Abstract
Purpose
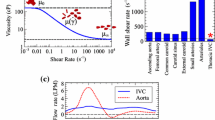
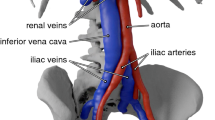
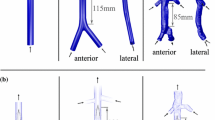
Robust experimental data for performing validation of fluid–structure interaction (FSI) simulations of the transport of deformable solid bodies in internal flow are currently lacking. This in vitro experimental study characterizes the clot trapping efficiency of a new generic conical-type inferior vena cava (IVC) filter in a rigid anatomical model of the IVC with carefully characterized test conditions, fluid rheological properties, and clot mechanical properties.
Methods
Various sizes of spherical and cylindrical clots made of synthetic materials (nylon and polyacrylamide gel) and bovine blood are serially injected into the anatomical IVC model under worst-case exercise flow conditions. Clot trapping efficiencies and their uncertainties are then quantified for each combination of clot shape, size, and material.
Results
Experiments reveal the clot trapping efficiency increases with increasing clot diameter and length, with trapping efficiencies ranging from as low as approximately 42% for small 3.2 mm diameter spherical clots up to 100% for larger clot sizes. Because of the asymmetry of the anatomical IVC model, the data also reveal the iliac vein of clot origin influences the clot trapping efficiency, with the trapping efficiency for clots injected into the left iliac vein up to a factor of 7.5 times greater than that for clots injected into the right iliac (trapping efficiencies of approximately 10% versus 75%, respectively).
Conclusion
Overall, this data set provides a benchmark for validating simulations predicting IVC filter clot trapping efficiency and, more generally, low-Reynolds number FSI modeling.








Similar content being viewed by others
References
ASME V&V 40-2018. Assessing Credibility of Computational Modeling Through Verification and Validation: Application to Medical Devices. New York: American Society of Mechanical Engineers, 2018.
Aycock, K. I., R. L. Campbell, F. C. Lynch, K. B. Manning, and B. A. Craven. The importance of hemorheology and patient anatomy on the hemodynamics in the inferior vena cava. Ann. Biomed. Eng. 44(12):3568–3582, 2016.
Aycock, K. I., R. L. Campbell, F. C. Lynch, K. B. Manning, and B. A. Craven. Computational predictions of the embolus-trapping performance of an IVC filter in patient-specific and idealized IVC geometries. Biomech. Model. Mechanobiol. 16:1957–1969, 2017.
Aycock, K. I., R. L. Campbell, K. B. Manning, and B. A. Craven. A resolved two-way coupled CFD/6-DOF approach for predicting embolus transport and the embolus-trapping efficiency of IVC filters. Biomech. Model. Mechanobiol. 16:851–869, 2017.
Aycock, K. I., R. L. Campbell, K. B. Manning, S. P. Sastry, S. M. Shontz, F. C. Lynch, and B. A. Craven. A computational method for predicting inferior vena cava filter performance on a patient-specific basis. J. Biomech. Eng. 136(8):081003, 2014.
Aycock, K. I., P. Hariharan, and B. A. Craven. Particle image velocimetry measurements in an anatomical vascular model fabricated using inkjet 3D printing. Exp. Fluids 58(11):154, 2017.
Brown, L. D., T. T. Cat, and A. DasGupta. Interval estimation for a proportion. Stat. Sci. 16:101–133, 2001.
Cheng, C. P., R. J. Herfkens, and C. A. Taylor. Inferior vena caval hemodynamics quantified in vivo at rest and during cycling exercise using magnetic resonance imaging. Am. J. Physol. Heart Circ. 284:1161–1167, 2003.
Craven, B. A., K. I. Aycock, and K. B. Manning. Steady flow in a patient-averaged inferior vena cava: Part II—computational fluid dynamics verification and validation. Cardiovasc. Eng. Techn. 9(4):654–673, 2018.
De Nayer, G., A. Kalmbach, M. Breuer, S. Sicklinger, and R. Wüchner. Flow past a cylinder with a flexible splitter plate: A complementary experimental–numerical investigation and a new FSI test case (FSI-PfS-1a). Comput. Fluids. 99:18–43, 2014.
Dria, S. J., and M. D. Eggers. In vitro evaluation of clot capture efficiency of an absorbable vena cava filter. J. Vasc. Surg-Venous L. 4(4):472–478, 2016.
Elbing, B. R., S. D. Young, M. L. Jonson, R. L. Campbell, B. A. Craven, R. F. Kunz, and K. L. Koudela. Experimental characterization of high-amplitude fluid–structure interaction of a flexible hydrofoil at high Reynolds number. J. Vib. Acoust 142(4):041014, 2020.
FDA. (1999) Guidance for industry and FDA staff: Guidance for cardiovascular intravascular filter 510(k) submissions. https://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/ucm073777.pdf.
Fung, Y. C., and S. C. Cowin. Biomechanics: Mechanical Properties of Living Tissues (2nd ed.). New York: Springer, 1993.
Gallagher, M. B., K. I. Aycock, B. A. Craven, and K. B. Manning. Steady flow in a patient-averaged inferior vena cava—Part I: Particle image velocimetry measurements at rest and exercise conditions. Cardiovasc. Eng. Techn. 9:641–653, 2018.
Gao, X., J. Zhang, B. Chen, H. Yu, J. Li, S. Zhang, Z. Feng, L. Ye, and J. Han. A new self-convertible inferior vena cava filter: Experimental in-vitro and in-vivo evaluation. J. Vasc. Interv. Radiol. 22(6):829–834, 2010.
Gomes, J. P., S. Yigit, H. Lienhart, and M. Schäfer. Experimental and numerical study on a laminar fluid–structure interaction reference test case. J. Fluid. Struct. 27(1):43–61, 2011.
Günther, R. W., J. Neuerburg, A. Mossdorf, J. Pfeffer, R. Høj, A. Mølgaard-Nielsen, A. Bücker, and T. Schmitz-Rode. New optional IVC filter for percutaneous retrieval - In vitro evaluation of embolus capturing efficiency. Rofo. 177(5):632–636, 2005.
Hammer, F. D., H. P. Rousseau, F. G. Joffre, B. P. Sentenac, T. Tran-Van, and R. P. Barthelemy. In vitro evaluation of vena cava filters. J. Vasc. Interv. Radiol. 5(6):869–876, 1994.
Hessenthaler, A., N. R. Gaddum, O. Holub, R. Sinkus, O. Röhrle, and D. Nordsletten. Experiment for validation of fluid–structure interaction models and algorithms. Int. J. Numer. Method. Biomed. Eng. 33(9):e2848, 2017.
Holzapfel, G. A., T. C. Gasser, and M. Stadler. A structural model for the viscoelastic behavior of arterial walls: Continuum formulation and finite element analysis. Eur. J. Mech. A 21(3):441–463, 2002.
Hosaka, J., S. Roy, K. Kvernebo, I. Enge, and F. Laerum. In vitro function of an adjustable temporary venous spring filter: Comparison with the temporary RF02 filter and the permanent Greenfield vena cava filter. Acad. Radiol. 5(9):620–625, 1998.
Humphrey, J. D. Continuum biomechanics of soft biological tissues. Proc. R. Soc. Lond. Series A 459(2029):3–46, 2003.
ISO 25539-3:2011ISO 25539-3:2011. Cardiovascular implants — endovascular devices — part 3: Vena cava filters. Geneva: International Organization for Standardization, 2011.
Jaeger, H. J., S. Kolb, T. Mair, M. Geller, A. Christmann, R. K. Kinne, and K. D. Mathias. In vitro model for the evaluation of inferior vena cava filters: Effect of experimental parameters on thrombus-capturing efficacy of the Vena Tech-LGM filter. J. Vasc. Interv. Radiol. 9(2):295–304, 1998.
Jaeger, H. J., T. Mair, M. Geller, R. K. Kinne, A. Christmann, and K. D. Mathias. A physiologic in vitro model of the inferior vena cava with a computer-controlled flow system for testing of inferior vena cava filters. Invest. Radiol. 32(9):511–522, 1997.
Joseph, A. A., D. Voit, and J. Frahm. Inferior vena cava revisited – Real time flow MRI of respiratory maneuvers. NMR Biomed. 33(4):e4232, 2020.
Katsamouris, A. A., A. C. Waltman, M. A. Delichatsios, and C. A. Athanasoulis. Inferior vena cava filters: In vitro comparison of clot trapping and flow dynamics. Radiology 166(2):351–366, 1988.
Kaufman, J. A., A. C. Waltman, S. M. Rivitz, and S. C. Geller. Anatomical observations on the renal veins and inferior vena cava at magnetic resonance angiography. CardioVasc. Interv. Radiol. 18(3):153–157, 1995.
Lopez, J. M., G. Fortuny, D. Puigjaner, J. Herrero, and F. Marimon. A comparative CFD study of four inferior vena cava filters. Int. J. Numer. Method. Biomed. Eng. 34(7):e2990, 2018.
Lorch, H., A. Dallmann, M. Zwaan, and H. Weiss. Efficacy of permanent and retrievable vena cava filters: experimental studies and evaluation of a new device. Cardiovasc. Intervent. Radiol. 25(3):193–199, 2002.
Lorch, H., M. Zwaan, C. Kulke, and H. D. Weiss. In vitro studies of temporary vena cava filters. Cardiovasc. Intervent Radiol. 21(2):146–150, 1998.
Mahnken, A. H., J. Pfeffer, S. Stanzel, A. Mossdorf, R. W. Günther, and T. Schmitz-Rode. In vitro evaluation of optionally retrievable and permanent IVC filters. Invest Radiol. 42(7):529–535, 2007.
Montgomery, J. P., and J. A. Kaufman. A critical review of available retrievable inferior vena cava filters and future directions. Semin. Intervent. Rad. 33(2):79–87, 2016.
Nahirnyak, V. M., S. W. Yoon, and C. K. Holland. Acousto-mechanical and thermal properties of clotted blood. J. Acoust. Soc. Am. 119(6):3766–3772, 2006.
Neuerburg, J., A. Haupt-Pichler, F. J. Katterbach, R. Eilers, T. Siess, K. Buro, C. von Pichler, K. Mottaghy, and R. W. Günther. Determining the effectiveness of percutaneous cava filters: Experimental studies. Rofo. 164(4):331–337, 1996.
Nicolas, M., M. Malve, E. Peña, M. A. Martinez, and R. Leask. In vitro comparison of Günther Tulip and Celect filters. Testing filtering efficiency and pressure drop. J. Biomech. 48(3):504–511, 2015.
Nicolas, M., V. Palero, E. Pena, M. Arroyo, M. Martinez, and M. Malve. Numerical and experimental study of the fluid flow through a medical device. Int. Commun. Heat Mass 61(2):170–178, 2015.
Rahbar, E., D. Mori, and J. Moore, Jr. Three-dimensional analysis of flow disturbances caused by clots in inferior vena cava filters. J. Vasc. Interv. Radiol. 22(6):835–842, 2011.
Ren, Z., S. L. Wang, and M. A. Singer. Modeling hemodynamics in an unoccluded and partially occluded inferior vena cava under rest and exercise conditions. Med. Biol. Eng. Comput. 50:277–287, 2012.
Robinson, R. A., L. H. Herbertson, S. S. Das, R. A. Malinauskas, W. F. Pritchard, and L. W. Grossman. Limitations of using synthetic blood clots for measuring in vitro clot capture efficiency of inferior vena cava filters. Med. Devices 10(6):49–57, 2013.
Rogers, F. B., G. Strindberg, S. R. Shackford, T. M. Osler, C. S. Morris, M. A. Ricci, K. E. Najarian, R. D’Agostino, and D. B. Pilcher. Five-year follow-up of prophylactic vena cava filters in high-risk trauma patients. Arch. Surg. 133(4):406–412, 1998.
Simon, M., D. J. Rabkin, S. Kleshinski, D. Kim, and B. J. Ransil. Comparative evaluation of inferior vena cava filters with an in vitro physiologic simulation of the vena cava. Radiology 189(3):769–774, 1993.
Singer, M. A., W. D. Henshaw, and S. L. Wang. Computational modeling of blood flow in the TrapEase inferior vena cava filter. J. Vasc. Interv. Radiol. 20(6):799–805, 2009.
Singer, M. A., and S. L. Wang. Modeling blood flow in a tilted inferior vena cava filter: Does tilt adversely affect hemodynamics? J. Vasc. Interv. Radiol. 22(2):229–235, 2011.
Singer, M. A., S. L. Wang, and D. P. Diachin. Design optimization of vena cava filters: An application to dual filtration devices. J. Biomech. Eng. 132(10):101006, 2010.
Stewart, S. F. C., R. A. Robinson, R. A. Nelson, and R. A. Malinauskas. Effects of thrombosed vena cava filters on blood flow: Flow visualization and numerical modeling. Ann. Biomed. Eng. 36(11):1764–1781, 2008.
Stoneham, G. W., B. E. Burbridge, and S. F. Millward. Temporary inferior vena cava filters: In vitro comparison with permanent IVC filters. J. Vasc. Interv. Radiol. 6(5):731–736, 1995.
Swaminathan, T. N., H. H. Hu, and A. A. Patel. Numerical analysis of the hemodynamics and embolus capture of a Greenfield vena cava filter. J. Biomech. Eng. 128(3):360–370, 2006.
Tedaldi, E., C. Montanari, K. I. Aycock, F. Sturla, A. Redaelli, and K. B. Manning. An experimental and computational study of the inferior vena cava hemodynamics under respiratory-induced collapse of the infrarenal IVC. Med. Eng. Phys. 54(4):44–55, 2018.
Tse, J. R., and A. J. Engler. Preparation of hydrogel substrates with tunable mechanical properties. Curr. Protoc. Cell Biol. 47(1):1–16, 2010.
Turek, S., and J. Hron. Proposal for numerical benchmarking of fluid–structure interaction between an elastic object and laminar incompressible flow. In: Fluid–Structure Interaction, edited by H. J. Bungartz, and M. Schäfer. Berlin: Springer, 2006, pp. 371–385.
Tutwiler, V., A. D. Peshkova, I. A. Adrianova, D. R. Khasanova, J. W. Weisel, and R. I. Litvinov. Contraction of blood clots is impaired in acute ischemic stroke. Arterioscl. Thromb. Vasc. Biol. 37(2):271–279, 2017.
Wang, S. L., and M. A. Singer. Toward an optimal position for inferior vena cava filters: Computational modeling of the impact of renal vein inflow with Celect and TrapEase filters. J. Vasc. Interv. Radiol. 21(3):367–374, 2010.
Yang, C., F. Ma, C. Gao, Y. Kang, G. Zhang, P. Liu, H. Jiang, and Z. Chang. Design and evaluation of a novel biodegradable inferior vena cava filter. J. Biomater. Appl. 33(8):1060–1069, 2019.
Acknowledgments
The authors thank Dr. Luke Herbertson for providing the protocol to create the polyacrylamide clots, Sailahari Ponnaluri, who helped with the creation of the polyacrylamide clots, and Dr. Frank Lynch for technical consultation on best practices for IVC filter deployment. The authors also thank Confluent Medical Technologies for manufacturing the generic nitinol IVC filter. This study was funded by the U.S. FDA Center for Devices and Radiological Health (CDRH) Critical Path program. The research was supported in part by an appointment to the Research Participation Program at the U.S. FDA administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and FDA. The findings and conclusions in this article have not been formally disseminated by the FDA and should not be construed to represent any agency determination or policy. The mention of commercial products, their sources, or their use in connection with material reported herein is not to be construed as either an actual or implied endorsement of such products by the Department of Health and Human Services.
Author Contributions
JMR, NSP, HMS, and BCG performed the experimental work with KIA, BAC, and KBM providing input on experimental design. JMR, BCG, KIA, BAC, and KBM performed data analysis and writing of the manuscript, editing, and approving the manuscript submission.
Data Availability
The data that support the findings of this study are available from the corresponding author, KBM, upon reasonable request.
Funding
This study was funded by the U.S. FDA Center for Devices and Radiological Health (CDRH) Critical Path program.
Conflict of interest
J.M. Riley, N.S. Price, H.M. Saaid, B.C. Good, K.I. Aycock, and B.A. Craven declare that they have no conflict of interest. K.B. Manning serves as a consultant for Cordis Inc.
Research Involving Human and Animal Rights
No human studies were carried out by the authors for this article. Approval from the Penn State IACUC was received to conduct the bovine blood studies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Patrick Segers oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary material 3 (MOV 740 kb)
Supplementary material 4 (MOV 610 kb)
Supplementary material 5 (MOV 856 kb)
Supplementary material 6 (MOV 682 kb)
Supplementary material 7 (MOV 1210 kb)
Supplementary material 8 (MOV 896 kb)
Supplementary material 9 (MOV 957 kb)
Supplementary material 10 (MOV 946 kb)
Rights and permissions
About this article
Cite this article
Riley, J.M., Price, N.S., Saaid, H.M. et al. In Vitro Clot Trapping Efficiency of the FDA Generic Inferior Vena Cava Filter in an Anatomical Model: An Experimental Fluid–Structure Interaction Benchmark. Cardiovasc Eng Tech 12, 339–352 (2021). https://doi.org/10.1007/s13239-021-00524-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-021-00524-z