Abstract
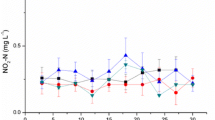
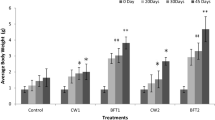
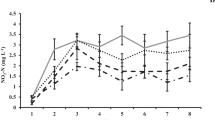
A 120-day experiment was conducted to understand the growth and immunomodulatory response of Penaeus vannamei reared in biofloc-integrated periphyton system–like natural substrates like bamboo (NS1), sugarcane bagasse (NS2), artificial substrates like polyvinyl chloride mat (AS1), agricultural shed net (AS2), and a control group without biofloc and substrate. Experiments were carried out in 500-L fiber-reinforced plastic (FRP) tanks at a stocking density of 100 nos/m3 (0.256 g). Shrimps reared in natural substrate-based treatments, (NS1 and NS2) recorded highest average body weight (ABW) and survival followed by artificial ones (AS1 and AS2), and were significantly higher (P < 0.01) compared with control. Significantly higher chlorophylla, ash-free dry matter (AFDM), and THB load were recorded in NS (1 and 2) followed by AS (1 and 2). Contrastingly, the Vibrio load was higher in control than treatments. Relative percentage survival was higher (P < 0.01) in treatments when challenged against V. parahaemolyticus. Serum protein level, phenoloxidase activity, and superoxide dismutase assay were significantly higher in natural and artificial substrate groups when compared to control. Immunomodulatory effects through immune gene study revealed a two fold to four fold upregulation in cytMn-SOD and mtMn-SOD genes in NS groups. Similarly, the lysozyme and prophenoloxidase genes were significantly upregulated in the treatment groups. The results conclude that biofloc with natural followed by artificial periphytic substrates were observed to deliver better performances, protective response, and immunomodulatory effects in pacific white shrimp P. vannamei culture.




Similar content being viewed by others
References
Anand PS, Kohli MP, Roy SD, Sundaray JK, Kumar S, Sinha A, Pailan GH, Kumar SM (2013) Effect of dietary supplementation of periphyton on growth performance and digestive enzyme activities in Penaeus monodon. Aquaculture 392:59–68. https://doi.org/10.1016/j.aquaculture.2013.01.029
Anand PS, Kohli MP, Kumar S, Sundaray JK, Roy SD, Venkateshwarlu G, Sinha A, Pailan GH (2014) Effect of dietary supplementation of biofloc on growth performance and digestive enzyme activities in Penaeus monodon. Aquaculture 418:108–115. https://doi.org/10.1016/j.aquaculture.2013.09.051
APHA (1995) Standard methods for the examination of water and wastewater. Washington, USA, 12th ed., DC
APHA (1998) Standard methods for the examination of water and wastewater. Washington, USA, 20th ed., DC
Araneda M, Pérez EP, Gasca-Leyva E (2008) White shrimp Penaeus vannamei culture in freshwater at three densities: condition state based on length and weight. Aquaculture 283:13–18. https://doi.org/10.1016/j.aquaculture.2008.06.030
Arnold SJ, Sellars MJ, Crocos PJ, Coman GJ (2006) Intensive production of juvenile tiger shrimp Penaeus monodon: an evaluation of stocking density and artificial substrates. Aquaculture 261:890–896. https://doi.org/10.1016/j.aquaculture.2006.07.036
Asaduzzaman M, Wahab MA, Verdegem MC, Huque S, Salam MA, Azim ME (2008) C/N ratio control and substrate addition for periphyton development jointly enhance freshwater prawn Macrobrachium rosenbergii production in ponds. Aquaculture 280:117–123. https://doi.org/10.1016/j.aquaculture.2008.04.019
Asokan R, Arumugam M, Mullainadhan P (1998) Functional analysis of plasma prophenoloxidase system in the marine mussel Perna viridis. Comp Biochem Phys A 120:753–762. https://doi.org/10.1016/S1095-6433(98)10098-3
Audelo-Naranjo JM, Martínez-Córdova LR, Voltolina D, Gómez-Jiménez S (2011) Water quality, production parameters and nutritional condition of Litopenaeus vannamei (Boone, 1931) grown intensively in zero water exchange mesocosms with artificial substrates. Aquac Res 42:1371–1377. https://doi.org/10.1111/j.1365-2109.2010.02725.x
Avnimelech Y (2011) Tilapia production using biofloc technology (BFT). In: Proc 9th Int Symposium on Tilapia in Aquaculture, pp 362–366
Azim ME, Wahab MA (2005) 1 2 Periphyton-based pond polyculture. Periphyton: Ecology, exploitation and management. 18:207
Azim ME, Verdegem MC, Khatoon H, Wahab MA, Van Dam AA, Beveridge MC (2002) A comparison of fertilization, feeding and three periphyton substrates for increasing fish production in freshwater pond aquaculture in Bangladesh. Aquaculture 212:227–243. https://doi.org/10.1016/S0044-8486(02)00093-5
Ballester EL, WasieleskyJr W, Cavalli RO, Abreu PC (2007) Nursery of the pink shrimp Farfantepenaeus paulensis in cages with artificial substrates: biofilm composition and shrimp performance. Aquaculture 269:355–362. https://doi.org/10.1016/j.aquaculture.2007.04.003
Banerjee S, Khatoon H, Shariff M, Yusoff FM (2010) Enhancement of Penaeus monodon shrimp postlarvae growth and survival without water exchange using marine Bacillus pumilus and periphytic microalgae. Fisheries Sci 76:481–487. https://doi.org/10.1007/s12562-010-0230-x
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Campa-Córdova AI, Hernández-Saavedra NY, Ascencio F (2002) Superoxide dismutase as modulator of immune function in American white shrimp (Litopenaeus vannamei). CompBiochemPhys C 133:557–565. https://doi.org/10.1016/S1532-0456(02)00125-4
Chiu CH, Guu YK, Liu CH, Pan TM, Cheng W (2007) Immune responses and gene expression in white shrimp, Litopenaeus vannamei, induced by Lactobacillus plantarum. Fish Shellfish Immunol 23:364–377. https://doi.org/10.1016/j.fsi.2006.11.010
Crab R, Kochva M, Verstraete W, Avnimelech Y (2009) Bio-flocs technology application in over-wintering of tilapia. Aquac Eng 40:105–112. https://doi.org/10.1016/j.aquaeng.2008.12.004
Dash P, Avunje S, Tandel RS, Panigrahi A (2017) Biocontrol of luminous vibriosis in shrimp aquaculture: a review of current approaches and future perspectives. Rev Fish Sci Aquac 25:245–255. https://doi.org/10.1080/23308249.2016.1277973
Ekasari J, Azhar MH, Surawidjaja EH, Nuryati S, De Schryver P, Bossier P (2014) Immune response and disease resistance of shrimp fed biofloc grown on different carbon sources. Fish Shellfish Immunol 41:332–339. https://doi.org/10.1016/j.fsi.2014.09.004
Elnady MA, Alkobaby AI, Salem MA, Abdel-Salam M, Asran BM (2010) Effect of fertilization and low quality feed on water quality dynamics and growth performance of Nile tilapia (Oreochromis niloticus). J Am Sc 6:1044–1054 http://www.jofamericanscience.org/journals/amsci/am0610/123_3693am0610_1044_1054.pdf
Fagutao FF, Koyama T, Kaizu A, Saito-Taki T, Kondo H, Aoki T, Hirono I (2009) Increased bacterial load in shrimp hemolymph in the absence of prophenoloxidase. FEBS J 276:5298–5306. https://doi.org/10.1111/j.1742-4658.2009.07225.x
Fan L, Chen J, Liu Q, Wu W, Meng S, Song C (2015) Exploration of three heterotrophic nitrifying strains from a tilapia pond for their characteristics of inorganic nitrogen use and application in aquaculture water. J Biosci 119:303–309. https://doi.org/10.1016/j.jbiosc.2014.09.006
Ferreira GS, Bolívar NC, Pereira SA, Guertler C, do Nascimento Vieira F, Mouriño JL, Seiffert WQ (2015) Microbial biofloc as source of probiotic bacteria for the culture of Litopenaeus vannamei. Aquaculture 448:273–279. https://doi.org/10.1016/j.aquaculture.2015.06.006
Ferreira LM, Lara G, WasieleskyJr W, Abreu PC (2016) Biofilm versus biofloc: are artificial substrates for biofilm production necessary in the BFT system? Aquacult int 24:921–930. https://doi.org/10.1007/s10499-015-9961-0
González-Félix ML, Gómez-Jiménez S, Perez-Velazquez M, Davis DA, Velazco-Rameños JG (2007) Nitrogen budget for a low salinity, zero-water exchange culture system: I. effect of dietary protein level on the performance of Litopenaeus vannamei (Boone). Aquac Res 38:798–808. https://doi.org/10.1111/j.1365-2109.2007.01657.x
Green BW, Schrader KK, Perschbacher PW (2014) Effect of stocking biomass on solids, phytoplankton communities, common off-flavors, and production parameters in a channel catfish biofloc technology production system. Aquac Res 45:1442–1458. https://doi.org/10.1111/are.12096
Halfhide T, Åkerstrøm A, Lekang OI, Gislerød HR, Ergas SJ (2014) Production of algal biomass, chlorophyll, starch and lipids using aquaculture wastewater under axenic and non-axenic conditions. Algal Res 6:152–159. https://doi.org/10.1016/j.algal.2014.10.009
Hernández-López J, Gollas-Galván T, Vargas-Albores F (1996) Activation of the prophenoloxidase system of the brown shrimp Penaeus californiensis Holmes. Comp Biochem PhysC 113:61–66 Activation_of_the_prophenoloxidase_syste20160331-4020-1f6k76u.pdf
Hose JE, Martin GG, Gerard AS (1990) A decapod haemocyte classification scheme integrating morphology, cytochemistry, and function. Biol Bull 178:33–45 http://www.jstor.org/stable/1541535
Hultmark D, Steiner H, Rasmuson T, Boman HG (1980) Insect immunity. Purification and properties of three inducible bactericidal proteins from hemolymph of immunized pupae of Hyalophora cecropia. Eur J Biochem 106:7–16. https://doi.org/10.1111/j.1432-1033.1980.tb05991.x
Iwanaga S, Lee BL (2005) Recent advances in the innate immunity of invertebrate animals. BMB Rep 38:128–150. https://doi.org/10.5483/BMBRep.2005.38.2.128
Ji PF, Yao CL, Wang ZY (2011) Reactive oxygen system plays an important role in shrimp Litopenaeus vannamei defense against Vibrio parahaemolyticus and WSSV infection. Dis Aquat Org 96:9–20. https://doi.org/10.3354/dao02373
Jiravanichpaisal P, Lee BL, Söderhäll K (2006) Cell-mediated immunity in arthropods: hematopoiesis, coagulation, melanization and opsonization. Immunobiology 211:213–236. https://doi.org/10.1016/j.imbio.2005.10.015
Ju ZY, Forster I, Conquest L, Dominy W, Kuo WC, David Horgen F (2008) Determination of microbial community structures of shrimp floc cultures by biomarkers and analysis of floc amino acid profiles. Aquac Res 39:118–133. https://doi.org/10.1111/j.1365-2109.2007.01856.x
Khatoon H, Banerjee S, Yusoff FM, Shariff M (2009) Evaluation of indigenous marine periphytic Amphora, Navicula and Cymbella grown on substrate as feed supplement in Penaeus monodon postlarval hatchery system. Aquac Nutr 15:186–193. https://doi.org/10.1111/j.1365-2095.2008.00582.x
Kim SK, Pang Z, Seo HC, Cho YR, Samocha T, Jang IK (2014) Effect of bioflocs on growth and immune activity of Pacific white shrimp, Litopenaeus vannamei postlarvae. Aquaculture Research 45:362–371. https://doi.org/10.1111/are.12319
Kuhn DD, Lawrence AL, Boardman GD, Patnaik S, Marsh L, Flick GJ Jr (2010) Evaluation of two types of bioflocs derived from biological treatment of fish effluent as feed ingredients for Pacific white shrimp, Litopenaeus vannamei. Aquaculture 303:28–33. https://doi.org/10.1016/j.aquaculture.2010.03.001
Kumar S, Anand PS, Ravichandran P, Panigrahi A, Dayal JS, Raja RA, Deo AD, Ghoshal TK, Ponniah AG (2015) Effect of periphyton on microbial dynamics, immune responses and growth performance in black tiger shrimp Penaeus monodon Fabricius, 1798. Indian J Fish 62:67–74 http://krishi.icar.gov.in/jspui/handle/123456789/11182
Kumar VS, Pandey PK, Anand T, Bhuvaneswari R, Kumar S (2017) Effect of periphyton (aquamat) on water quality, nitrogen budget, microbial ecology, and growth parameters of Litopenaeus vannamei in a semi-intensive culture system. Aquaculture 479:240–249. https://doi.org/10.1016/j.aquaculture.2017.05.048
Lee C, Kim S, Lim SJ, Lee KJ (2017) Supplemental effects of biofloc powder on growth performance, innate immunity, and disease resistance of Pacific white shrimp Litopenaeus vannamei. Fish Aquat Sc 20:15. https://doi.org/10.1186/s41240-017-0059-7
Li J, Tan B, Mai K (2009) Dietary probiotic Bacillus OJ and isomaltooligosaccharides influence the intestine microbial populations, immune responses and resistance to white spot syndrome virus in shrimp (Litopenaeus vannamei). Aquaculture 291:35–40. https://doi.org/10.1016/j.aquaculture.2009.03.005
Little TJ, Hultmark D, Read AF (2005) Invertebrate immunity and the limits of mechanistic immunology. Nat Immunol 6:651. https://doi.org/10.1038/ni1219
Liu CH, Yeh ST, Cheng SY, Chen JC (2004) The immune response of the white shrimp Litopenaeus vannamei and its susceptibility to Vibrio infection in relation with the moult cycle. Fish Shellfish Immunol 16:151–161. https://doi.org/10.1016/S1050-4648(03)00058-5
Moss KR, Moss SM (2004) Effects of artificial substrate and stocking density on the nursery production of pacific white shrimp Litopenaeus vannamei. J World Aquacult Soc 35:536–542. https://doi.org/10.1111/j.1749-7345.2004.tb00121.x
MPEDA (2017–2018) https://mpeda.gov.in/wp-content/uploads/2020/11/1550120514MPEDAAR201718.pdf
Naylor RL, Goldburg RJ, Mooney H, Beveridge M, Clay J, Folke C, Kautsky N, Lubchenco J, Primavera J, Williams M (1998) Nature’s subsidies to shrimp and salmon farming. Science 282:883–884. https://doi.org/10.1126/science.282.5390.883
NRC (2011) Nutrient requirements of fish and shrimp. The National Academies Press, Washington, DC. https://doi.org/10.17226/13039
Otta SK, Arulraj R, EzhilPraveena P, Manivel R, Panigrahi A, Bhuvaneswari T, Ravichandran P, Jithendran KP, Ponniah AG (2014) Association of dual viral infection with mortality of Pacific white shrimp (Litopenaeus vannamei) in culture ponds in India. Virus Disease 25:63–68. https://doi.org/10.1007/s13337-013-0180-x
Panigrahi A, Kiron V, Puangkaew J, Kobayashi T, Satoh S, Sugita H (2005) The viability of probiotic bacteria as a factor influencing the immune response in rainbow trout Oncorhynchus mykiss. Aquaculture 243(1–4):241–254. https://doi.org/10.1016/j.aquaculture.2004.09.032
Panigrahi A, Viswanath K, Satoh S (2011) Real-time quantification of the immune gene expression in rainbow trout fed different forms of probiotic bacteria Lactobacillus rhamnosus. Aquac Res 42(7):906–917
Panigrahi A, Sundaram M, Ravichandran P, Gopal C (2014) Microbial soup-eco based approach forshrimp culture and management. ENVIS Newsletter 12:3–6 http://krishi.icar.gov.in/jspui/handle/123456789/12139
Panigrahi A, Sundaram M, Jebha J, Otta SK, Bhuvaneshwari T, Saraswathy R, Rajababu D, Saranya C, Gopal C, Ravichandran P (2017) Biofloc based nutrient dense culture system for nursery and grow-out farming of Pacific white shrimp Penaeus vannamei Boone, 1931. Indian J Fish 64:22–32 http://krishi.icar.gov.in/Publication/handle/123456789/11253
Panigrahi A, Saranya C, Sundaram M, Kannan SV, Das RR, Kumar RS, Rajesh P, Otta SK (2018) Carbon: nitrogen (C: N) ratio level variation influences microbial community of the system and growth as well as immunity of shrimp (Litopenaeus vannamei) in biofloc based culture system. Fish Shellfish Immunol 81:329–337. https://doi.org/10.1016/j.fsi.2018.07.035
Panigrahi A, Sundaram M, Saranya C, Kumar RS, Dayal JS, Saraswathy R, OttaSK APS, Rekha PN, Gopal C (2019a) Influence of differential protein levels of feed on production performance and immune response of pacific white leg shrimp in a biofloc–based system. Aquaculture 503:118–127. https://doi.org/10.1016/j.aquaculture.2018.12.036
Panigrahi A, Sundaram M, Saranya C, Satishkumar R, Syama Dayal J, Gopal C (2019b) Effect of carbon and nitrogen ratio (C: N) manipulation on the production performance and immunity of Pacific white shrimp Litopenaeus vannamei (Boone, 1931) in a biofloc-based rearing system. Aquac Res 50(1):29–41
Panigrahi A, Sundaram M, Saranya C, Swain S, Dash RR, Dayal JS (2019c) Carbohydrate sources deferentially influence growth performances, microbial dynamics and immunomodulation in Pacific white shrimp (Litopenaeus vannamei) under biofloc system. Fish & shellfish immunology 86:1207–1216
Panigrahi A, Esakkiraj P, Jayashree S, Saranya C, Das RR, Sundaram M (2019d) Colonization of enzymatic bacterial flora in biofloc grown shrimp Penaeus vannamei and evaluation of their beneficial effect. Aquaculture International 27(6):1835–1846. https://doi.org/10.1016/j.aquaculture.2018.12.036
Panigrahi A, Das RR, Saranya C, Sivakumar MR, Saravanan A, Kumaraguru Vasagam KP, Mahalakhsmi P, Kannappan K, Gopikrishna G (2020) Bio-augmentation of heterotrophic bacteria in biofloc system improves growth, survival, and immunity of Indian white shrimp Penaeus indicus. Fish Shellfish Immunol. https://doi.org/10.1016/j.fsi.2020.01.021
Panjaitan P (2011) Effect of C: N ratio levels on water quality and shrimp production parameters in Penaeus monodon shrimp culture with limited water exchange using molasses as a carbon source. ILMU KELAUTAN: Indones J Mar Sc 16:1–8. https://doi.org/10.14710/ik.ijms.16.1.1-8
Pervin R, Haque MR, Akter M (2014) Effects of C/N controlled periphyton based organic farming of freshwater prawn on water quality parameters and biotic factors. J Fish 2:125–134. https://doi.org/10.17017/jfish.v2i2.2014.37
Qi C, Wang L, Liu M, Jiang K, Wang M, Zhao W, Wang B (2017) Transcriptomic and morphological analyses of Litopenaeus vannamei intestinal barrier in response to Vibrio parahemolyticus infection reveals immune response signatures and structural disruption. Fish Shellfish Immunol 70:437–450. https://doi.org/10.1016/j.fsi.2017.09.004
Rai S, Yi Y (2012) Nibbling frequency of carps in periphyton-based aquaculture systems with and without supplemental feed. Isr J Aquacult 6:4818–4822 http://hdl.handle.net/10524/31835
Ramesh MR, Shankar KM, Mohan CV (1999) Varghese TJ Comparison of three plant substrates for enhancing carp growth through bacterial biofilm. Aquacult Eng 19:119-131. https://doi.org/10.1016/S0144-8609(98)00046-6
Rao R, Zhu YB, Alinejad T, Tiruvayipati S, Thong KL, Wang J, Bhassu S (2015) RNA-seq analysis of Macrobrachium rosenbergii hepatopancreas in response to Vibrio parahaemolyticus infection. Gut Pathog 7:6. https://doi.org/10.1186/s13099-015-0052-6
Rezende PC, Schleder DD, da Silva HV, Henriques FM, de Lorenzo MA, Seiffert WQ, Andreatta ER, do Nascimento Vieira F (2018) Prenursery of the Pacific white shrimp in a biofloc system using different artificial substrates. Aquac Eng 82:25–30. https://doi.org/10.1016/j.aquaeng.2018.04.001
Samocha TM, Patnaik S, Speed M, Ali AM, Burger JM, Almeida RV, Ayub Z, Harisanto M, Horowitz A, Brock DL (2007) Use of molasses as carbon source in limited discharge nursery and grow-out systems for Litopenaeus vannamei. AquacultEng 36:184–191. https://doi.org/10.1016/j.aquaeng.2006.10.004
Schrader KK, Green BW, Perschbacher PW (2011) Development of phytoplankton communities and common off-flavors in a biofloc technology system used for the culture of channel catfish (Ictalurus punctatus). Aquac Eng 5:118–126. https://doi.org/10.1016/j.aquaeng.2011.08.004
Schveitzer R, Arantes R, Costódio PF, do Espírito Santo CM, Arana LV, Seiffert WQ, Andreatta ER (2013) Effect of different biofloc levels on microbial activity, water quality and performance of Litopenaeus vannamei in a tank system operated with no water exchange. Aquac Eng 56:59–70. https://doi.org/10.1016/j.aquaeng.2013.04.006
Smith VH, Tilman GD, Nekola JC (1999) Eutrophication: impacts of excess nutrient inputs on freshwater, marine, and terrestrial ecosystems. Environ Pollut 100:179–196. https://doi.org/10.1016/S0269-7491(99)00091-3
Söderhall K, Smith VJ (1983) Separation of the haemocyte of Carcinus maenas and other decapod crustaceans and phenoloxidase distribution. Dev Comp Immunol 7:229–239. https://doi.org/10.1016/0145-305X(83)90004-6
Sundaram M, Panigrahi A, Ganesh J, Rekha PN, Sivagnanam S, Rajamanickam S, Gopal C (2017) Evaluation of different probiotic strains for growth performance and immunomodulation in Pacific white shrimp Penaeus vannamei Boone, 1931. Indian J Fish 64:89–96 http://krishi.icar.gov.in/jspui/handle/123456789/12156
Sung HH, Chang HJ, Her CH, Chang JC, Song YL (1998) Phenoloxidase activity of haemocytes derived from Penaeus monodon and Macrobrachium rosenbergii. J Invertebrpatho l71:26–33. https://doi.org/10.1006/jipa.1997.4703
Supamattaya K, Kiriratnikom S, Boonyaratpalin M, Borowitzka L (2005) Effect of a Dunaliellaextract on growth performance, health condition, immune response and disease resistance in black tiger shrimp (Penaeus monodon). Aquaculture 248:207–216. https://doi.org/10.1016/j.aquaculture.2005.04.014
Tepaamorndech S, Chantarasakha K, Kingcha Y, Chaiyapechara S, Phromson M, Sriariyanun M, Kirschke CP, Huang L, Visessanguan W (2019) Effects of Bacillus aryabhattai TBRC8450 on vibriosis resistance and immune enhancement in Pacific white shrimp, Litopenaeus vannamei. Fish Shellfish immunol 86:4–13. https://doi.org/10.1016/j.fsi.2018.11.010
Thompson FL, Abreu PC, Wasielesky W (2002) Importance of biofilm for water quality and nourishment in intensive shrimp culture. Aquaculture 203:263–278. https://doi.org/10.1016/S0044-8486(01)00642-1
Tseng DY, Ho PL, Huang SY, Cheng SC, Shiu YL, Chiu CS, Liu CH (2009) Enhancement of immunity and disease resistance in the white shrimp, Litopenaeus vannamei, by the probiotic, Bacillus subtilis E20. Fish Shellfish Immunol 26:339–344. https://doi.org/10.1016/j.fsi.2008.12.003
van Dam AA, Beveridge MC, Azim ME, Verdegem MC (2002) The potential of fish production based on periphyton. Rev Fish Biol Fisher 12:1–31. https://doi.org/10.1023/A:1022639805031
Xu WJ, Pan LQ (2012) Effects of bioflocs on growth performance, digestive enzyme activity and body composition of juvenile Litopenaeus vannamei in zero-water exchange tanks manipulating C/N ratio in feed. Aquaculture 356:147–152. https://doi.org/10.1016/j.aquaculture.2012.05.022
Xu WJ, Morris TC, Samocha TM (2016) Effects of C/N ratio on biofloc development, water quality, and performance of Litopenaeus vannamei juveniles in a biofloc-based, high-density, zero-exchange, outdoor tank system. Aquaculture 453:169–175. https://doi.org/10.1016/j.aquaculture.2015.11.021
Zhang B, Lin W, Wang Y, Xu R (2010) Effects of artificial substrates on growth, spatial distribution and non-specific immunity factors of Litopenaeus vannamei in the intensive culture condition. Turk J Fish Aquat Sc 10:491–497 URL: http://jifro.ir/article-1-9-en.html
Zhao CS, Huang D, Peng T, Huang MZ, Xie CY, Chen J, Kong JR, Xie RC, Liu Y, Wang WN (2017) Molecular cloning, characterization and function of a germinal center kinase MST4 gene from Litopenaeus vannamei in response to Vibrio alginolyticus challenge in TLR-TRAF6 signaling pathway. Dev Comp Immunol 73:206–219. https://doi.org/10.1016/j.dci.2017.03.026
Acknowledgements
The facilities provided by the Director, ICAR-Central Institute of Brackishwater Aquaculture (CIBA) to carry out this research are gratefully acknowledged.
Funding
The authors received financial support from the National Fisheries Development Board, Hyderabad, India, and Department of Biotechnology, Ministry of Science and Technology, New Delhi, India (F. No. DBT/PR11721/AAQ/3/683/2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
The research undertaken complies with the current animal welfare laws in India. Care and treatment of the experimental animal used in this study were in accordance with the guidelines of the CPCSEA [Committee for the Purpose of Control and Supervision of Experiments on Animals, Ministry of Environment & Forests (Animal Welfare Division), Govt. of India] on care and use of animals in scientific research. The study was undertaken with approval of statutory authorities of the Central Institute of Brackishwater Aquaculture, Chennai, India. The experimental animal Penaeus vannamei is not an endangered shrimp; the provisions of the Govt. of India’s Wildlife Protection Act of 1972 are not applicable for experiments on this fish.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mani, S., Mullaivanam Ramasamy, S., Chakrapani, S. et al. The effect of natural and artificial periphytic substrates with biofloc system on shrimp Penaeus vannamei (Boone 1931) culture: growth and immune response. Aquacult Int 29, 651–668 (2021). https://doi.org/10.1007/s10499-021-00646-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00646-0