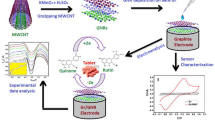
Abstract
Resveratrol has a variety of biological functions, however, a limited number of studies have assessed its interaction with cell surface receptors. In this study, a sandwich-type rat small intestine tissue sensor (RSIT-sensor) was fabricated to detect the response current from receptor stimulation by different resveratrol concentrations via electrochemical workstation. The results showed that with detection limit of 1 × 10–13 mol/L, the maximum rate of change of the response current was found at the concentration of 8.5 × 10–12 mol/L, indicating that the resveratrol-related receptor was saturated. With comparing the response values of prepared biosensor and bare electrode with resveratrol, it can be concluded that the response value of small intestinal cells to resveratrol has obviously been amplified by the intracellular signal transmission system, and its magnification was about 100 times. In the current research, for the first time, kinetics of the interaction between resveratrol and its receptors and the transmission of signals to the body could be quantitatively measured by a biosensor. Our findings may provide new ideas for resveratrol-related receptor analysis, separation and purification, signal transmission, and evaluation of biological function.








Similar content being viewed by others
References
I. Ahmad, M. Hoda, Attenuation of diabetic retinopathy and neuropathy by resveratrol: Review on its molecular mechanisms of action. Life Sci 24515, 117350 (2020)
J.M. Allaire, S.M. Crowley, H.T. Law, S.-Y. Chang, H.-J. Ko, B.A. Vallance, The intestinal epithelium: central coordinator of mucosal immunity. Trends Immunol. 39(9), 677–696 (2018)
M. Athar, J.H. Back, L. Kopelovich, D.R. Bickers, A.L. Kim, Multiple molecular targets of resveratrol: Anti-carcinogenic mechanisms. Arch. Biochem. Biophys. 486(215), 95–102 (2009)
N. Bostanghadiri, A. Pormohammad, A.S. Chirani, R. Pouriran, A. Hashemi, Comprehensive review on the antimicrobial potency of the plant polyphenol resveratrol. Biomed. Pharmacother. 95, 1588–1595 (2017)
K.S. Bhullar, C.C. Udenigwe, Clinical evidence of resveratrol bioactivity in cardiovascular disease. Curr. Opin. Food. Sci. 8, 68–73 (2016)
G.T. Diaz-Gerevini, G. Repossi, A. Dain, M.C. Tarres, M.D.F.A.M.S Undurti Narasimha Das, A.R. Eynard, Beneficial action of resveratrol: How and why? Nutrition 32(2), 174–178 (2016)
M. Elshaer, Y. Chen, X.-J. Wang, X. Tang, Resveratrol: An overview of its anti-cancer mechanisms. Life Sci. 20715, 340–349 (2018)
M. Jang, L. Cai, G.O. Udeani, K.V. Slowing, C.F. Thomas, C.W. Beecher, H.H. Fong, N.R. Farnsworth, A.D. Kinghorn, R.G. Mehta, R.C. Moon, J.M. Pezzuto, Cancer chemopreventive activity of resveratrol natural product derived from grapes. Science 275, 218–200 (1997)
C.-W. Kim, K.-A. Hwang, K.-C. Choi, Anti-metastatic potential of resveratrol and its metabolites by the inhibition of epithelial-mesenchymal transition, migration, and invasion of malignant cancer cells. Phytomedicine 23(14), 1787–1796 (2016)
S.S. Kulkarni, C. Cantó, The molecular targets of resveratrol. BBA-Mol. Basis Dis. 1852(6), 1114–1123 (2015)
H. Li, N. Xia, U. Förstermann, Cardiovascular effects and molecular targets of resveratrol. Nitric Oxide 26(2), 102–110 (2012)
M. Ligt, S. Timmers, P. Schrauwen, Resveratrol and obesity: Can resveratrol relieve metabolic disturbances? BBA-Mol. Basis Dis. 1852(6), 1137–1144 (2015)
S.A.M. Martins, J.R.C. Trabuco, G.A. Monteiro, V. Chu, J.P. Conde, D.M.F. Prazeres, Towards the miniaturization of GPCR-based live-cell screening assays. Trends Biotechnol. 30(11), 566–574 (2012)
R. Méjard, H.J. Griesser, B. Thierry, Optical biosensing for label-free cellular studies. TrAC, Trend Anal. Chem. 53, 178–186 (2014)
M. Ndiaye, C. Philippe, H. Mukhtar, N. Ahmad, The grape antioxidant resveratrol for skin disorders: Promise, prospects, and challenges. Arch. Biochem. Biophys. 508(215), 164–170 (2011)
M.R. Oliveira, S.F. Nabavi, A. Manayi, M. Daglia, Z. Hajheydari, S.M. Nabavi, Resveratrol and the mitochondria: from triggering the intrinsic apoptotic pathway to inducing mitochondrial biogenesis, a mechanistic view. BBA-Gen. Subjects 1860(4), 727–745 (2016)
E. Öztürk, A.K.K. Arslan, M.B. Yerer, A. Bishayee, Resveratrol and diabetes: A critical review of clinical studies. Biomed. Pharmacother. 95, 230–234 (2017)
M.M. Poulsen, K. Fjeldborg, M.J. Ornstrup, T.N. Kjær, M.K. Nøhr, S.B. Pedersen, Resveratrol and inflammation: challenges in translating pre-clinical findings to improved patient outcomes. BBA-Mol. Basis Dis. 1852(6), 1124–1136 (2015)
L. Qiao, L. Jiao, G. Pang, J. Xie, A novel pungency biosensor prepared with fixing taste-bud tissue of rats. Biosens. Bioelectron. 68, 454–461 (2015)
R. Ren, D. Lu, G. Pang, Development of a new and simple method for the detection of histidine-tagged proteins based on thionine-chitosan/gold nanoparticles/horseradish peroxidase. Biomed. Microdevices 22(11), 1–10 (2020)
G. Repossi, U.N. Das, A.R. Eynard, Molecular basis of the beneficial actions of resveratrol. Arch. Med. Res. 51(2), 105–114 (2020)
B.A. Sandoval-Ramírez, Ú. Catalán, A. Pedret, R.M. Valls, M.J. Motilva, L. Rubió, R. Solà, Exploring the effects of phenolic compounds to reduce intestinal damage and improve the intestinal barrier integrity: A systematic review of in vivo animal studies. Clin. Nutr. 2020, In Press
L. Subramanian, S. Youssef, S. Bhattacharya, J. Kenealey, A.S. Polans, P.R. van Ginkel, Resveratrol: challenges in translation to the clinic–a critical discussion. Clin. Cancer Res. 16(24), 5942–5948 (2010)
H.Y. Tsai, C.T. Ho, Y.K. Chen, Biological actions and molecular effects of resveratrol, pterostilbene, and 3’-hydroxypterostilbene. J. Food Drug Anal. 25(1), 134–147 (2017)
S. Ushiama, Y. Ishimaru, M. Narukawa, M. Yoshioka, C. Kozuka, N. Watanabe, M. Tsunoda, N. Osakabe, T. Asakura, H. Masuzaki, K. Abe, Catecholamines facilitate fuel expenditure and protect against obesity via a novel network of the gut-brain axis in transcription factor skn-1-deficient mice. Ebiomedicine 8, 60–71 (2016)
A.J. Venkatakrishnan, X. Deupi, G. Lebon, C.G. Tate, G.F. Schertler, M.M. Babu, Molecular signatures of G-protein-coupled receptors. Nature 493(7436), 185–194 (2013)
A. Vojdani, L.R. Gushgari, E. Vojdani, Interaction between food antigens and the immune system: Association with autoimmune disorders. Autoimmun. Rev. 19(3), 102459 (2020)
L. Wei, L. Qiao, G. Pang, J. Xie, A kinetic study of bitter taste receptor sensing using immobilized porcine taste bud tissues. Biosens. Bioelectron. 92, 74–80 (2017)
S. Xiao, Y. Zhang, P. Song, J. Xie, G. Pang, The investigation of allosteric regulation mechanism of analgesic effect using SD rat taste bud tissue biosensor. Biosens. Bioelectron. 126, 815–823 (2019)
Q. Xu, D. Lu, G. Pang, Study on bombykol receptor self-assembly and universality of G protein cellular signal amplification system. ACS Sensors 4, 257–264 (2019)
Y. Ye, H. Guo, X. Sun, Recent progress on cell-based biosensors for analysis of food safety and quality control. Biosens. Bioelectron. 126, 389–404 (2019)
Acknowledgments
This study was financially supported by the National Natural Science Foundation of China (Grant No. 31901782).
Author information
Authors and Affiliations
Contributions
Ruijuan Ren participated in drafting the manuscript, as well as revision. Tingting Liu contributed to study design and drafting the primary version of the manuscript. The study was conducted under Dingqiang Lu’s supervision. All authors have read and approved the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ren, R., Lu, D. & Liu, T. Development of a sandwich-type rat small intestine tissue sensor for detecting resveratrol and its receptors. Biomed Microdevices 23, 13 (2021). https://doi.org/10.1007/s10544-021-00554-3
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-021-00554-3