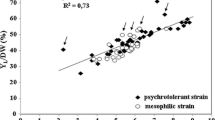
Abstract
Biodiesel is a renewable alternative fuel and glycerol as a main byproduct of the manufacturing process. Lipids could be produced from crude glycerol by using yeasts. The ability of 107 yeast strains to utilize glycerol was screened and 92 of these were selected. 60 strains were determined as a potential for lipid production by Sudan Black B staining. After secondary screening 25 of them showed specific growth rates (OD 600), high biomass production and lipid content. These strains were identified as Pichia cactophila, P. fermentans, P. anomala, Rhodotorula mucilaginosa, R. dairenensis, Clavispora lusitaniae, Saccharomyces cerevisiae, Wickerhamomyces anomalus, Candida glabrata, C. inconspicua, C. albicans, Yarrowia lipolytica with molecular identifications based on ITS and D1/D2 26S rDNA sequences. The results showed that P. cactophila accumulated lipid up to 64.94%, the highest lipid content. C16:0, C18:0, C18:1 and C18:2 essential fatty acids for biodiesel production were detected by GC–MS in the lipids accumulated by all strains. P. cactophila and C. lusitaniae were reported for the first time as lipid-producing yeasts. The results suggest that selected 25 isolates have the ability to grow on crude glycerol and especially P. cactophila produce lipid that has potential use as a feedstock for second generation biodiesel production.
Graphic Abstract



Similar content being viewed by others
References
Areesirisuk, A., Yen, T.B., Chiu, C.H., Liu, C.H., Guo, J.H.: Optimization on yeast lipid production of psuedozyma sp. with response surface methodology for biodiesel manufacturing. J. Adv. Agric. Technol. 2(1), 13–18 (2015). https://doi.org/10.12720/joaat.2.1.13-18
Easterling, E.R., French, W.T., Hemandez, R., Licha, M.: The effect of glycerol as a sole and secondary substrate on the growth and fatty acid composition of Rhodotorula glutinis. Bioresour. Technol. 100(1), 356–361 (2009). https://doi.org/10.1016/j.biortech.2008.05.030
Morita, T., Konishi, M., Fukuoka, T., Imura, T., Kitomato, D.: Microbial conversation of glycerol into glcolipid biosurfactants, mannosylerythritol lipids, by a basidiomycete yeast, Pseudozyma antarctica JCM 10317. Biosci. Bioeng. 104, 78–81 (2007). https://doi.org/10.1263/jbb.104.78
Li, C., Lesnik, K.L., Liu, H.: Microbial conversation of waste glycerol from biodiesel production into value-added products. Energies 6, 4739–4768 (2013). https://doi.org/10.3390/en6094739
Taccari, M., Canonico, L., Comitini, F., Mannazzu, I., Ciani, M.: Screening of yeasts for growth on crude glycerol and optimization of biomass production. Bioresour. Technol. 110, 488–495 (2012). https://doi.org/10.1016/j.biortech.2012.01.109
Sriwongchai, S., Pokethitiyook, P., Kruatrachue, M., Bajwa, K.P., Lee, H.: Screening of selected oleaginous yeasts for lipid production from glycerol and some factors which affect lipid production by Yarrowia lipolytica strains. J. Microbiol. Biotechnol. Food Sci. 2(5), 2344–2348 (2013)
Hwangbo, M., Chu, K.H.: Recent advances in production and extraction of bacterial lipids for biofuel production. Sci. Total Environ. 734, 139420 (2020). https://doi.org/10.1016/j.scitotenv.2020.139420
Thangavelu, K., Sundararaju, P., Srinivasan, N., Muniraj, I., Uthandi, S.: Simultaneous lipid production for biodiesel feedstock and decontamination of sago processing wastewater using Candida tropicalis ASY2. Biotechnol. Biofuels 13(1), 1–14 (2020). https://doi.org/10.1186/s13068-020-01676-1
Li, W., Du, W., Li, Y.H., Liu, D.H., Zhao, Z.B.: Enzymatic transesterification of yeast oil for biodiesel fuel production. Chin. J. Process Eng. 7(4), 137–140 (2007)
Papanikolaou, S., Chatzifragkou, A., Fakas, S., Galiotoupanayotou, M., Komaitis, M., Nicaud, J., Aggelis, G.: Biosynthesis of lipids and organic acids by Yarrowia lipolytica strains cultivated on glucose. Eur. J. Lipid Sci. Technol. 111(12), 1–12 (2009). https://doi.org/10.1002/ejlt.200900055
Ayadi, I., Kamoun, O., Trigui-Lahiani, H., Hdiji, A., Gargouri, A., Belghith, H., Guerfali, M.: Single cell oil production from a newly isolated Candida viswanathii YE4 and agro-industrial by-products valorization. J. Ind. Microbiol. Biotechnol. 43, 901–914 (2016). https://doi.org/10.1007/s10295-016-1772-4
Arous, F., Azabou, S., Triantaphyllidou, I.-E., Aggelis, G., Jaouani, A., Nasri, M., Mechichi, T.: Newly isolated yeasts from Tunisian microhabitats: lipid accumulation and fatty acid composition. Eng. Life Sci. 17, 226–236 (2017). https://doi.org/10.1002/elsc.201500156
Maina, S., Pateraki, C., Kopsahelis, N., Paramithiotis, S., Drosinos, E.H., Papanikolaou, S., Koutinas, A.A.: Microbial oil production from various carbon sources by newly isolated oleaginous yeasts. Eng. Life Sci. 17, 333–344 (2017). https://doi.org/10.1002/elsc.201500153
Filippousi, R., Antoniou, D., Tryfinopoulou, P., Nisiotou, A.A., Nychas, G.J., Koutinas, A.A., Papanikolaou, S.: Isolation, identification and screening of yeasts towards their ability to assimilate biodiesel-derived crude glycerol: microbial production of polyols, endopolysaccharides and lipid. J. Appl. Microbiol. 127(4), 1080–1100 (2019). https://doi.org/10.1111/jam.14373
Uysal, Ü.D., Öncü, E.M., Berikten, D., Yilmaz, N., Tuncel, N.B., Kıvanc, M., Tuncel, M.: Time and temperature dependent microbiological and mycotoxin (ochratoxin-A) levels in boza. Int. J. Food Microbiol. 130(1), 43–48 (2009). https://doi.org/10.1016/j.ijfoodmicro.2008.12.032
Kitcha, S., Cheirsilp, B.: Screening of oleaginous yeasts and optimization for lipid production using crude glycerol as carbon source. Energy Procedia 9, 274–282 (2011). https://doi.org/10.1016/j.egypro.2011.09.029
Pan, L.X., Yang, D.F., Shao, L., Li, W., Chen, G.G., Liang, Z.Q.: Isolation of the oleaginous yeasts from the soil and studies of their lipid producing capacities. Food Technol. Biotechnol. 47(2), 215–220 (2009)
Sluiter, A., Hames, B., Ruiz, R., Scarlata, C., Sluiter, J., Templeton, D.: Determination of sugars, byproducts, and degradation products in liquid fraction process samples.laboratory analytical procedure (LAP), Technical Report NREL/TP-510-42623 (2008)
Bozan, B., Temelli, F.: Supercritical CO2 extraction of flaxseed. J. Am. Oil Chem. Soc. 79(3), 231–235 (2002)
Maoura, N., Mbaiguinam, M., Nguyen, H.V., Gaillardin, C., Pourquie, J.: Identification and typing of the yeast strains isolated from bili bili, a traditional sorghum beer of chad. Afr. J. Biotechnol. 4(7), 646–656 (2005)
White, T., Burns, T., Lee, S., Taylor, J.: Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.K. (eds.) PCR Protocols: A Guide to Methods and Applications, pp. 315–322. Academic Press, London (1990)
Dobrowolski, A., Drzymała, K., Rzechonek, D.A., Mituła, P., Mironczuk, A.M.: Lipid production from waste materials in seawater-based medium by the yeast Yarrowia lipolytica. Front. Microbiol. 10, 547 (2019). https://doi.org/10.3389/fmicb.2019.00547
Rakicka, M., Lazar, Z., Thierry, D., Patrick, F., Jean, M.N.: Lipid production by the oleaginous yeast Yarrowia lipolytica using industrial by-products under different culture conditions. Biotechnol. Biofuels 8, 104 (2015). https://doi.org/10.1186/s13068-015-0286-z
Kosamia, N.M., Samavi, M., Uprety, B.K., Rakshit, S.K.: Valorization of biodiesel byproduct crude glycerol for the production of bioenergy and biochemicals. Catalysts 10(6), 609 (2020). https://doi.org/10.3390/catal10060609
Kumar, L.R., Yellapu, S.K., Tyagi, R.D., Drogui, P.: Microbial lipid and biodiesel production from municipal sludge fortified with crude glycerol medium using pH-based fed-batch strategy. J. Environ. Chem. Eng. (2021). https://doi.org/10.1016/j.jece.2021.105032
Neto, C.D., Sydney, E.B., Candeo, E.S., de Souza, E.B.S., Camargo, D., Sydney, A.C.N., Soccol, C.R.: New method for the extraction of single-cell oils from wet oleaginous microbial biomass: efficiency, oil characterisation and energy assessment. Waste Biomass Valoriz. 11, 3443–3452 (2020). https://doi.org/10.1007/s12649-019-00705-x
Papanikolaou, S., Aggelis, G.: Modelling lipid accumulation and degredation in Yarrowia lipolytica cultivated on industrial fats. Curr. Microbiol. 46, 398–402 (2003). https://doi.org/10.1007/s00284-002-3907-2
Robles-Rodríguez, C.E., Muñoz-Tamayo, R., Bideaux, C., Gorret, N., Guillouet, S.E., Molina-Jouve, C., Roux, G., Aceves-Lara, C.A.: Modeling and optimization of lipid accumulation by Yarrowia lipolytica from glucose under nitrogen depletion conditions. Biotechnol. Bioeng. 115(5), 1137–1151 (2018). https://doi.org/10.1002/bit.26537
Beopoulos, A., Cescut, J., Haddouche, R., Uribelarrea, J.L., Molina-Jouve, C., Nicaud, J.M.: Yarrowia lipolytica as a model for bio-oil production. Prog. Lipid Res. 48, 375–387 (2009). https://doi.org/10.1016/j.plipres.2009.08.005
Sestric, R., Munch, G., Cicek, N., Sparling, R., Levin, D.B.: Growth and neutral lipid synthesis by Yarrowia lipolytica on various carbon substrates under nutrient-sufficient and nutrient-limited conditions. Bioresour. Technol. 164, 41–46 (2014). https://doi.org/10.1016/j.biortech.2014.04.016
Leiva-Candia, D.E., Tsakona, S., Kopsahelis, N., Garcia, I.L., Papanikolaou, S., Dorado, M.P., Koutinas, A.A.: Biorefining of by-product streams from sunflower-based biodiesel production plants for integrated synthesis of microbial oil and value-added co-products. Bioresour. Technol. 190, 57–65 (2015). https://doi.org/10.1016/j.biortech.2015.03.114
Kitcha, S., Cheirsilp, B.: Enhancing lipid production from crude glycerol by newly isolated oleaginous yeasts: strain selection, process optimization, and fed-batch strategy. Bioenergy Res. 6, 300–310 (2013). https://doi.org/10.1007/s12155-012-9257-4
Saenge, C., Cheirsilp, B., Suksaroge, T.T., Bourtoom, T.: Potential use of oleaginous red yeast Rhodotorula glutinis for the bioconversion of crude glycerol from biodiesel plant to lipids and carotenoids. Process Biochem. 46, 210–218 (2011). https://doi.org/10.1016/j.procbio.2010.08.009
Fakas, S., Papanikolaou, S., Batsos, A., Galiotou-Panayotou, M., Mallouchos, A., Aggelis, G.: Evaluating renewable carbon sources as substrates for single cell oil production by Cunninghamella echinulata and Mortierella isabellina. Biomass Bioenergy 33, 573–580 (2009). https://doi.org/10.1016/j.biombioe.2008.09.006
Liu, L., Hu, Y., Lou, W., Li, N., Wu, H., Zong, M.: Use of crude glycerol as sole carbon source for microbial lipid production by oleaginous yeasts. Appl. Biochem. Biotechnol. 182, 495–510 (2017). https://doi.org/10.1007/s12010-016-2340-0
Mast, B., Zöhrens, N., Schmidl, F., Hernandez, R., French, W.T., Merkt, N., Graeff-Hönninger, S.: Lipid production for microbial biodiesel by the oleagenious yeast Rhodotorula glutinis using hydrolysates of wheat straw and miscanthus as carbon sources. Waste Biomass Valoriz. 5(6), 955–962 (2014). https://doi.org/10.1007/s12649-014-9312-9
Knothe, G.: Improving biodiesel fuel properties by modifying fatty ester composition. Energy Environ. Sci. 2(7), 759–766 (2009). https://doi.org/10.1039/B903941D
Bandhu, S., Dasgupta, D., Akhter, J., Kanaujia, P., Suman, S.K., Agrawal, D., Kaul, S., Adhikari, D.K., Ghosh, D.: Statistical design and optimization of single cell oil production from sugarcane bagasse hydrolysate by an oleaginous yeast Rhodotorula sp. IIP-33 using response surface methodology. SpringerPlus 3(1), 691 (2014). https://doi.org/10.1186/2193-1801-3-691
Acknowledgements
This research was financially supported by the Anadolu University Scientific Research Projects Coordination Unit, Turkey (under Grant Number 1410F411).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Berikten, D., Hoşgün, E.Z., Gökdal Otuzbiroğlu, A. et al. Lipid Production from Crude Glycerol by Newly Isolated Oleaginous Yeasts: Strain Selection, Molecular Identification and Fatty Acid Analysis. Waste Biomass Valor 12, 5461–5470 (2021). https://doi.org/10.1007/s12649-021-01405-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-021-01405-1