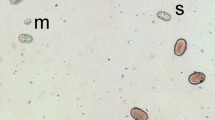
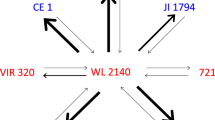
Abstract
Five accessions representing divergent lineages of wild peas (Pisum sativum subsp. elatius) were crossed with each other in both directions and also artificially pollinated with own pollen, to evaluate reproductive barriers inside Pisum sativum which may be important for pea pre-breeding. The outcome of hybrid seeds was evaluated for each combination of crosses. Reciprocal classes of F1 hybrids were compared for pollen and seed fertility and quantitative traits including seed productivity, total biomass etc. Pollination of accession VIR320 with two accessions resulted in hybrids with drastically reduced leaves and pigmentation, resembling known cases of conflict of the nucleus and plastids. Four pairs of reciprocal F1 hybrids showed strong differences in male and female fertility, supposedly a milder manifestation of cytonuclear conflict. Male and female fertility of hybrids correlated with each other, the former appearing more convenient for evaluating genetic disturbance of gametogenesis. Only two hybrid classes showed fully fertile pollen. Some of the accessions studied have been previously reported to differ in a reciprocal translocation and their hybrids were expected to have half-sterile pollen. However accession VIR320 seems to ‘bridge’ both karyological classes showing relatively high pollen fertility in some crosses with representatives of both of them, the reasons for which is discussed. Generally, strong and usually asymmetric cases of incompatibility manifested in the drop of fertility were revealed and their pattern did not correlate with phylogenetic relatedness of the accessions. Hence, wild representatives of P. sativum can hardly be subdivided into natural biological taxa below species rank and their involvement in pea pre-breeding can be complicated by unexpected crossing barriers.




Similar content being viewed by others
Availability of data and materials
The numerical datasets generated and analysed in the course of this study are available from the corresponding author on reasonable request.
References
Abbo S, Zesak I, Schwartz E, Lev-Yadun S, Gopher A (2008) Experimental harvesting of wild peas in Israel: implications for the origins of Near East farming. J Archaeol Sci 35:922–929. https://doi.org/10.1016/j.jas.2007.06.016
Abbo S, Lev-Yadun S, Heun M, Gopher A (2013) On the ‘lost crops’ of the neolithic Near East. J Exp Bot 64:815–822. https://doi.org/10.1093/jxb/ers373
Ben-Ze’ev N, Zohary D (1973) Species relationship in the genus Pisum L. Isr J Bot 22:73–91
Berdnikov VA, Gorel FL, Kosterin OE, Bogdanova VS (2003) Tertiary trisomics in the garden pea as a model of B chromosome evolution in plants. Heredity 91:577–583
Blixt S (1972) Mutation genetics in Pisum. Agri Hortique Genet 30:1–294
Bogdanova VS (2020) Genetic and molecular genetic basis for nuclear-plastid incompatibilities. Plants 9(1):23. https://doi.org/10.3390/plants9010023
Bogdanova VS, Kosterin OE (2006) A case of anomalous chloroplast inheritance in crosses of garden pea involving an accession of wild subspecies. Dokl Biol Sci 406(1–6):44–46
Bogdanova VS, Galieva ER, Kosterin OE (2009) Genetic analysis of nuclear-cytoplasmic incompatibility in pea associated with cytoplasm of an accession of wild subspecies Pisum sativum subsp. elatius (Bieb.) Schmahl. Theor Appl Genet 118(4):801–809
Bogdanova VS, Galieva ER, Yadrikhinskiy AK, Kosterin OE (2012) Inheritance and genetic mapping of two nuclear genes involved in nuclear-cytoplasmic incompatibility in peas (Pisum sativum L.). Theor Appl Genet 124:1503–1512. https://doi.org/10.1007/s00122-012-1804-z
Bogdanova VS, Kosterin OE, Yadrikhinskiy AK (2014) Wild peas vary in their cross-compatibility with cultivated pea (Pisum sativum subsp. sativum L.) depending on alleles of a nuclear-cytoplasmic incompatibility locus. Theor Appl Genet 127:1163–1172. https://doi.org/10.1007/s00122-014-2288-9
Bogdanova VS, Zaytseva OO, Mglinets AV, Shatskaya NV, Kosterin OE, Vasiliev GV (2015) Nuclear-cytoplasmic conflict in pea (Pisum sativum L.) is associated with nuclear and plastidic candidate genes encoding Acetyl-CoA carboxylase subunits. PLoS ONE 10(3):e0119835. https://doi.org/10.1371/journal.pone.0119835
Bogdanova VS, Mglinets AV, Shatskaya NV, Kosterin OE, Solovyev VI, Vasiliev GV (2018) Cryptic divergences in the genus Pisum L. (peas), as revealed by phylogenetic analysis of plastid genomes. Mol Phyl Evol 129:280–290. https://doi.org/10.1016/j.ympev.2018.09.002
Bogdanova VS, Shatskaya NV, Mglinets AV, Kosterin OE, Vasiliev GV (2020) Discordant evolution of organellar genomes in peas (Pisum L.). BioRxiv. https://doi.org/10.1101/2020.05.19.104224
Copenhaver GP, Keith KC, Preuss D (2000) Tetrad analysis in higher plants. A budding technology. Plant Physiol 124:7–16. https://doi.org/10.1104/pp.124.1.7
Coulot P, Rabaute P (2016) Monographie de Leguminosae de France. Tome 4. Tribus des Fabeae, des Cicereae et des Genisteae. Bulletin de la Société Botanique du Centre-Ouest 46:1–902
Coulot P, Rabaute P (2017) Deuxièmes compléments à la Monographie des Leguminosae de France. Le Monde des Plantes 516:11–35
Ellis THN, Poyser SJ, Knox MR, Vershinin AV, Ambrose MJ (1998) Polymorphism of insertion sites of Ty1-copia class retrotransposons and its use for linkage and diversity analysis in pea. Mol Gen Genet 260:9–19. https://doi.org/10.1007/PL00008630
Janco J (1958) Statistické tabulky. Nakladatelství Akademie věd, Praha
Jing R, Johnson R, Seres A, Kiss G, Ambrose MJ, Knox R, Ellis THN, Flawell A (2007) Gene-based sequence diversity analysis of wild pea (Pisum). Genetics 177:2263–2275. https://doi.org/10.1534/genetics.107.081323
Jing R, Vershinin A, Grzebota J, Shaw P, Smýkal P, Marshall D, Ambrose MJ, Ellis THN, Flavell AJ (2010) The genetic diversity and evolution of field pea (Pisum) studied by high throughput retrotransposon based insertion polymorphism (RBIP) marker analysis. BMC Evol Biol 10:44. https://doi.org/10.1186/1471-2148-10-44
Konovalov F, Toshchakova E, Gostimsky S (2005) A CAPS marker set for mapping in linkage group III of pea (Pisum sativum L). Cell Mol Biol Lett 10:163–171
Kosterin OE (2017a) Evolution and genogeography of the wild forms of the pea genus (Pisum L.). Dissertation, Institute of Ctyology & Genetics SB RAS, Novosibirsk (in Russian). https://doi.org/https://doi.org/10.13140/RG.2.2.28701.82404
Kosterin OE (2017b) Abyssinian pea (Lathyrus schaeferi Kosterin nom. nov. pro Pisum abyssinicum A. Br.) is a problematic taxon. Vavilovskii Zhurnal Genetiki i Selektsii Vavilov J Genet Breed 1(2):158–169. https://doi.org/10.18699/VJ17.234
Kosterin OE, Bogdanova VS (2008) Relationship of wild and cultivated forms of Pisum L. as inferred from an analysis of three markers, of the plastid, mitochondrial and nuclear genomes. Genet Resour Crop Evol 55:735–755. https://doi.org/10.1007/s10722-007-9281-y
Kosterin OE, Bogdanova VS (2014) Efficiency of hand pollination in different pea (Pisum) species and subspecies. Indian J Genet Plant Breed 74:50–55. https://doi.org/10.5958/j.0975-6906.74.1.007
Kosterin OE, Bogdanova VS (2015) Reciprocal compatibility within the genus Pisum L. as studied in F1 hybrids: 1. Crosses involving P. sativum L. subsp. sativum. Genet Resour Crop Evol 62(5):691–709. https://doi.org/10.1007/s10722-014-0189-z
Kosterin OE, Bogdanova VS (2020) Reciprocal compatibility within the genus Pisum L. as studied in F1 hybrids: 3. Crosses involving P. abyssinicum A. Br. Genet Res Crop Evol 67(4):967–983. https://doi.org/10.1007/s10722-020-00895-7
Kosterin OE, Bogdanova VS, Gorel’ FL, Berdnikov VA (1995) Inheritance of a pollen protein and a probable case of inversion in the pea chromosome corresponding to linkage group I. Pisum Genet 27:5–8
Kosterin OE, Pukhnacheva NV, Gorel FL, Berdnikov VA (1999) Location of the breakpoints of four reciprocal translocations involving linkage group V and their influence on recombination distances between neighbouring markers. Pisum Genet 31:13–20
Kosterin OE, Zaytseva OO, Bogdanova VS, Ambrose M (2010) New data on three molecular markers from different cellular genomes in Mediterranean accessions reveal new insights into phylogeography of Pisum sativum L. subsp. elatuis (Beib.) Schmalh. Genet Resour Crop Evol 57:733–739. https://doi.org/10.1007/s10722-009-9511-6
Kosterin OE, Bogdanova VS, Galieva ER (2019) Reciprocal compatibility within the genus Pisum L. as studied in F1 hybrids: 2. Crosses involving P. fulvum Sibth. et Smith. Genet Resour Crop Evol. https://doi.org/10.1007/s10722-018-0714-6
Kosterin OE, Bogdanova VS, Mglinets AV (2020) Wild pea (Pisum sativum L. subsp. elatius Aschers. et Graebn. s. l.) at the periphery of its range: Zagros Mountains. Vavilovskii Zhurnl Genetiki i Selektsii Vavilov J Genet Breed 24(1):60–68. https://doi.org/10.18699/VJ20.596
Kreplak K, Madoui MA, Cápal P, Novák P, Labadie K, Aubert G, Bayer PE, Gali KK, Syme RA, Main D, Klein A, Bérard A, Vrbová I, Fournier C, d’Agata L, Belser C, Berrabah W, Toegelová H, Milec Z, Vrána J, Lee H, Kougbeadjo A, Térézol M, Huneau C, Turo CJ, Mohellibi N, Neumann P, Falque M, Gallardo K, McGee R, Tar’an B, Bendahmane A, Aury JM, Batley J, Le Paslier MC, Ellis N, Warkentin TD, Coyne CJ, Salse J, Edwards D, Lichtenzveig J, Macas J, Doležel J, Wincker P, Burstin J (2019) A reference genome for pea provides insight into legume genome evolution. Nat Genet 51:1411–1422. https://doi.org/10.1038/s41588-019-0480-1
Lamm R (1951) Cytogenetical studies of translocations in Pisum. Hereditas 37:356–372. https://doi.org/10.1111/j.1601-5223.1951.tb02899.x
Makasheva RKh (1979) Kul’turnaya flora SSSR [Cultivated Flora of the USSR], vol 4, Leningrad (in Russian)
Maxted N, Ambrose M (2001) Peas (Pisum L.). In: Maxted N, Bennett SJ (eds) Plant genetic resources of legumes in the Mediterranean. Current plant science and biotechnology in agriculture, vol 39. Kluwer Academic Publishers, Dordrecht, pp 181–190
Postel Z, Touzet P (2020) Cytonuclear genetic incompatibilities in plant speciation. Plants 9(4):487. https://doi.org/10.3390/plants9040487
Schaefer H, Hechenleitner P, Santos-Guerra A, Menezes de Sequeira M, Pennington RT, Kenicer G, Carine MA (2012) Systematics, biogeography, and character evolution of the legume tribe Fabeae with special focus on the middle-Atlantic island lineages. BMC Evol Biol 12:250. https://doi.org/10.1186/1471-2148-12-250
Singh RJ (2003) Plant cytogenetics, 2nd edn. CRC Press, Boca Raton
Smýkal P, Aubert G, Burstin J, Coyne CJ, Ellis NT, Flavell AJ, Ford R, Hýbl M, Macas I, Neumann P, McPhee KE, Redden RJ, Rubiales D, Weller JL, Warkentin TD (2012) Pea (Pisum sativum L.) in the genomic era. Agronomy 2:74–115. https://doi.org/10.3390/agronomy2020074
Smýkal P, Trněný O, Brus J, Hanáček P, Rathore A, Roma RD, Pechanek V, Douchoslav M, Battacharrya D, Bariotakis M, Pirintsos S, Berger J, Toker C (2018) Genetic structure of wild pea (Pisum sativum subsp. elatius) populations in the northern part of the Fertile Crescent reflects moderate cross-pollination and strong effect of geographic but not environmental distance. PLoS ONE 13(3):e0194056. https://doi.org/10.1371/journal.pone.0194056
Soldano A (1992) Riproposizione di taxa sottospecifici prioritari dovuti a botanici italiani. Natura Bresciana (Ann Mus Civ Sci Nat Brescia) 27:51–56
Sutton AW (1914) Results obtained by crossing a wild pea from Palestine with commercial types. J Linn Soc Bot 42:427–433
Vershinin AV, Allnutt TR, Knox MR, Ambrose MJ (2003) Transposable elements reveal the impact of introgression, rather than transposition, in Pisum diversity, evolution, and domestication. Mol Biol Evol 20:2067–2075. https://doi.org/10.1093/molbev/msg220
Weeden NF, Brauner S, Przyborowski JA (2002) Genetic analysis of pod dehiscence in pea (Pisum sativum L.). Cell Mol Biol Lett 7:657–663. https://doi.org/10.2307/2332510
Welch BL (1947) The generalisation of “Student’s” problem when several different population variances are involved. Biometrika 34:28–35. https://doi.org/10.2307/2332510
Zaytseva OO, Bogdanova VS, Kosterin OE (2012) Phylogenetic reconstruction at the species and intraspecies levels in the genus Pisum L. (peas) using a histone H1 gene. Gene 504:192–202. https://doi.org/10.1016/j.gene.2012.05.026
Zaytseva OO, Gunbin KV, Mglinets AV, Kosterin OE (2015) Divergence and population traits in evolution of the genus Pisum L. as reconstructed using genes of two histone H1 subtypes showing different phylogenetic resolution. Gene 556:235–244. https://doi.org/10.1016/j.gene.2014
Zaytseva OO, Bogdanova VS, Mglinets AV, Kosterin OE (2017) Refinement of the collection of wild peas (Pisum L.) and search for the area of pea domestication with a deletion in the plastidic psbA-trnH spacer. Genet Res Crop Evol 64:1417–1430. https://doi.org/10.1007/s10722-016-0446-4
Zohary D, Hopf M (2000) Domestication of plants in the old world, 3rd edn. Clarendon Press, Oxford
Acknowledgements
The work was supported by the Russian State Scientific Project № 0259-2021-0012 and the Project 19-04-00162 of the Russian Fund for Fundamental Research. We thank Lyudmila P. Romkina for care of the experimental plants. The plants were grown in the Artificial Plant Growing Facility, ICG SB RAS.
Funding
The work was supported by the Russian State Scientific Project № 0259-2021-0012 and the Project 19-04-00162 of the Russian Fund for Fundamental Research.
Author information
Authors and Affiliations
Contributions
Both authors equally contributed to all parts of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest or competing interests to declare that are relevant to the content of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kosterin, O.E., Bogdanova, V.S. Reciprocal compatibility within the genus Pisum L. as studied in F1 hybrids: 4. Crosses within P. sativum L. subsp. elatius (Bieb.) Aschers. et Graebn.. Genet Resour Crop Evol 68, 2565–2590 (2021). https://doi.org/10.1007/s10722-021-01151-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-021-01151-2