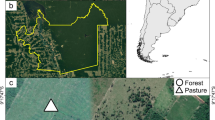
Abstract
The Altitudinal Atlantic Forest is comprised of humid forest enclaves on top of the mountains surrounded by the Caatinga's xeric shrubland vegetation. It is considered a humidity refuge of endemism in semi-arid regions, and it is under intense anthropogenic pressure. Dung beetle communities are widely used as an ecological indicator of environmental conservation. However, dung beetle communities of the Altitudinal Atlantic Forest are poorly studied, and their value for biodiversity conservation in semi-arid regions is unknown. We describe and compare dung beetle communities of Altitudinal Atlantic Forest remnants located in the Brazilian Northeast semi-arid region to evaluate whether diversity patterns and species composition differ among the remnants. We captured 1,333 beetles belonging to 24 species. Only one remnant showed a statistically significant difference in diversity based on species richness and abundance, and beta diversity showed a high dissimilarity among remnants. The remnants had in common only one species, and the dominant species were distinct among them. Species composition was 16% similar among the sampled remnants, and we registered species commonly found in the Atlantic Forest, the Caatinga, and Amazonia. The structure of the dung beetle community in each remnant could be related to the historical origin and maintenance of the Altitudinal Atlantic Forest. Species compositions changed as a result of geographic location and surrounding environments, which act as filters to dung beetle species flow and establishment. We highlight the importance of the Altitudinal Atlantic Forest for the conservation and maintenance of biodiversity in the semi-arid region.




Similar content being viewed by others
References
Andrade-Lima D (1982) Present-day forest refuges in northeastern Brazil. In: Andrade-Lima D, Prance GT (eds) Biological diversification in the tropics, 1st edn. Columbia University Press, Nova York, pp 245–251
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity: Partitioning beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A (2012) The relationship between species replacement, dissimilarity derived from nestedness, and nestedness. Glob Ecol Biogeogr 21:1223–1232. https://doi.org/10.1111/j.1466-8238.2011.00756.x
Baselga A (2013) Separating the two components of abundance‐based dissimilarity: balanced changes in abundance vs. abundance gradients. Methods Ecol Evol 4:552–557. https://doi.org/10.1111/2041-210X.12029
Baselga A, Orme CDL (2012) betapart: an R package for the study of beta diversity. Methods Ecol Evol 3:808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Calderón-Patrón JM, Moreno CE (2019) Diversidad beta basada en indices de disimilitud: Su partición en componentes de recambio y diferencias en riqueza. In: Moreno CE (ed) La biodiversidad en un mundo cambiante: Fundamentos teóricos y metodológicos para su estudio, 1st edn. Universidad Autónoma del Estado de Hidalgo/Libermex, Ciudad de México, pp 203–222
Chao A, Gotelli NJ, Hsieh T et al (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67. https://doi.org/10.1890/13-0133.1
Chao A, Jost L (2012) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547. https://doi.org/10.1890/11-1952.1
Chao A, Ma K, Hsieh T (2016) iNEXT (iNterpolation and EXTrapolation) online: software for interpolation and extrapolation of species diversity. Program and user’s guide. Methods Ecol Evol 7:1451–1456. https://doi.org/10.1111/2041-210X.12613
Clarke K, Gorley R (2006) Primer. PRIMER-e, Plymouth
Colwell RK, Chao A, Gotelli NJ et al (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. Plant Ecol 5:3–21. https://doi.org/10.1093/jpe/rtr044
Costa CMQ, Silva FAB, Farias ÂID, Moura RDCD (2009) Diversidade de Scarabaeinae (Coleoptera: Scarabaeidae) coletados com armadilha de interceptação de vôo no Refúgio Ecológico Charles Darwin, Igarassu-PE. Brasil Rev Bras Entomol 53:88–94. https://doi.org/10.1590/S0085-56262009000100021
Costa FC, Pessoa KKT, Liberal CN et al (2013) What is the importance of open habitat in a predominantly closed forest area to the dung beetle (Coleoptera, Scarabaeinae) assemblage ? Rev Bras Entomol 57:329–334. https://doi.org/10.1590/S0085-56262013000300012
da Silva PG, Lobo JM, Hensen MC, Vaz-de-Mello FZ, Hernández MIM (2018) Turnover and nestedness in subtropical dung beetle assemblages along an elevational gradient. Divers Distrib 24:1277–1290. https://doi.org/10.1111/ddi.12763
Edmonds WD, Zídek J (2010) A taxonomic review of the neotropical genus Coprophanaeus Olsoufieff, 1924 (Coleoptera: Scarabaeidae, Scarabaeinae). Insecta Mundi 0129:1–111
Endres AA, Creão-duarte AJ, Isabel M, Hernández M (2007) Diversidade de Scarabaeidae s. str. (Coleoptera) da Reserva Biológica Guaribas, Mamanguape, Paraíba, Brasil: uma comparação entre Mata Atlântica e Tabuleiro Nordestino. Rev Bras Entomol 51:67–71
Faith DP, Minchin PR, Belbin L (1987) Compositional dissimilarity as a robust measure of ecological distance. Vegetatio 69:57–68. https://doi.org/10.1007/BF00038687
Filgueiras BK, Iannuzzi L, Vaz-de-Mello FZ (2011) First report of Oxysternon silenus Castelnau (Scarabaeidae, Scarabaeinae, Phanaeini) in the Brazilian Atlantic Forest. Rev Bras Entomol 55:283–328. https://doi.org/10.1590/S0085-56262011005000010
Filho GAP, Montingelli GG (2011) Check list of snakes from the Brejos de Altitude of Paraíba and Pernambuco, Brazil. Biota Neotrop 11:145–151. https://doi.org/10.1590/S1676-06032011000300011
Gardner TA, Hernández MIM, Barlow J, Peres CA (2008) Understanding the biodiversity consequences of habitat change : the value of secondary and plantation forests for Neotropical dung beetles. J Appl Ecol 45:883–893. https://doi.org/10.1111/j.1365-2664.2008.01454.x
Gomes MA (2004) Parque Ecológico Vasconcelos Sobrinho e a reprodução socioambiental do insustentável. In: Pôrto KC, Cabral JJP, Tabarelli M (eds) Brejos de altitude em Pernambuco e Paraíba: história natural, ecologia e conservação, 1st edn. Ministério do Meio Ambiente e Universidade Federal de Pernambuco, Brasília-Brasil, pp 49–78
Halffter G (1991) Historical and ecological factors determining the geographical distribution of beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Biogeographia 15:11–40. https://doi.org/10.21426/B615110376
Halffter G, Arellano L (2002) Response of dung beetle diversity to human-induced changes in a tropical landscape. Biotropica 34:144–154. https://doi.org/10.1111/j.1744-7429.2002.tb00250.x
Halffter G, Favila ME (1993) The Scarabaeinae an animal group for analysing, inventorying and monitoring biodiversity in tropical rainforest and modified landscapes. Biol Intl 27:15–21
Halffter G, Matthews EG (1966) The natural history of dung beetles of the subfamily Scarabaeinae (Coleoptera, Scarabaeidae), 1st edn. Folia Entomol. Mex, Mexico
Hanski I, Cambefort Y (1991) Dung Beetle Ecology, 1st edn. Princeton University Press, Princeton
Hernández MI, Barreto PS, Costa VH et al (2014) Response of a dung beetle assemblage along a reforestation gradient in Restinga forest. J Insect Conserv 18:539–546. https://doi.org/10.1007/s10841-014-9645-5
Hernández MIM (2007) Besouros escarabeíneos (Coloptera: Scarabaeidae) da Caatinga paraibana, Brasil. Oecol Bras 11:356–364. https://www.researchgate.net/profile/Malva_Hernandez/publication/28224192_Besouros_escarabeineos_Coleoptera_Scarabaeidae_da_Caatinga_paraibana_Brasil/links/0c960533a4a639b6b3000000/Besourosescarabeineos-Coleoptera-Scarabaeidae-da-Caatinga-paraibana-Brasil.pdf. Accessed 26 June 2019
Hernández MIM, Da Silva PG, Niero MM, Alves VM et al (2019) Ecological characteristics of Atlantic Forest dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae) in the state of Santa Catarina, southern Brazil. Coleopts Bull 73:693–709. https://doi.org/10.1649/0010-065X-73.3.693
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–432. https://doi.org/10.2307/1934352
Jost L (2006) Entropy and diversity. Oikos 113:36–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Krell F-T, Krell-Westerwalbesloh S, Weiß I, Euggleton P, Linsenmair KE (2003) Spatial separation of Afrotropical dung beetle guilds: a trade-off between competitive superiority and energetic constraints (Coleoptera: Scarabaeidae). Ecography 26:210–222. https://doi.org/10.1034/j.1600-0587.2003.03278.x
Larsen TH, Lopera A, Forsyth A (2008) Understanding trait-dependent community disassembly: Dung beetles, density functions, and forest fragmentation. Conserv Biol 22:1288–1298. https://doi.org/10.1111/j.1523-1739.2008.00969.x
Liberal CN, Farias ÂMID, Meiado MV et al (2011) How habitat change and rainfall affect dung beetle diversity in Caatinga, a Brazilian semi-arid ecosystem. J Insect Sci 11:1–11. https://doi.org/10.1673/031.011.11401
Lumaret JP, Kirk A (1987) Ecology of dung beetles in the French Mediterranean region (Coleoptera: Scarabaeidae). Acta Zool Mex 19:1–55. https://doi.org/10.21829/azm.1987.192420381987.19242038
Medina A, Lopes P (2014) Resource utilization and temporal segregation of Scarabaeinae (Coleoptera, Scarabaeidae) community in a Caatinga fragment. Neotrop Entomol 43:127–133. https://doi.org/10.1007/s13744-014-0198-9
Moreno CE, Sánchez-Rojas G, Pineda E, Escobar F (2007) Shortcuts for biodiversity evaluation: a review of terminology and recommendations for the use of target groups, bioindicators and surrogates. Int J Environ Health 1:71–86
Nekola JC, White PS (1999) The distance decay of similarity in biogeography and ecology. J Biogeogr 26:867–878. https://doi.org/10.1046/j.1365-2699.1999.00305.x
Nichols E, Larsen T, Spector S et al (2007) Global dung beetle response to tropical forest modification and fragmentation: A quantitative literature review and meta-analysis. Biol Conserv 137:1–19. https://doi.org/10.1016/j.biocon.2007.01.023
Novais SMA, Evangelista LA, Reis-Júnior R, Neves FS (2016) How does dung beetle (Coleoptera: Scarabaeidae) diversity vary along a rainy season in a tropical dry forest? J Insect Sci 16:81. https://doi.org/10.1093/jisesa/iew069
Nunes CA, Braga RF, Figueira JEC, Neves FS, Fernandes GW (2016) Dung beetles along a tropical altitudinal gradient: environmental filtering on taxonomic and functional diversity. PLoS One 11:e0157442. https://doi.org/10.1371/journal.pone.0157442
Pinto S, Santos A, Tabarelli M (2009) Seed predation by rodents and safe sites for large-seeded trees in a fragment of the Brazilian Atlantic forest. Braz J Biol 69:763–771. https://doi.org/10.1590/S1519-69842009000400003
Prance GT (1979) The taxonomy and phytogeography of the Chrysobalanaceae of the Atlantic Coastal Forest in Brasil. Rev Bras Bot 2:19–39
R Core Team (2019) R: a language and environment for statistical computing, r foundation for statistical computing. Austria, Vienna
Ribeiro MC, Martensen AC, Metzger JP et al (2011) The Brazilian Atlantic Forest: A Shrinking Biodiversity Hotspot. In: Zachos F, Habel J (eds) Biodiversity hotspots, 1st edn. Springer, Berlin, pp 405–434
Ridgley R, Tudor G (1994) The birds of South America. Volume II: The suboscine passerines. University of Texas Press, Austin. http://books.google.com.br/books?id=FPrCwEeHGkIC&printsec=frontcover&dq=ridgely+and+tudor+1994&hl=pt-BR&sa=X&ei=IT_WT9_JMqaQ0QHUxK2SAw&ved=0CD. Accessed 5 Sept 2019
Rodal MJN, Sales MF (2007) Composição da flora vascular em um remanescente de floresta montana no semi-árido do nordeste do Brasil. Hoehnea 34:433–446. https://doi.org/10.1590/S2236-89062007000400002
Rodal MJN, Sales MF, Silva MJ, Silva AG (2005) Flora de um Brejo de Altitude na escarpa oriental do planalto da Borborema, PE. Brasil Act Bot Bras 19:843–858. https://doi.org/10.1590/S0102-33062005000400020
Rosa RS, Groth F (2004) Ictiofauna dos ecossistemas de brejos de altitude de Pernambuco e Paraíba. In: Pôrto KC, Cabral JJP, Tabarelli M (eds) Brejos de altitude em Pernambuco e Paraíba: história natural, ecologia e conservação, 1st edn. Ministério do Meio Ambiente e Universidade Federal de Pernambuco, Brasília-Brasil, pp 201–210
Salomão RP, Iannuzzi L (2017) How Do regeneration stages of Caatinga forests influence the structure of dung beetle (Coleoptera: Scarabaeidae) assemblage? Coleopts Bull 71:578–589. https://doi.org/10.1649/0010-065X-71.3.578
Santos A (2002) Distribuição de plantas lenhosas e relações históricas entre a floresta Amazônica, a floresta Atlântica costeira e os brejos de altitude do nordeste brasileiro. Dissertation, Universidade Federal de Pernambuco
Scarano FR (2012) Biomas brasileiros: retrato de um país plural. Casa da Palavra, Rio de Janeiro
Silva AP, Pontes ARM (2008) The effect of a mega-fragmentation process on large mammal assemblages in the highly-threatened Pernambuco Endemism Centre, North-eastern Brazil. Biodivers Conserv 17:1455–1464. https://doi.org/10.1007/s10531-008-9353-0
Silva F, Hernández MIM, Ide S, Moura RC (2007) Comunidade de escarabeíneos (Coleoptera, Scarabaeidae) copro-necrófagos da região de Brejo Novo, Caruaru, Pernambuco. Brasil Rev Bras entomol 51:228–233. https://doi.org/10.1590/S0085-56262007000200014
Silva FAB, Costa CMQ, Moura RC, Farias AI (2010) Study of the dung beetle (Coleoptera: Scarabaeidae) community at two sites: Atlantic forest and clear-cut, Pernambuco. Brazil Environ Entomol 39:359–367. https://doi.org/10.1603/EN09180
Silva JMC, Tabarelli M (2000) Tree species impoverishment and the future flora of the Atlantic forest of Northeast Brazil. Nature 404:72–74. https://doi.org/10.1038/35003563
Silva RJ, Ribeiro HV, Souza MF, Vaz-De-Mello FZ (2015) Influência da granulometria do solo na estrutura de guildas funcionais de besouros rola-bostas (Coleoptera: Scarabaeidae: Scarabaeinae) em florestas semideciduais no estado do Mato Grosso, Brasil. Bioscience 31:601–612. https://doi.org/10.14393/BJ-v31n1a2015-235252015-23525
Silva PGD, Di Mare RA (2012) Escarabeíneos copro-necrófagos (Coleoptera, Scarabaeidae, Scarabaeinae) de fragmentos de Mata Atlântica em Silveira Martins, Rio Grande do Sul, Brasil. Iheringia. Sér Zool 102:197–205. https://doi.org/10.1590/S0073-47212012000200012
SOS Mata Atlântica (1993) Mapa de remanescentes da floresta Atlântica nordestina. In: Sociedade Nordestina de Ecologia, Conservation International & Fundação Biodiversitas (eds) Workshop Prioridades para a Conservação da Floresta Atlântica do Nordeste. Brazil
Sousa MAN, Langguth A, do Amaral Gimenez E (2004) Mamíferos dos brejos de altitude Paraíba e Pernambuco. In: Pôrto KC, Cabral JJP, Tabarelli M (eds) Brejos de altitude em Pernambuco e Paraíba: história natural, ecologia e conservação, 1st edn. Ministério do Meio Ambiente e Universidade Federal de Pernambuco, Brasília-Brasil, pp 229–254
Tabarelli M, Aguiar AV, Ribeiro MC et al (2010) Prospects for biodiversity conservation in the Atlantic Forest: lessons from aging human-modified landscapes. Biol Conserv 143:2328–2340. https://doi.org/10.1016/j.biocon.2010.02.005
Tabarelli M, Santos AMM (2004) Uma breve descrição sobre a história natural dos Brejos Nordestinos. In: Pôrto KC, Cabral JJP, Tabarelli M (eds) Brejos de altitude em Pernambuco e Paraíba: história natural, ecologia e conservação, 1st edn. Ministério do Meio Ambiente e Universidade Federal de Pernambuco, Brasília-Brasil, p 342
Tavares M, Rodal M, Melo A, Araújo M (2000) Fitossociologia do componente arbóreo de um trecho de floresta ombrófila montana do Parque Ecológico João Vasconcelos Sobrinho, Caruaru, Pernambuco. Naturalia 25:243–265
Teixeira D, Nacinovic J, Pontual F (1986) Notes on some birds of northeastern Brazil (2). Bull Br Ornithol Club 107:151–157
Vieira I, Louzada JNC, Spector S (2008) Effects of degradation and replacement of southern Brazilian coastal sandy vegetation on the dung beetles (Coleoptera: Scarabaeidae). Biotropica 40:719–727. https://ci.nii.ac.jp/naid/10027913359/en/. Accessed 16 Feb 2018
Vivo M (1997) Mammalian evidence of historical ecological change in the Caatinga semiarid vegetation of northeastern Brazil. J Comp Biol 2:65–73
Wolda H (1981) Similarity indices, sample size and diversity. Oecologia 50:296–302. https://doi.org/10.1007/BF00344966
Acknowledgments
We thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) by scientific initiation scholarship to JCSC and funding to RCM (CNPQ-PQ2 305298/2014-3), and LGBI (Laboratório de Genética e Biodiversidade de Insetos) team for their support with the field and laboratory work. We appreciate the comments of the evaluators, they helped to improve the document.
Funding
This study was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (grant number CNPQ-PQ2 305298/2014–3).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Janaina Camara S. da Cunha and Julliana Barretto. Funding acquisition and resources: Rita de Cássia Moura and Fernando Silva. Julliana Barretto wrote the manuscript first draft, and all authors commented on previous versions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Barretto, J., da Cunha, J.C.S., Silva, F. et al. Dung beetle communities of altitudinal Atlantic forest remnants: diversity and composition. Int J Trop Insect Sci 41, 2873–2881 (2021). https://doi.org/10.1007/s42690-021-00471-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-021-00471-1