Abstract
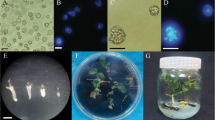
An effective culture system for producing embryos was developed from isolated microspores of sweet pepper (Capsicum annuum L.). The present work aimed to define exogenous arabinogalactan proteins (AGPs) on the embryogenesis of sweet pepper under the growth environments of donor plants. The experiments were performed with the cultivars Arancia, California wonder, Californium, Camelot, Inspiration, Plato, and Taranto under three different growth environments of the donor plants. The impacts of the different numbers of sweet pepper ovaries (0, 5, 10, and 15) were evaluated on microspore embryogenesis of cultivars in a factorial design with five replications. The simultaneous application of 10 ovaries in stress (sucrose starvation; temperature) and induction media resulted in the highest embryogenesis of the seven aforementioned cultivars under field conditions. Also, the impacts of 0, 80, 120, and 200 mg L−1 gum arabic were studied on embryogenesis in stress and induction media. The treatments containing 120 mg L−1 gum arabic showed the highest embryogenesis across all cultivars, except the California Wonder cultivar in the field conditions. Also, the simultaneous impact of the number of ovaries and concentrations of gum arabic on embryogenesis were studied. A combination of ten ovaries and 120 mg L−1 gum arabic generated the highest embryogenesis in the majority of cultivars. The results revealed that ovaries of sweet pepper and gum arabic had a significant impact on the efficiency of microspore embryogenesis in sweet pepper plants under field conditions.



Similar content being viewed by others
References
Akiyama Y, Eda S, Katō K (1984) Gum Arabic is a kind of arabinogalactan–protein. Agric Biol Chem 48:235–237. https://doi.org/10.1080/00021369.1984.10866126
Broughton S (2008) Ovary co-culture improves embryo and green plant production in anther culture of Australian spring wheat (Triticum aestivum L.). Plant Cell Tiss Org Cult 95:185–195. https://doi.org/10.1007/s11240-008-9432-7
Chauhan H, Khurana P (2011) Use of doubled haploid technology for development of stable drought tolerant bread wheat (Triticum aestivum L.) transgenics. Plant Biotechnol J 9:408–417. https://doi.org/10.1111/j.1467-7652.2010.00561.x
Cheng Y, Ma RL, Jiao YS, Qiao N, Li TT (2013) Impact of genotype, plant growth regulators and activated charcoal on embryogenesis induction in microspore culture of pepper (Capsicum annuum L.). S Afr J Bot 88:306–309. https://doi.org/10.1016/j.sajb.2013.08.012
Cistué L, Soriano M, Castillo AM, Valles MP, Sanz JM, Echavarri B (2006) Production of doubled haploids in durum wheat (Triticum turgidum L.) through isolated microspore culture. Plant Cell Rep 25:257–264. https://doi.org/10.1007/s00299-005-0047-8
Corral-Martínez P, Seguí-Simarro JM (2014) Refining the method for eggplant microspore culture: effect of abscisic acid, epibrassinolide, polyethylene glycol, naphthaleneacetic acid, 6-benzylaminopurine and arabinogalactan proteins. Euphytica 195:369–382. https://doi.org/10.1007/s10681-013-1001-4
Devaux P, Pickering R (2005) Haploids in the improvement of Poaceae. Biotechnology in agriculture and forestry, vol 56. Springer, Berlin, pp 215–242. https://doi.org/10.1007/3-540-26889-8_11
Doležel J, Binarova P, Lucretti S (1989) Analysis of nuclear DNA content in plant cells by flow cytometry. Biol Plant 31:113–120. https://doi.org/10.1007/BF02907241
Dunwell JM, Perry E (1973) The influence of in vivo growth conditions of Nicotiana tabacum plants on the in vitro embryogenetic potential of their anthers. John Innes Inst Rep 64:69–70
Ellis M, Egelund J, Schultz CJ, Bacic A (2010) Arabinogalactan-proteins: key regulators at the cell surface? Plant Physiol 153:403–419. https://doi.org/10.1104/pp.110.156000
El-Tantawy AA, Solís MT, Da Costa ML, Coimbra S, Risueño MC, Testillano PS (2013) Arabinogalactan protein profiles and distribution patterns during microspore embryogenesis and pollen development in Brassica napus. Plant Reprod 26:1–13. https://doi.org/10.1007/s00497-013-0217-8
Ercan N, Sensoy FA, Sensoy AS (2006) Influence of growing season and donor plant age on anther culture response of some pepper cultivars (Capsicum annuum L.). Sci Hortic 110:16–20. https://doi.org/10.1016/j.scienta.2006.06.007
Eudes F, Amundsen E (2005) Isolated microspore culture of Canadian 69 triticale cultivars. Plant Cell Tiss Org Cult 82:233–241. https://doi.org/10.1007/s11240-005-0867-9
Ferrie AMR, Caswell KL (2011) Isolated microspore culture techniques and recent progress for haploid and doubled haploid plant production. Plant Cell Tiss Org Cult 104:301–309. https://doi.org/10.1007/s11240-010-9800-y
Ferrie AMR, Mollers C (2011) Haploid and doubled haploids in Brassica spp. for genetic and genomic research. Plant Cell Tiss Org Cult 104:375–386. https://doi.org/10.1007/s11240-010-9831-4
Germana MA (2011) Gametic embryogenesis and haploid technology as valuable support to plant breeding. Plant Cell Rep 30:839–857
Germana MA, Lambardi M (2016) In vitro embryogenesis in higher plants in vitro embryogenesis in higher plants. Humana Press, New York. https://doi.org/10.1007/978-1-4939-3061-6
Irikova T, Grozeva S, Rodeva V (2011) Anther culture in pepper (Capsicum annuum L.) in vitro. Acta Physiol Plant 33:1559–1570. https://doi.org/10.1007/s11738-011-0736-6
Jain SM, Sopory SK, Veilleux RE (1996) In vitro haploid production in higher plants. Volume 2: Applications, vol 24. Springer Science & Business Media, Berlin. https://doi.org/10.1007/978-94-017-1860-8
Kim M, Jang IC, Kim JA, Park EJ, Yoon M, Lee Y (2008) Embryogenesis and plant regeneration of hot pepper (Capsicum annuum L.) through isolated microspore culture. Plant Cell Rep 27:425–434. https://doi.org/10.1007/s00299-007-0442-4
Kim M, Park EJ, An D, Lee Y (2013) High-quality embryo production and plant regeneration using a two-step culture system in isolated microspore cultures of hot pepper (Capsicum annuum L.). Plant Cell Tiss Org Cult 112:191–201. https://doi.org/10.1007/s11240-012-0222-x
Kothari SL, Joshi A, Kachhwaha S, Ochoa-Alejo N (2010) Chilli peppers: a review on tissue culture and transgenesis. Biotechnol Adv 28:35–48. https://doi.org/10.1016/j.biotechadv.2009.08.005
Kristiansen K, Andersen SB (1993) Effect of donor plant-temperature, photoperiod, and age on anther culture response of Capsicum annuum L. Euphytica 67:105–109. https://doi.org/10.1007/BF00022732
Lantos C, Juhász AG, Somogyi G, Ötvös K, Vági P, Mihály R, Kristóf Z, Somogyi N, Pauk J (2009) Improvement of isolated microspore culture of pepper (Capsicum annuum L.) via co-culture with ovary tissues of pepper or wheat. Plant Cell Tiss Org Cult 97:285–293. https://doi.org/10.1007/s11240-009-9527-9
Lantos C, Juhász AG, Vági P, Mihály R, Kristóf Z, Pauk J (2012) Androgenesis induction in microspore culture of sweet pepper (Capsicum annuum L.). Plant Biotechnol Rep 6:123–132. https://doi.org/10.1007/s11816-011-0205-0
Letarte J, Simon E, Miner M, Kasha KJ (2006) Arabinogalactans, and arabinogalactan-proteins induce embryogenesis in wheat (Triticum aestivum L.) microspore culture. Plant Cell Rep 24:691–698. https://doi.org/10.1007/s00299-005-0013-5
Li H, Devaux P (2001) Enhancement of microspore culture efficiency of recalcitrant barley genotypes. Plant Cell Rep 20:475–481. https://doi.org/10.1007/s002990100368
Li WL, Liu Y, Douglas CJ (2017) Role of glycosyltransferases in pollen wall primexine formation and exine patterning. Plant Physiol 173:167–182. https://doi.org/10.1104/pp.16.00471
Makowska K, Kałużniak M, Oleszczuk S, Zimny J, Czaplicki A, Konieczny R (2017) Arabinogalactan proteins improve plant regeneration in barley (Hordeum vulgare L.) anther culture. Plant Cell Tiss Org Cult 131:247–257. https://doi.org/10.1007/s11240-017-1280-x
Mejza SJ, Morgant V, DE DB (1993) Wong JR Plant regeneration from isolated microspores of Triticum aestivum. Plant Cell Rep 12:149–153. https://doi.org/10.1007/BF00239096
Monnier M (1995) Culture of Zygotic Embryos. In: Thorpe TA (ed) In vitro embryogenesis in plants. Current Plant Science and Biotechnology in Agriculture, vol 20. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-0485-2_4
Nitsch C, Nitsch JP (1967) The induction of flowering in vitro in stem segments of Plumbago indica L. I. The production of vegetative buds. Planta 72:355–370. https://doi.org/10.1007/BF00390147
Lichter R (1981) Anther culture of Brassica napus in a liquid culture medium. Z für Pflanzenphysiol 103:229–237. https://doi.org/10.1016/S0044-328X(81)80155-9
Paire A, Devaux P, Lafitte C, Dumas C, Matthys-Rochon E (2003) Proteins produced by barley microspores and their derived androgenic structures promote in vitro zygotic maize embryo formation. Plant Cell Tiss Org Cult 73:167–176. https://doi.org/10.1023/A:1022805623167
Phillips GO (2009) Molecular association and function of arabinogalactan protein complexes from tree exudates. Struct Chem 20:309–315. https://doi.org/10.1007/s11224-009-9422-3
Pilarska M, Knox JP, Konieczny R (2013) Arabinogalactan-protein and pectin epitopes in relation to an extracellular matrix surface network and somatic embryogenesis and callogenesis in Trifolium nigrescens Viv. Plant Cell Tiss Org Cult 115:35–44. https://doi.org/10.1007/s11240-013-0337-8
Razdan MK (2003) Introduction to plant tissue culture. Science Publishers, Inc., Plymouth
Sall J, Stephens ML, Lehman A, Loring S (2017) JMP start statistics: a guide to statistics and data analysis using JMP. SAS Institute, Cary
Scagliusi SM (2014) Establishing isolated microspore culture to produce doubled haploid plants in Brazilian wheat (Triticum aestivum L.). Aust J Crop Sci 8:887–894
Seifert GJ, Roberts K (2007) The biology of arabinogalactan proteins. Annu Rev Plant Biol 58:137–161. https://doi.org/10.1146/annurev.arplant.58.032806.103801
Shumilina D, Kornyukhin D, Domblides E, Soldatenko A, Artemyeva A (2020) Effects of genotype and culture conditions on microspore embryogenesis and plant regeneration in Brassica Rapa Ssp. Rapa L Plants 278:1–12. https://doi.org/10.3390/plants9020278
Simonović AD, Filipović BK, Trifunović MM, Malkov SN, Milinković VP, Jevremović SB, Subotić AR (2015) Plant regeneration in leaf culture of Centaurium erythraea Rafn. Part 2: the role of arabinogalactan proteins. Plant Cell Tiss Org Cult 121:721–739. https://doi.org/10.1007/s11240-015-0741-3
Sriskandarajah S, Sameri M, Lerceteau-Köhler E, Westerbergh A (2015) Increased recovery of green doubled haploid plants from barley anther culture. Crop Sci 55:2806–2812. https://doi.org/10.2135/cropsci2015.04.0245
Supena EDJ, Suharsono S, Jacobsen E, Custers JBM (2006) The successful development of a shed-microspore culture protocol for doubled haploid production in Indonesian hot peppers (Capsicum annuum L.). Plant Cell Rep 25:1–10. https://doi.org/10.1007/s00299-005-0028-y
Swanson EB (1990) Microspore culture in Brassica. In: Pollard JW, Walker JM (eds) Methods in molecular biology, Plant Cell and Tissue Culture. Humana Press, New Jersey, pp 159–170
Tadesse W, Tawkaz S, Inagaki MN, Picard E, Baum M (2013) Methods and applications of doubled haploid technology in wheat breeding, vol 36. ICARDA, Aleppo
Tran F, Penniket C, Patel RV, Provart NJ, Laroche A, Rowland O, Robert LS (2013) Developmental transcriptional profiling reveals key insights into Triticeae reproductive development. Plant J 74:971–988. https://doi.org/10.1111/tpj.12206
Whittle CA, Malik MR, Li R, Krochko JE (2010) Comparative transcript analyses of the ovule, microspore, and mature pollen in Brassica napus. Plant Mol Biol 72:279–299. https://doi.org/10.1007/s11103-009-9567-x
Yu M, Zhao J (2012) The cytological changes of tobacco zygote and proembryo cells induced by beta-glucosyl Yariv reagent suggest the involvement of arabinogalactan proteins in cell division and cell plate formation. BMC Plant Biol 126:1–16. https://doi.org/10.1186/1471-2229-12-126
Zhong J, Ren Y, Yu M, Ma T, Zhang X, Zhao J (2011) Roles of arabinogalactan proteins in cotyledon formation and cell wall deposition during embryo development of Arabidopsis. Protoplasma 248:551–563. https://doi.org/10.1007/s00709-010-0204-y
Funding
The authors gratefully acknowledge the Research Deputy of Tarbiat Modares University, Tehran, for financial support of this research project.
Author information
Authors and Affiliations
Contributions
AM conceived, designed, and managed the research. AP performed the experiments. AP and HD analyzed the data. SRM participated in the design of the study, AP wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Additional information
Editor: Baochun Li
Supplementary Information
ESM 1
(DOCX 983 kb)
Rights and permissions
About this article
Cite this article
Pourmohammad, A., Moieni, A., Dehghani, H. et al. Field-grown donor plants and arabinogalactan proteins improve microspore embryogenesis in sweet pepper (Capsicum annuum L.). In Vitro Cell.Dev.Biol.-Plant 57, 510–518 (2021). https://doi.org/10.1007/s11627-020-10152-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-020-10152-2