Abstract
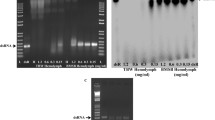
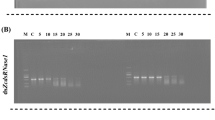
The southern green stink bug, Nezara viridula, is a worldwide pest of many important crops. RNA from N. viridula nymphs and adults was sequenced and assembled into a transcriptome. Orthologs of genes coding for proteins involved in RNA interference (RNAi) pathways and potential target genes for RNAi-mediated control of this pest were identified by searching the transcriptome. The watery saliva, lumen and hemolymph collected from N. viridula showed lower dsRNase activity than that in the lumen and hemolymph collected from the fall armyworm, Spodoptera frugiperda, which is refractory to RNAi. Seven housekeeping genes, 60S RP L12, 40S RP S17, 18S rRNA, EF1-α, Actin, Ubiquitin and β-Tubulin, were evaluated across developmental stages and dsRNA treatments and identified 60S RP L12 and 40S RP S17 as the best reference genes for use in reverse transcriptase quantitative PCR (RT-qPCR). One microgram of dsRNA targeting the orthologs of seven known RNAi target genes (SNF7, IAP1, IAP2, IAP5, PP1, GPCR and ATPase1) was tested in N. viridula. Three out of seven dsRNAs tested induced 67–100% mortality. Injection of dsRNA targeting SNF7 into N. viridula adults caused 100% mortality, while feeding dsSNF7 induced 60% mortality in both adults and nymphs. Delivery of dsRNA by either feeding or injection efficiently knocked down target gene expression. These data demonstrate that RNAi pathway components are present in N. viridula and delivering dsRNA by either injection or feeding induces silencing of target gene expression and mortality, suggesting that RNAi-based methods could be developed to manage this pest.




Similar content being viewed by others
References
Bansal R, Mittapelly P, Chen Y, Mamidala P, Zhao C, Michel A (2016) Quantitative RT-PCR gene evaluation and RNA interference in the Brown Marmorated Stink Bug. PLoS ONE 11(5):e0152730. https://doi.org/10.1371/journal.pone.0152730
Baum JA, Bogaert T, Clinton W et al (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25(11):1322–1326. https://doi.org/10.1038/nbt1359
Bolognesi R, Ramaseshadri P, Anderson J et al (2012) Characterizing the mechanism of action of double-stranded RNA activity against western corn rootworm (Diabrotica virgifera virgifera LeConte). PLoS ONE 7(10):e47534. https://doi.org/10.1371/journal.pone.0047534
Bustin SA, Benes V, Garson JA et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55(4):611–622. https://doi.org/10.1373/clinchem.2008.112797
Butte AJ, Dzau VJ, Glueck SB (2001) Further defining housekeeping, or “maintenance,” genes Focus on “A compendium of gene expression in normal human tissues.” Phys Genom 7(2):95–96. https://doi.org/10.1152/physiolgenomics.2001.7.2.95
Castellanos NL, Smagghe G, Sharma R, Oliveira EE, Christiaens O (2019) Liposome encapsulation and EDTA formulation of dsRNA targeting essential genes increase oral RNAi-caused mortality in the Neotropical stink bug. Euschistus Heros Pest Manag Sci 75(2):537–548. https://doi.org/10.1002/ps.5167
Christiaens O, Swevers L, Smagghe G (2014) DsRNA degradation in the pea aphid (Acyrthosiphon pisum) associated with lack of response in RNAi feeding and injection assay. Peptides 53:307–314. https://doi.org/10.1016/j.peptides.2013.12.014
Davis-Vogel C, Van Allen B, Van Hemert JL, Sethi A, Nelson ME, Sashital DG (2018) Identification and comparison of key RNA interference machinery from western corn rootworm, fall armyworm, and southern green stink bug. PLoS ONE 13(9):e0203160. https://doi.org/10.1371/journal.pone.0203160
Esquivel JF, Musolin DL, Jones WA, Rabitsch W, Greene JK, Toews MD, Schwertner CF, Grazia J, McPherson RM (2018) Nezara viridula (L.). Invasive stink bugs and related species (Pentatomoidea): biology, higher systematics, semiochemistry, and management, vol 17. CRC Press, Boca Raton, pp 351–423
Gao FB, Brenman JE, Jan LY, Jan YN (1999) Genes regulating dendritic outgrowth, branching, and routing in Drosophila. Genes Dev 13(19):2549–2561. https://doi.org/10.1101/gad.13.19.2549
Ghosh SK, Hunter WB, Park AL, Gundersen-Rindal DE (2017) Double strand RNA delivery system for plant-sap-feeding insects. PLoS ONE 12(2):e0171861. https://doi.org/10.1371/journal.pone.0171861
Ghosh SKB, Hunter WB, Park AL, Gundersen-Rindal DE (2018) Double-stranded RNA oral delivery methods to induce RNA interference in Phloem and plant-sap-feeding Hemipteran Insects. J Vis Exp 135:57390. https://doi.org/10.3791/57390
Gurusamy D, Howell JL, Chereddy SCRR, Koo J, Palli SR (2020) Transport of orally delivered dsRNA in southern green stink bug, Nezara viridula. Arch Insect Biochem Phys 104:e21692. https://doi.org/10.1002/arch.21692
Howell JL, Mogilicherla K, Gurusamy D, Palli SR (2020) Development of RNAi methods to control the harlequin bug, Murgantia histrionica. Arch Insect Biochem Phys. https://doi.org/10.1002/arch.21690
Huvenne H, Smagghe G (2010) Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: a review. J Insect Phys 56(3):227–235. https://doi.org/10.1016/j.jinsphys.2009.10.004
Kim DW, Sung H, Shin D et al (2011) Differential physiological roles of ESCRT complexes in Caenorhabditis elegans. Mol Cells 31(6):585–592. https://doi.org/10.1007/s10059-011-1045-z
Lavore A, Perez-Gianmarco L, Esponda-Behrens N et al (2018) Nezara viridula (Hemiptera: Pentatomidae) transcriptomic analysis and neuropeptidomics. Sci Rep 8(1):17244. https://doi.org/10.1038/s41598-018-35386-4
Lee JA, Beigneux A, Ahmad ST, Young SG, Gao FB (2007) ESCRT-III dysfunction causes autophagosome accumulation and neurodegeneration. Curr Biol 17(18):1561–1567. https://doi.org/10.1016/j.cub.2007.07.029
Liu S, Lomate PR, Bonning BC (2018) Tissue-specific transcription of proteases and nucleases across the accessory salivary gland, principal salivary gland and gut of Nezara viridula. Insect Biochem Mol Biol 103:36–45. https://doi.org/10.1016/j.ibmb.2018.10.003
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lomate PR, Bonning BC (2016) Distinct properties of proteases and nucleases in the gut, salivary gland and saliva of southern green stink bug. Nezara viridula Sci Rep 6:27587. https://doi.org/10.1038/srep27587
Lu Y, Chen M, Reding K, Pick L (2017) Establishment of molecular genetic approaches to study gene expression and function in an invasive hemipteran. Halyomorpha Halys Evodevo 8:15. https://doi.org/10.1186/s13227-017-0078-6
Mogilicherla K, Howell JL, Palli SR (2018) Improving RNAi in the Brown Marmorated Stink Bug: identification of target genes and reference genes for RT-qPCR. Sci Rep 8(1):3720. https://doi.org/10.1038/s41598-018-22035-z
O’Neal ST, Anderson TD, Wu-Smart JY (2018) Interactions between pesticides and pathogen susceptibility in honey bees. Curr Opin Insect Sci 26:57–62. https://doi.org/10.1016/j.cois.2018.01.006
Palli SR (2014) RNA interference in Colorado potato beetle: steps toward development of dsRNA as a commercial insecticide. Curr Opin Insect Sci 6:1–8. https://doi.org/10.1016/j.cois.2014.09.011
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26(6):509–515. https://doi.org/10.1023/B:BILE.0000019559.84305.47
Puntener W (1981) Manual for Field Trials in Plant Protection. Ciba-Geigy Limited, Basle, Switzerland, p 205
Ramaseshadri P, Segers G, Flannagan R et al (2013) Physiological and cellular responses caused by RNAi-mediated suppression of Snf7 orthologue in western corn rootworm (Diabrotica virgifera virgifera) larvae. PLoS ONE 8(1):e54270. https://doi.org/10.1371/journal.pone.0054270
Riga M, Denecke S, Livadaras I, Geibel S, Nauen R, Vontas J (2020) Development of efficient RNAi in Nezara viridula for use in insecticide target discovery. Arch Insect Biochem Phys 103(3):e21650. https://doi.org/10.1002/arch.21650
Sharma R, Christiaens O, Taning CN, Smagghe G (2020) RNAi-mediated mortality in southern green stinkbug Nezara viridula by oral delivery of dsRNA. Pest Manag Sci. https://doi.org/10.1002/ps.6017
Shukla JN, Kalsi M, Sethi A et al (2016) Reduced stability and intracellular transport of dsRNA contribute to poor RNAi response in lepidopteran insects. RNA Biol 13(7):656–669. https://doi.org/10.1080/15476286.2016.1191728
Singh IK, Singh S, Mogilicherla K, Shukla JN, Palli SR (2017) Comparative analysis of double-stranded RNA degradation and processing in insects. Sci Rep 7(1):17059. https://doi.org/10.1038/s41598-017-17134-2
Sweeney NT, Brenman JE, Jan YN, Gao FB (2006) The coiled-coil protein shrub controls neuronal morphogenesis in Drosophila. Curr Biol 16(10):1006–1011. https://doi.org/10.1016/j.cub.2006.03.067
Terenius O, Papanicolaou A, Garbutt JS et al (2011) RNA interference in Lepidoptera: an overview of successful and unsuccessful studies and implications for experimental design. J Insect Physiol 57(2):231–245. https://doi.org/10.1016/j.jinsphys.2010.11.006
Tomoyasu Y, Miller SC, Tomita S, Schoppmeier M, Grossmann D, Bucher G (2008) Exploring systemic RNA interference in insects: a genome-wide survey for RNAi genes in Tribolium. Genome Biol 9(1):R10. https://doi.org/10.1186/gb-2008-9-1-r10
Urquhart W, Mueller GM, Carleton S et al (2015) A novel method of demonstrating the molecular and functional equivalence between in vitro and plant-produced double-stranded RNA. Regul Toxicol Pharmacol 73(2):607–612. https://doi.org/10.1016/j.yrtph.2015.09.004
Vaccari T, Rusten TE, Menut L et al (2009) Comparative analysis of ESCRT-I, ESCRT-II and ESCRT-III function in Drosophila by efficient isolation of ESCRT mutants. J Cell Sci 122:2413–2423. https://doi.org/10.1242/jcs.046391
Vandesompele J, De Preter K, Pattyn F et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7):RESEARCH0034. https://doi.org/10.1186/gb-2002-3-7-research0034
Winter V, Hauser MT (2006) Exploring the ESCRTing machinery in eukaryotes. Trends Plant Sci 11(3):115–123. https://doi.org/10.1016/j.cub.2007.07.029
Xie F, Xiao P, Chen D, Xu L, Zhang B (2012) miRDeepFinder: a miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol Biol 80:75–84. https://doi.org/10.1007/s11103-012-9885-2
Yoon JS, Gurusamy D, Palli SR (2017) Accumulation of dsRNA in endosomes contributes to inefficient RNA interference in the fall armyworm, Spodoptera frugiperda. Insect Biochem Mol Biol 90:53–60. https://doi.org/10.1016/j.ibmb.2017.09.011
Yoon JS, Mogilicherla K, Gurusamy D, Chen X, Chereddy SCRR, Palli SR (2018) Double-stranded RNA binding protein, Staufen, is required for the initiation of RNAi in coleopteran insects. Proc Natl Acad Sci USA 115(33):8334–8339. https://doi.org/10.1073/pnas.1809381115
Zhang J, Khan SA, Hasse C, Ruf S, Heckel DG, Bock R (2015) Pest control full crop protection from an insect pest by expression of long double-stranded RNAs in plastids. Science 347(6225):991–994. https://doi.org/10.1126/science.1261680
Zhang J, Khan SA, Heckel DG, Bock R (2017) Next-Generation insect-resistant plants: RNAi-mediated crop protection. Trends Biotechnol 35(9):871–882. https://doi.org/10.1016/j.tibtech.2017.04.009
Zhu F, Xu J, Palli R, Ferguson J, Palli SR (2011) Ingested RNA interference for managing the populations of the Colorado potato beetle, Leptinotarsa decemlineata. Pest Manag Sci 67(2):175–182. https://doi.org/10.1002/ps.2048
Acknowledgements
The authors thank Dr. Amit Sethi from Corteva Agriscience, Dr. Guo-Qing Tang from Syngenta and Dr. Sven Geibel from Bayer Crop Science for discussions and advice.
Funding
This material is based upon work supported by the National Science Foundation I/UCRC, the Center for Arthropod Management Technologies under Grant IIP-1821936 and by industry partners and the USDA/NIFA (under Hatch Project 2351177000).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Guy Smagghe.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gurusamy, D., Howell, J.L., Chereddy, S.C.R.R. et al. Improving RNA interference in the southern green stink bug, Nezara viridula. J Pest Sci 94, 1461–1472 (2021). https://doi.org/10.1007/s10340-021-01358-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-021-01358-3