Abstract
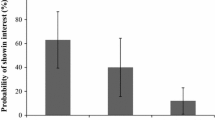
Insect-plant interactions involve physiological adaptations by insects to secondary metabolic compounds synthetized by host plants, which are considered essential for the determination of resources partitioning of these insects. Data on such phenomena are important to understand evolutionary and ecological processes. However, climatic factors also seem to play a key role in affecting these patterns. The present study aimed to investigate the influence of seasonal variation on patterns of host plant use (Passifloraceae) by Heliconiini butterflies (Nymphalidae: Heliconiinae) at a Neotropical site in Southeastern Brazil. A total of 12 species of Heliconiini were reported, with nine of them being resident and using five species of Passiflora (Passifloraceae) as larval host plants. Three host plant species accounted for 97% of the total use, and the use varied along the seasons highlighting the plasticity boundaries in Heliconiini and possible limiting factors.



Similar content being viewed by others
References
Abarca M (2019) Herbivore seasonality responds to conflicting cues: untangling the effects of host, temperature, and photoperiod. PLoS One 14(9):e0222227. https://doi.org/10.1371/journal.pone.0222227
Alexander AJ (1961) A study of the biology and behavior of the caterpillars, pupae and emerging butterflies of the Heliconiinae in Trinidad, West Indies. Part I. Some aspects of larval behavior. Zool 46(1):1–24
Beebe W, Crane J, Fleming H (1960) A comparison of eggs, larvae and pupae in fourteen species of heliconiine butterflies from Trinidad. W.I. Zool 45:111–154
Benson WW (1978) Resource partitioning in passion vine butterflies. Evol 32:493–518
Benson WW, Brown KS, Gilbert LE (1975) Coevolution of plant and herbivores: passion flower butterflies. Evol 29(4):659–680. https://doi.org/10.2307/2407076
Boggs CL, Watt WB, Ehrlich PR (2003) Butterflies: ecology and evolution taking flight. The University of Chicago Press, Chicago, p 756
Brower AVZ (1997) The evolution of ecologically important characters in Heliconius butterflies (Lepidoptera: Nymphalidae): a cladistic review. Zool J Linnean Soc 119(4):457–472. https://doi.org/10.1111/j.1096-3642.1997.tb00143.x
Brown KS (1981) The biology of Heliconius and related genera. Annu Rev Entomol 26:427–457. https://doi.org/10.1146/annurev.en.26.010181.002235
Brown KS, Benson WW (1974) Adaptive polymorphism associated with multiple Müllerian mimicry in Heliconius numata (Lepidoptera: Nymphalidae). Biotropica 6(4):205–228. https://doi.org/10.2307/2989666
Caldas A (2014) Species traits of relevance for climate vulnerability and the prediction of phenological responses to climate change. Journal of the Lepidopterists’. Soc 68(3):197–202. https://doi.org/10.18473/lepi.v68i3.a7
Darragh K, Vanjari S, Mann F, Gonzalez-Rojas MF, Morrison CR, Salazar C, Pardo-Diaz C, Merrill RM, McMillan WO, Schulz S, Jiggins CD (2017) Male sex pheromone components in Heliconius butterflies released by the androconia affect female choice. PeerJ 5:e3953. https://doi.org/10.7717/peerj.3953
De Nardin J, Buffon V, Revers LF, Araújo AM (2018) Association between molecular markers and behavioral phenotypes in the immatures of a butterfly. Genet Mol Biol 41:243–252. https://doi.org/10.1590/1678-4685-gmb-2017-0073
Dell’Erba R, Kaminski LA, Moreira GRP (2005) O estágio do ovo dos Heliconiini (Lepidoptera, Nymphalidae) do Rio Grande do Sul, Brasil. Iheringia Série Zool 95(1):29–46. https://doi.org/10.1590/S0073-47212005000100006
Dennis RLH, Shreeve TG, Arnold HR, Roy DB (2005) Does diet breadth control herbivorous insect distribution size? Life history and resource outlets for specialist butterflies. J Insect Conserv 9:187–200. https://doi.org/10.1007/s10841-005-5660-x
Dyer LA, Singer MS, Lill JT, Stireman JO, Gentry GL, Marquis RJ, Ricklefs RE, Greeney HF, Wagner DL, Morais HC, Diniz IR, Kursar TA, Coley PD (2007) Host specificity of Lepidoptera in tropical and temperate forests. Nature 448:696–700. https://doi.org/10.1038/nature05884
Ehrlich PR, Gilbert LE (1973) Population structure and dynamics of the tropical butterfly Heliconius ethilla. Biotropica 5(2):69–82. https://doi.org/10.2307/2989656
Ehrlich PR, Raven PH (1964) Butterflies and plants: a study in coevolution. Evol 18(4):586–608. https://doi.org/10.1111/j.1558-5646.1964.tb01674.x
Feeny P (1970) Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecol 51:565–581
Francini RB, Duarte M, Mielke OHH, Caldas A, Freitas AVL (2011) Butterflies (Lepidoptera, Papilionoidea and Hesperioidea) of the “Baixada Santista” region, coastal São Paulo, southeastern Brazil. Revista Brasileira de Entomologia 55(1):55–68. https://doi.org/10.1590/S0085-56262011000100010
Gilbert LE (1975) Ecological consequences of a coevolved mutualism between butterflies and plants. In: Gilbert LE, Raven PH (eds) Coevolution of animals and plants. University of Texas Press, Austin, pp 210–240
Gilbert LE (1991) Biodiversity of a Central American Heliconius community: pattern, process, and problems. In: Price PW, Lewinsohn TM, Fernandes GW, Benson WW (eds) Plant-animal interactions: evolutionary ecology in tropical and temperate regions. John Wiley and sons, New York, pp 403–427
Gilbert LE, Smiley JT (1978) Determinants of local diversity in phytophagous insects: host specialists in tropical environments. In: Mound LA, Waloff N (eds) Diversity of insect faunas: Symposia of the Royal Entomological Society of London number 9, pp 89–104
Grimaldi D, Engel MS (2005) Evolution of the insects. Cambridge University Press, New York, NY, p 772
Gotelli NJ, Ellison AM (2013) A primer of ecological statistics, Second edn. Sinauer Associates, Massachusetts USA, p 615
Heliconius Genome Consortium (2012) Butterfly genome reveals promiscuous exchange of mimicry adaptations among species. Nature 487(7405):94–98. https://doi.org/10.1038/nature11041
Hijmans RJ, Guarino L, Mathur P (2012) DIVA-GIS 7.5 Software. http://www.diva-gis.org/ Accessed Mai 2012
Jiggins CD (2008) Ecological speciation in mimetic butterflies. Bioscience 58(6):541–548. https://doi.org/10.1641/b580610
Jiggins CD (2017) The ecology and evolution of Heliconius butterflies. Oxford University Press, New York, USA, p 321
Jorge LR, Cordeiro-Estrela P, Klaczko LB, Moreira GRP, Freitas AVL (2011) Host-plant dependent wing phenotypic variation in the neotropical butterfly Heliconius erato. Biol J Linn Soc 102(4):765–774. https://doi.org/10.1111/j.1095-8312.2010.01610.x
Joron M, Jiggins CD, Papanicolaou A, McMillan WO (2006) Heliconius wing patterns: an evo-devo model for understanding phenotypic diversity. Heredity 97:157–167. https://doi.org/10.1038/sj.hdy.6800873
Kaminski LA, Freitas AVL, Oliveira PS (2010) Interaction between mutualisms: ant-tended butterflies exploit enemy-free space provided by ant-treehopper associations. Am Nat 176(3):322–334. https://doi.org/10.1086/655427
Krebs CJ (1999) Ecological Methodology, 2nd edition Addison Wesley Longman, Menlo Park, CA 620p
Kristensen NP, Scoble MJ, Karsholt O (2007) Lepidoptera phylogeny and systematics: the state of inventorying moth and butterfly diversity. Zootaxa 1668(1):699–747. 10.11646/zootaxa.1668.1.30
Kronforst MR, Papa R (2015) The functional basis of wing patterning in Heliconius butterflies: the molecules behind mimicry. Genetics 200(1):1–19. https://doi.org/10.1534/genetics.114.172387
Lancaster LT (2020) Host use diversification during range shifts shapes global variation in Lepidopteran dietary breadth. Nature Ecol and Evol 4:963–969. https://doi.org/10.1038/s41559-020-1199-1
McMillan WO, Monteiro A, Kapan DD (2002) Development and evolution on the wing. Trends Ecol Evol 17(3):125–133. https://doi.org/10.1016/S0169-5347(01)02427-2
Menna-Barreto Y, Araújo AM (1985) Evidence for host plant preferences in Heliconius erato phyllis from southern Brazil (Nymphalidae). Journal of Research on the Lepidoptera 24:41–46
Montejo-Kovacevich G, Martin SH, Meier JI, Bacquet CN, Monllor M, Jiggins CD, Nadeau NJ (2020) Microclimate buffering and thermal tolerance across elevations in a tropical butterfly. J Exp Biol 223:1–19 jeb220426. https://doi.org/10.1242/jeb.220426
Müller J, Stadler J, Jarzabek-Müller A, Hacker H, ter Braak C, Brandl R (2011) The predictability of phytophagous insect communities: Host specialists as habitat specialists. PLoS One 6(10):e25986. https://doi.org/10.1371/journal.pone.0025986
Navarro-Cano JA, Karlsson B, Posledovich D, Toftegaard T, Wiklund C, Ehrlén J, Gotthard K (2015) Climate change, phenology, and butterfly host plant utilization. AMBIO 44(suppl. 1):S78–S88
Nylin S, Nygren GH, Windig JJ, Janz N, Bergström A (2005) Genetics of host-plant preference in the comma butterfly Polygonia c-album (Nymphalidae), and evolutionary implications. Biol J Linn Soc 84(4):755–765. https://doi.org/10.1111/j.1095-8312.2004.00433.x
Périco E (1995) Interação entre quatro espécies de Heliconiini (Lepidoptera: Nymphalidae) e suas plantas hospedeiras (Passifloraceae) em uma floresta secundária do Rio Grande do Sul, Brasil. Biociências 3(2):3–18
Périco E, Araújo AM (1991) Suitability of host plants (Passifloraceae) and their acceptableness by Heliconius erato and Dryas iulia (Lepidoptera: Nymphalidae). Evol Biol 5:59–74
Ramos RR, Freitas AVL (1999) Population biology and wing color variation in Heliconius erato phyllis (Nymphalidae). Journal of the Lepidopterists’ Society 53(1):11–21
Ramos RR, Rodrigues D, Freitas AVL (2012) Oviposition preference and larval performance in a Heliconius erato phyllis (Lepidoptera: Nymphalidae) population from southeastern Brazil: is there a positive relationship? J Nat Hist 46(11-12):669–681. https://doi.org/10.1080/00222933.2011.651633
Rodrigues D, Moreira GRP (1999) Feeding preference of Heliconius erato (Lep.: Nymphalidae) in relation to leaf age and consequences for larval performance. Journal of the Lepidopterists’ Society 53(3):108–113
Rodrigues D, Moreira GRP (2004) Seasonal variation in larval host plants and consequences for Heliconius erato (Lepidoptera: Nymphalidae) adult body size. Austral Ecol 29(4):437–445. https://doi.org/10.1111/j.1442-9993.2004.01381.x
Rosser N, Phillimore AB, Huertas B, Willmott KR, Mallet J (2012) Testing historical explanations for gradients in species richness in heliconiine butterflies of tropical America. Biol J Linn Soc 105:479–497. https://doi.org/10.1111/j.1095-8312.2011.01814.x
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect–plant biology, 2nd edn. United States by Oxford University Press, Oxford, p 421
Silva AK, Gonçalves GL, Moreira GRP (2014) Larval feeding choices in heliconians: induced preferences are not constrained by performance and host plant phylogeny. Anim Behav 89:155–162. https://doi.org/10.1016/j.anbehav.2013.12.027
Singer MC (1986) The definition and measurement of oviposition preference in plant-feeding insects. In: Miller JR, Miller TA (eds) Insect-plant interactions Springer Series in Experimental Entomology. Springer, New York, NY, pp 65–94
Speight MR, Hunter MD, Watt AD (2008) Ecology of insects: concepts and applications. UK, Oxford, p 640
Spencer KC (1988) Chemical mediation of coevolution. Academic Press, Inc., Harcourt Brace Jovanovich Publishers, San Diego, California, p 609
Turlure C, Schtickzelle N, Dubois Q, Baguette M, Roger LHD, Van Dyck H (2019) Suitability and transferability of the resource-based habitat concept: a test with an assemblage of butterflies. Front Ecol Evol 7:127. https://doi.org/10.3389/fevo.2019.00127
Turner JRG (1981) Adaptation and evolution in Heliconius: a defense of neo Darwinism. Annu Rev Ecol Syst 12:99–121. https://doi.org/10.1146/annurev.es.12.110181.000531
Ururahy JC, Collares JER, Santos MM, Barreto RAA (1987) Vegetação. In: Projeto RADAMBRASIL. Vol. 32. fls. sf 23-24 (Rio de Janeiro/Vitória). Edit. IBGE/Ministério das Minas e Energia, Brasília, DF
Uyi OO, Zachariades C, Heshula LU, Hill MP (2018) Developmental and reproductive performance of a specialist herbivore depend on seasonality of, and light conditions experienced by, the host plant. PLoS One 13(1):e0190700. https://doi.org/10.1371/journal.pone.0190700
VanKuren NW, Massardo D, Nallu S, Kronforst MR (2019) Butterfly mimicry polymorphisms highlight phylogenetic limits of gene reuse in the evolution of diverse adaptations. Mol Biol Evol 36(12):2842–2853. https://doi.org/10.1093/molbev/msz194
Acknowledgements
To Keith S. Brown Jr. for valuable suggestions during all phases of the present study. To Neil Rosser, who revised and much improved the final version. To two anonymous referees that gave valuable suggestions for improving the manuscript. To Paulo Eduardo Costa for allowing the field work at the Parque Ecológico do Voturuá.
Funding
Financial support was provided by the CNPq (process 302585/2011-7 and 421248/2017-3), and by FAPESP (grant 2012/50260-6). This publication is part of the RedeLep ‘Rede Nacional de Pesquisa e Conservação de Lepidópteros’ SISBIOTA Brasil/CNPq (563332/2010-7), and of the BIOTA-FAPESP Program (grants 2011/50225-3 and 2013/50297-0). RRR thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) - Finance Code 001. The present study is registered at the SISGEN: A2641E3.
Author information
Authors and Affiliations
Contributions
RRR and AVLF conceived the ideas; RRR performed most of the field work, with contributions of AVLF and RBF. RRR and RBF analyzed the data. RRR and AVLF led the writing, with contributions of all authors. All authors read and approved the final manuscript.
Corresponding authors
Additional information
Edited by Diego F Segura
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ramos, R.R., Francini, R.B., Habib, M.ED.M. et al. Seasonal Patterns of Host Plant Use in an Assemblage of Heliconiini Butterflies (Lepidoptera: Nymphalidae) in a Neotropical forest. Neotrop Entomol 50, 358–365 (2021). https://doi.org/10.1007/s13744-021-00855-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-021-00855-5