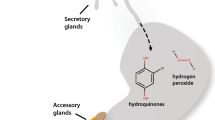
Abstract
Of the approximately one million described insect species, ground beetles (Coleoptera: Carabidae) have long captivated the attention of evolutionary biologists due to the diversity of defensive compounds they synthesize. Produced using defensive glands in the abdomen, ground beetle chemicals represent over 250 compounds including predator-deterring formic acid, which has evolved as a defensive strategy at least three times across Insecta. Despite being a widespread method of defense, formic acid biosynthesis is poorly understood in insects. Previous studies have suggested that the folate cycle of one-carbon (C1) metabolism, a pathway involved in nucleotide biosynthesis, may play a key role in defensive-grade formic acid production in ants. Here, we report on the defensive gland transcriptome of the formic acid-producing ground beetle Harpalus pensylvanicus. The full suite of genes involved in the folate cycle of C1 metabolism are significantly differentially expressed in the defensive glands of H. pensylvanicus when compared to gene expression profiles in the rest of the body. We also find support for two additional pathways potentially involved in the biosynthesis of defensive-grade formic acid, the kynurenine pathway and the methionine salvage cycle. Additionally, we have found an array of differentially expressed genes in the secretory lobes involved in the biosynthesis and transport of cofactors necessary for formic acid biosynthesis, as well as genes presumably involved in the detoxification of secondary metabolites including formic acid. We also provide insight into the evolution of the predominant gene family involved in the folate cycle (MTHFD) and suggest that high expression of folate cycle genes rather than gene duplication and/or neofunctionalization may be more important for defensive-grade formic acid biosynthesis in H. pensylvanicus. This provides the first evidence in Coleoptera and one of a few examples in Insecta of a primary metabolic process being co-opted for defensive chemical biosynthesis. Our results shed light on potential mechanisms of formic acid biosynthesis in the defensive glands of a ground beetle and provide a foundation for further studies into the evolution of formic acid-based chemical defense strategies in insects.





Similar content being viewed by others
Change history
31 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10886-021-01268-9
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Arndt E, Beutel RG, Will K (2016) Carabidae Latreille, 1802. In: Beutel RG, Leschen RAB (eds) handbook of zoology. Vol. IV Arthropoda: Insecta, part 38. Coleoptera, beetles. Vol 1: morphology and systematics (Archostemata, Adephaga, Myxophaga, Polyphaga partim). Walter de Gruyter, Berlin, pp 119–144
Arsovski AA, Pradinuk J, Guo XQ, Wang S, Adams KL (2015) Evolution of Cis-regulatory elements and regulatory networks in duplicated genes of Arabidopsis. Plant Physiol 169(4):2982–2091
Attygalle AB, Smedley SR, Meinwald J, Eisner T (1993) Defensive secretion of two notodontid caterpillars (Schizura unicornis, S. badia). J. Chem Ecol. 19(10):2089–2104
Attygalle AB, Wu X, Will KW (2006) Biosynthesis of Tiglic, Ethacrylic, and 2-Methylbutyric acids in a Carabid beetle, Pterostichus (Hypherpes) californicus. J Chem Ecol 33(5):963–970
Attygalle AB, Xu S, Moore W, McManus R, Gill A, Will K (2020) Biosynthetic origin of benzoquinones in the explosive discharge of the bombardier beetle Brachinus elongatulus. Sci Nat 107:26
Badawy AA (2017) Kynurenine pathway of tryptophan metabolism: regulatory and functional aspects. Int J Tryptophan Res 10:1–20
Ben-Sahra I, Hoxhaj G, Ricoult SJH, Asara JM, Manning BD (2016) mTORC1 induces purine synthesis through control of the mitochondrial tetrahydrofolate cycle. Science. 351(6274):728–733
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc 57(1):289–300
Bray NL, Pimentel H, Melsted P, Pachter L (2016) Near-optimal probabilistic RNA-seq quantification. Nat Biotechnol 34:525–527
Brosnan ME, Brosnan JT (2016) Formate: the neglected member of one-carbon metabolism. Annu Rev Nutr 36:369–388
Brückner A, Parker J (2020) Molecular evolution of gland cell types and chemical interactions in animals in animals. J. Exp. Biol 223:jeb211938
Carreras CW, Santi DV (1995) The catalytic mechanism and structure of thymidylate synthase. Annu Rev Biochem 64:721–762
Carroll N, Pangilinan F, Molloy AM, Troendle J, Mills JL, Kirke PN, Brody LC, Scott JM, Parle-McDermott A (2009) Analysis of the MTHFD1 promoter and risk of neural tube defects. Hum Genet 125(3):247–256
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552
Chen S, Zhou Y, Taru C, Gu J (2018) Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:i884–i890
Conesa A, Madrigal P, Tarazona S, Gomez-Cabrero D, Cervera A, McPherson SMW, Gaffney DJ, Elo LL, Zhang X, Mortazavi A (2016) A survey of best practices for RNA-seq data analysis. Genome Biol 17:13
Coppin CW, Jackson CJ, Sutherland T, Hart PJ, Devonshire AL, Russell RJ, Oakeshott JG (2012) Testing the evolvability of an insect carboxylesterase for the detoxification of synthetic pyrethroid insecticides. Insect Biochem Mol Biol 42(5):343–352
Deuve T (1993) L'abdomen et les genitalia des femelles de Coleopteres Adephaga. Mém Mus Natl Hist Nat 155:1–184
Di Pietro E, Sirois J, Tremblay ML, MacKenzie RE (2002) Mitochondrial NAD-dependent Methylenetetrahydrofolate dehydrogenase-Methenyltetrahydrofolate Cyclohydrolase is essential for embryonic development. Mol Cell Biol 22(12):4158–4166
Eddy SR (1998) Profile hidden Markov models. Bioinformatics 14(9):755–763
Eisner T, Aneshansley D, del Campo ML, Eisner M, Frank JH, Deyrup M (2006) Effect of bombardier beetle spray on a wolf spider: repellency and leg autotomy. Chemoecology 16:185–189
Eisner T, Meinwald J, Monro A, Ghent R (1961) Defence mechanisms of arthropods— I the composition and function of the spray of the whipscorpion, Mastigoproctus giganteus (Lucas) (Arachnida, Pedipalpida). J Insect Physiol 6(4):272–292
Feyereisen R (2020) Origin and evolution of the CYP4G subfamily in insects, cytochrome P450 enzymes involved in cuticular hydrocarbon synthesis. Mol Phylogenet Evol 143:106695
Finn RD, Clements J, Eddy SR (2011) HMMER web server: interactive sequence similarity searching. Nucleic Acids Res 39(web server issue):W29–W37
Forsyth DJ (1972) The structure of the pygidial defence glands of Carabidae (Coleoptera). Trans Zool Soc Lond 32:249–309
Forsyth DJ (1968) The structure of the defence glands in the Dytiscidae, Noteridae, Haliplidae and Gyrinidae (Coleoptera). Trans R Ent Soc Lond 120:158–181
Forsyth DJ (1970) The structure of the defence glands of the Cicindelidae, Amphizoidae, and Hygrobiidae (Insecta: Coleoptera). J Zool Lond 160:51–69
Fox JT, Stover PJ (2008) Folate-mediated one-carbon metabolism. Vitam Horm 79:1–44
Francke W, Dettner K (2005) Chemical signalling in beetles. In: Schulz S (ed) The chemistry of pheromones and other semiochemicals II. Springer, Berlin, pp 85–166
Froese DS, Fowler B, Baumgartner MR (2019) Vitamin B12, folate, and the methionine remethylation cycle- biochemistry, pathways, and regulation. J Inherit Metab Dis 42(4):673–685
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-seq data without a reference genome. Nat Biotechnol 29(7):644–652
Gustafsson R, Jemth AS, Gustafsson NM, Färnegårdh K, Loseva O, Wiita E, Bonagas N, Dahllund L, Llona-Minguez S, Häggblad M, Henriksson M, Andersson Y, Homan E, Helleday T, Stenmark P (2017) Crystal structure of the emerging Cancer target MTHFD2 in complex with a substrate-based inhibitor. Cancer Res 77(4):937–948
Haas BJ, Papanicolaou A, Yassour M, Grabherr M, Blood PD, Bowden J, Couger MB, Eccles D, Li B, Lieber M, Macmanes MD, Ott M, Orvis J, Pochet N, Strozzi F, Weeks N, Westerman R, William T, Dewey CN, Henschel R, Leduc RD, Friedman N, Regev A (2013) De novo transcript sequence reconstruction from RNA-seq using the trinity platform for reference generation and analysis. Nat Protoc 8(8):1494–1512
Han Q, Robinson H, Li J (2012) Biochemical identification and crystal structure of kynurenine formamidase from Drosophila melanogaster. Biochem J 446(2):253–260
Haque MR, Higashiura A, Nakagawa A, Hirowatari A, Furuya S, Yamamoto K (2019) Molecular structure of a 5,10-methylenetetrahydrofolate dehydrogenase from the silkworm Bombyx mori. FEBS Open Bio 9(4):618–628
Hartman SC, Buchanan JM (1959) Biosynthesis of the purines. XXVI The identification of the formyl donors of the transformylation reactions. J Biol Chem 234(7):1812–1816
Hefetz A, Blum MS (1978a) Biosynthesis and accumulation of formic acid in the poison gland of the carpenter ant Camponotus pensylvanicus. Science 201(4354):454–455
Hefetz A, Blum MS (1978b) Biosynthesis of formic acid by the poison glands of formicine ants. Biochim Biophys Acta 543(4):484–496
Ivarsson P, Henrikson BI, Stenson JAE (1996) Volatile substances in the pygidial secretion of gyrinid beetles (Coleoptera: Gyrinidae). Chemoecology 7(4):191–193
Jackson CJ, Liu JW, Carr PD, Younus F, Coppin C, Meirelles T, Lethier M, Pandey G, Ollis DL, Russell RJ, Weik M, Oakeshott JG (2013) Structure and function of an insect α-carboxylesterase (αEsterase7) associated with insecticide resistance. Proc Natl Acad Sci U S A 110(25):10177–10182
Jaffe JJ, Chrin LR (1979) De novo syntesis of methionine in normal and brugia-infected Aedes aegypti. J Parasitol 65(4):550–554
Jensen LJ, Julien P, Kuhn M, von Mering C, Muller J, Doerks T, Bork P (2008) eggNOG: automated construction and annotation of orthologous groups of genes. Nucleic Acids Res 36(Database issue):D250–D254
Kalyaanamoorthy S, Minh BQ, Wong TKF, von Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14(6):587–589
Kanai M, Funakoshi H, Takahashi H, Hayakawa T, Mizuno S, Matsumoto K, Nakamura T (2009) Tryptophan 2,3-dioxygenase is a key modulator of physiological neurogenesis and anxiety-related behavior in mice. Mol Brain 2:8
Kanehisa K, Kawazu K (1982) Fatty acid components of the defensive substances in acid-secreting carabid beetles. Appl Entomol Zool 17(4):460–466
Kanehisa K, Kawazu K (1985) Differences in neutral components of the defensive secretion in formic acid-secreting carabid beetles. Appl Entomol Zool 20(3):299–304
Kanehisa K, Murase M (1977) Comparative study of the pygidial defensive systems of carabid beetles. Appl Entomol Zool 12:225–235
Kanehisa M, Goto S, Sato Y, Furumichi M, Tanabe M (2012) KEGG for integration and interpretation of large-scale molecular datasets. Nucleic Acids Res 40:D109–D114
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30(4):772–780
Krogh A, Larsson B, von Heijne G, Sonnhammer EL (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305(3):567–580
Kück P, Mayer C, Wägele JW, Misof B (2012) Long Branch effects distort maximum likelihood phylogenies in simulations despite selection of the correct model. PLoS One 7(5):e36593
Law CW, Chen Y, Shi W, Smyth GK (2014) Voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol 15:R29
Lečić S, Ćurčić S, Vujisić L, Ćurčić B, Ćurčić N, Nikolić Z, Anđelković B, Milosavljević S, Tešević V, Makarov S (2014) Defensive secretions in three ground-beetle species (Insecta: Coleoptera: Carabidae). Ann Zool Fenn 51:285–300
Li T, White KP (2003) Tissue-specific gene expression and ecdysone-regulated genomic networks in Drosophila. Dev Cell 5(1):59–72
Locher KP (2009) Structure and mechanism of ATP-binding cassette transporters. Philos Trans R Soc Lond Ser B Biol Sci 2364(1514):239–245
Long HK, Prescott SL, Wysocka J (2016) Ever-changing landscapes: transcriptional enhancers in development and evolution. Cell. 167(5):1170–1187
MacFarlane AJ, Perry CA, Girnary HH, Gao D, Allen RH, Stabler SP, Shane B, Stover PJ (2009) Mthfd1 is an essential gene in mice and alters biomarkers of impaired one-carbon metabolism. J Biol Chem 284(3):1533–1539
McCullough T (1966) Compounds found in the defensive scent glands of Harpalus caliginosus (Coleoptera: Carabidae). Ann Entomol Soc Am 59(5):1020–1021
Mehler AH, Knox WE (1950) The conversion of tryptophan to kynurenine in liver. II The enzymatic hydrolysis of formylkynurenine. J Biol Chem 187(1):431–438
Meiser J, Tumanov S, Maddocks O, Labuschagne CF, Athineos D, Van Den Broek N, Mackay GM, Gottlieb E, Blyth K, Vousden K, Kamphorst JJ, Vazquez A (2016) Serine one-carbon catabolism with formate overflow. Sci Adv 2(10):e1601273
Moore BP, Wallbank BE (1968) Chemical composition of the defensive secretion in carabid beetles and its importance as a taxonomic character. Proc R Ent Soc Lond 37(5–6):62–72
Moore BP (1979) Chemical defense in Carabids and its bearing on phylogeny. Carabid Beetles:193–203
Mulder GJ (1992) Glucuronidation and its role in regulation of biological activity of drugs. Annu Rev Pharmacol Toxicol 32:25–49
Nguyen LT, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32(1):268–274
Parkhitko AA, Jouandin P, Mohr SE, Perrimon N (2019) Methionine metabolism and methyltransferases in the regulation of aging and lifespan extension across species. Aging Cell 18(6):e13034
Peschke K, Eisner T (1987) Defensive secretion of the tenebrionid beetle Blaps mucronata physical and chemical determinants of effectiveness. J Comp Physiol A 161(3):377–388
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786
Philippe H, Zhou Y, Brinkmann H, Rodrigue N, Delsuc F (2005) Heterotachy and long-branch attraction in phylogenetics. BMC Evol Biol 5:50
Punta M, Coggill PC, Eberhardt RY, Mistry J, Tate J, Boursnell C, Pang N, Forslund K, Ceric G, Clements J, Heger A, Holm L, Sonnhammer EL, Eddy SR, Bateman A, Finn RD (2012) The Pfam protein families database. Nucleic Acids Res 40:D290–D301
Qiu Y, Tittiger C, Wicker-Thomas C, Le Goff G, Young S, Wajnberg E, Fricaux T, Taquet N, Blomquist GJ, Feyereisen R (2012) An insect-specific P450 oxidative decarbonylase for cuticular hydrocarbon biosynthesis. Proc Natl Acad Sci U S A 109(37):14858–11463
Quistgaard EM, Löw C, Guettou F, Nordlund P (2016) Understanding transport by the major facilitator superfamily (MFS): structures pave the way. Nat Rev Mol Cell Biol 17:123–132
Ranwez V, Harispe S, Delsuc F, Douzery EJP (2011) MACSE: multiple alignment of coding SEquences accounting for Frameshifts and stop codons. PLoS One 6(9):e22594
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47
Rork AM, Mikó I, Renner T (2019) Pygidial glands of Harpalus pensylvanicus (Coleoptera: Carabidae) contain resilin-rich structures. Arthropod Struct Dev 44(12):1069–1083
Rork AM, Renner T (2018) Carabidae Semiochemistry: current and future directions. J Chem Ecol 49:19–25
Rossini C, Attygalle AB, González A, Smedley SR, Eisner M, Meinwald J, Eisner T (1977) Defensive production of formic acid (80%) by a carabid beetle (Galerita lecontei). Proc Natl Acad Sci U S A 94:6792–6797
Roth LM, Eisner T (1962) Chemical defenses of arthropods. Annu Rev Entomol 7:107–136
Savarse TM, Shih-Hsi C, Ming-Yu C, Parks RE (1985) 5′-Deoxy-5′-methylthioadenosine phosphorylase—III: role of the enzyme in the metabolism and action of 5′-halogenated adenosine analogs. Biochem Pharmacol 34(3):361–367
Schildknecht H, Holoubek K (1961) Die Bombardierkäfer und ihre Explosionschemie V. Mitteilung über Insekten-Abwehrstoffe. Angew Chem 73(1):1–7
Schildknecht H, Maschwitz U, Winkler H (1968a) Zur Evolution der Carabiden-Wehrdrüsensekrete. Über Arthropoden-Abwehrstoffe XXXII Naturwissenschaften 55:112–117
Schildknecht H, Weis KH (1961) Die chemische natur des wehrsekretes von Pseudophonus pubescens und Ps. griseus. VIII. Mitteilung über insektenabwehrstoffe. Z Naturforsch B 16:361–363
Schildknecht H, Winkler H, Maschwitz U (1968b) Vergleichend chemische Untersuchungen der Inhaltsstoffe der Pygidialwehrblasen von Carabiden. Über Arthropoden-Abwehrstoffe XXXI Z Naturforsch B 23:637–644
Schildknecht H (1961) Über Insekten-und Pflanzenabwehrstoffe, ihre Isolierung und Aufklärung. Angew Chem 73(17-18):629
Schildknecht H (1964) Defensive substances of the arthropods, their isolation and identification. Angew Chem Int Ed 3(2):73–82
Schildknecht H (1970) The defensive chemistry of land and water beetles. Angew Chem Int Ed 9(1):1–9
Schuler MA, Berenbaum MR (2013) Structure and function of cytochrome P450s in insect adaptation to natural and synthetic toxins: insights gained from molecular modeling. J Chem Ecol 39(9):1232–1245
Schurch NJ, Schofield P, Gierliński M, Cole C, Sherstnev A, Singh V, Wrobel N, Gharbi K, Simpson GG, Owen-Hughes T, Blaxter M, Barton GJ (2016) How many biological replicates are needed in an RNA-seq experiment and which differential expression tool should you use? RNA. 22(6):839–851
Scott JG, Wen Z (2001) Cytochromes P450 of insects: the tip of the iceberg. Pest Manag Sci 57(10):958–967
Sekowska A, Ashida H, Danchin A (2019) Revisiting the methionine salvage pathway and its paralogues. Microb Biotechnol 12(1):77–97
Simāo FA, Waterhouse RM, Ioannidis P, Kriventseva EV, Zdobnov EM (2015) BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics. 31(19):3210–3212
Smith JD, McManus KF, Fraser HB (2013) A novel test for selection on cis-regulatory elements reveals positive and negative selection acting on mammalian transcriptional enhancers. Mol Biol Evol 30(11):2509–2518
Smith MD, Wertheim JO, Weaver S, Murrell B, Scheffler K, Kosakovsky Pond SL (2015) Less is more: an adaptive branch-site random effects model for efficient detection of episodic diversifying selection. Mol Biol Evol 32(5):1342–1353
Sonawane AR, Platig J, Fagny M, Chen CY, Paulson JN, Lopes-Ramos CM, DeMeo DL, Quackenbush J, Glass K, Kuijjer ML (2017) Understanding tissue-specific gene regulation. Cell Rep 21(4):1077–1088
Steuernagel L, Meckbach C, Heinrich F, Zeidler S, Schmitt AO, Gültas M (2019) Computational identification of tissue-specific transcription factor cooperation in ten cattle tissues. PLoS One 14(5):e021647
Storey JD (2002) A direct approach to false discovery rates. J R Stat Soc Series B 64:479–498
Talavera G, Castresana J (2007) Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst Biol 56:564–577
Tautz D, Domazet-Lošo T (2011) The evolutionary origin of orphan genes. Nat. Rev. Genet. 12(10):692–702
The Gene Ontology Consortium (2000) Gene ontology: tool for the unification of biology. Nat Genet 25:25–29
Wagner W, Breksa AP 3rd, Monzingo AF, Appling DR, Robertus JD (2005) Kinetic and structural analysis of active site mutants of monofunctional NAD-dependent 5,10-methylenetetrahydrofolate dehydrogenase from Saccharomyces cerevisiae. Biochemistry 44(39):13163–13171
West MG, Barlowe CK, Appling DR (1993) Cloning and characterization of the Saccharomyces cerevisiae gene encoding NAD-dependent 5,10-methylenetetrahydrofolate dehydrogenase. J Biol Chem 268(1):153–160
Wheeler TJ, Eddy SR (2013) Nhmmer: DNA homology search with profile HMMs. Bioinformatics 29(19):2487–2489
Will KW, Attygalle AB, Herath K (2000) New defensive chemical data for ground beetles (Coleoptera: Carabidae): interpretations in a phylogenetic framework. Biol J Linn Soc 71:459–481
Will KW, Gill AS, Lee H, Attygalle AB (2010) Quantification and evidence for mechanically metered release of pygidial secretions in formic acid-producing carabid beetles. J Insect Sci 10:1–17
Wilson EO, Regnier FE (1971) The evolution of the alarm-defense system in the formicine ants. Am Nat 105(943):279–289
Wray GA (2007) The evolutionary significance of cis-regulatory mutations. Nat Rev Genet 8:206–216
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14
Acknowledgements
We thank Yuka Imamura and Kimberly Moran for their assistance with RNA extraction, library preparation, and RNA-Seq. We would also like to thank Wendy Moore, Reilly McManus, Kipling Will, Sean Perez, and Aman Gill for thoughtful discussion on Carabidae biology and chemical defense mechanisms. Additionally, we thank Wendy Moore, Kipling Will, Kylie Bocklund, Chloe P. Drummond, Katie Nolan, Kadeem Gilbert, Arthi Bala, Dana Roberts, Jonah Ulmer, and István Mikó for discussion and critique of this project, its results, and interpretation. This work was supported by the National Science Foundation [DEB 1762760, 1556898].
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised: Modifications have been made to the sentences under Methods and figures 1-5 captions.
Rights and permissions
About this article
Cite this article
Rork, A.M., Xu, S., Attygalle, A. et al. Primary Metabolism co-Opted for Defensive Chemical Production in the Carabid Beetle, Harpalus pensylvanicus. J Chem Ecol 47, 334–349 (2021). https://doi.org/10.1007/s10886-021-01253-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01253-2