Abstract
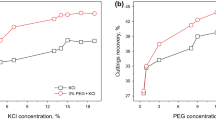
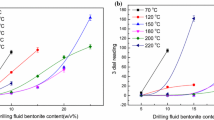
The inhibitors for shale gas can be divided into two categories, including inorganic inhibitors and organic inhibitors. The inorganic inhibitors usually include sodium chloride and potassium chloride, while the organic inhibitors include potassium sorbate, polyethylene glycol, polyetheramine, and diethylenetriamine. The inhibitive properties of these inhibitors for shale gas have been comprehensively compared in detail. The results show that the inhibitive effect of potassium chloride is much better than the other inorganic inhibitor sodium chloride, and polyetheramine exhibits the best inhibitive performance to other organic inhibitors. From the analysis of the inhibition mechanism, it can be found that both inorganic salt inhibitors and potassium sorbate, polyethylene glycol can effectively squeeze out the water molecules, adsorbed within clay layers, through ion exchange. As for polyetheramine and diethylenetriamine, the protonated ammonium ions can be adsorbed onto clay surface through electrostatic interaction to exchange hydrated sodium ions, thereby reduce the hydration repulsion of the clay.







Similar content being viewed by others
REFERENCES
Cao, C., Pu, X., Wang, G., and Huang, T., Chem. Technol. Fuels Oils, 2018, vol. 53, p. 966. https://doi.org/10.1007/s10553-018-0886-y
Bai, X., Zhang, X., Ning, T., Luo, Y., and Zhou, S., J. Inorg. Organomet. Polym. Mater. 2020, vol. 30, p. 1172. https://doi.org/10.1007/s10904-019-01270-5
Zhang, L., Li, T., Huang, L., Ye, Z., Ye, Z., Yan, X., Li, L., Deng, Q., Chen, G., Zhang, J., and Zhang, Z., Chem. Cent. J. 2018, vol. 12, p. 44. https://doi.org/10.1186/s13065-018-0410-9
Bai, X., Yang, Y., Xiao, D., Pu, X., and Wang, X., J. Appl. Polym. Sci., 2015, vol. 132, p. 41762/1. https://doi.org/10.1002/app.41762
Saboorian-Jooybari, H., Dejam, M., and Chen, Z., J. Pet. Sci. Eng., 2016, vol. 142, p. 85. https://doi.org/10.1016/j.petrol.2016.01.023
Olayiwola, S.O. and Dejam, M., Fuel, 2019, vol. 241, p. 1045. https://doi.org/10.1016/j.fuel.2018.12.122
Luo, Z., Wang, L., Yu, P., and Chen, Z., Appl. Clay Sci., 2017, vol. 150, p. 267. https://doi.org/10.1016/j.clay.2017.09.038
Jiang, G., Zhang, X., Dong, T., Xuan, Y., Wang, L., and Jiang, Q., J. Appl. Polym. Sci., 2018, vol. 135, no. 1, p. 45581/1. https://doi.org/10.1002/app.45584
Suter, J.L., Coveney, P.V., Anderson, R.L., Greenwell, H.C., and Cliffe, S., Energy Environ. Sci., 2011, vol. 4, p. 4572. https://doi.org/10.1039/c1ee01280k
van Oort, E., J. Pet. Sci. Eng., 2003, vol. 38, p. 213. https://doi.org/10.1016/S0920-4105(03)00034-2
Boulet, P., Bowden, A.A., Coveney, P.V., and Whiting, A., J. Mater. Chem., 2003, vol. 13, p. 2540. https://doi.org/10.1039/B307752G
Liu, X.-D. and Lu, X.-C., Angew. Chem., Int. Ed., 2006, vol. 118, p. 6300. https://doi.org/10.1002/anie.200601740
Naeimavi, M., Khazali, F., Abdideh, M., and Saadati, Z., Energy Sources, Part A, 2019. https://doi.org/10.1080/15567036.2019.1663303
Peng, B., Luo, P., Guo, W., and Yuan, Q., J. Appl. Polym. Sci., 2013, vol. 129, p. 1074. https://doi.org/10.1002/app.38784
Zhong, H., Huang, W., Qiu, Z., Cao, J., Xie, B., Wang, F., and Zheng, W., Energy Sources, Part A, 2015, vol. 37, p. 1971. https://doi.org/10.1080/15567036.2011.654315
Wang, L., Liu, S., Wang, T., and Sun, D.-J., Colloids Surf. A, 2011, vol. 381, p. 41. https://doi.org/10.1016/j.colsurfa.2011.03.008
Zhang, J., Hu, W., Zhang, L., Li, T., Cai, D., and Chen, G., Adsorpt. Sci. Technol., 2019, vol. 37, p. 49. https://doi.org/10.1177/0263617418809832
Tu, C., Li, N., Zhu, L., Zhou, L., Su, Y., Li, P., and Zhu, X., Polym. Chem., 2013, vol. 4, p. 393. https://doi.org/10.1039/C2PY20523H
Fan, J., Zhu, H., Li, R., and Chen, N., J. Dispersion Sci. Technol., 2015, vol. 36, p. 569. https://doi.org/10.1080/01932691.2014.917052
Hortal, A.R., Hurtado, P., Martínez-Haya, B., Arregui, A., and Bañares, L., J. Phys. Chem. B, 2008, vol. 112, p. 8530. https://doi.org/10.1021/jp802089r
Lin, J.-J., Cheng, I-J., Wang, R., and Lee, R.-J., Macromol., 2001, vol. 34, p. 8832. https://doi.org/10.1021/ma011169f
Greenwell, H.C., Harvey, M.J., Boulet, P., Bowden, A.A., Coveney, P.V., and Whiting, A., Macromol., 2005, vol. 38, p. 6189. https://doi.org/10.1021/ma0503817
Greenwell, H.C., Bowden, A.A., Chen, B., Boulet, P., Evans, J.R.G., Coveney, P.V., and Whiting, A., J. Mater. Chem., 2006, vol. 16, p. 1082. https://doi.org/10.1039/b505217c
Xi, Y., Ding, Z., He, H., and Frost, R.L., J. Colloid Interface Sci., 2004, vol. 277, p. 116. https://doi.org/10.1016/j.jcis.2004.04.053
Jiang, G., Liu, T., Ning, F., Tu, Y., Zhang, L., Yu, Y., and Kuang, L., Energies, 2011, vol. 4, no. 1, p. 140. https://doi.org/10.3390/en4010140
Zhang, S., Sheng, J., and Qiu, Z., Appl. Clay Sci., 2016, vol. 132, p. 635. https://doi.org/10.1016/j.clay.2016.08.015
Zhong, H., Qiu, Z., Sun, D., Zhang, D., and Huang, W., J. Nat. Gas Sci. Eng., 2015, vol. 26, p. 99. https://doi.org/10.1016/j.jngse.2015.05.029
Zhang, J., Hu, W., Zhang, L., Li, T., Cai, D., and Chen, G., Adsorpt. Sci. Technol., 2019, vol. 37, p. 49. https://doi.org/10.1177/0263617418809832
Bai, X., Wang, H., Luo, Y., Zheng, X., Zhang, X., Zhou, S., and Pu, X., J. Appl. Polym. Sci., 2017, vol. 134, no. 46, p. 45466/1. https://doi.org/10.1002/app.45466
Akhtarmanesh, S., Shahrabi, M.J.A., and Atashnezhad, A., J. Pet. Sci. Eng., 2013, vol. 112, p. 290. https://doi.org/10.1016/j.petrol.2013.11.017
Luo, Z., Pei, J., Wang, L., Yu, P., and Chen, Z., Appl. Clay Sci., 2017, vol. 136, p. 96. https://doi.org/10.1016/j.clay.2016.11.015
Wang, L., Liu, S.-Y., Wang, T., and Sun, D.-J., Colloids Surf. A, 2011, vol. 381, p. 41. https://doi.org/10.1016/j.colsurfa.2011.03.008
Huang, X., Shen, H., Sun, J., Lv, K., Liu, J., Dong, X., and Luo, S., ACS Appl. Mater. Interfaces, 2018, vol. 10, p. 33252. https://doi.org/10.1021/acsami.8b11419
Wu, W., Clays Clay Miner., 2001, vol. 49, p. 446. https://doi.org/10.1346/CCMN.2001.0490511
Funding
The work was supported by the National Natural Science Foundation of China (no. 51104122), and Applied Basic Research Program of Science and Technology Department of Sichuan Province (no. 2018JY0302).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest requiring disclosure in this article.
Rights and permissions
About this article
Cite this article
Bai, X., Xu, Y., Zhang, X. et al. Comparison on the Inhibitive Properties of Different Inhibitors in Water-Based Drilling Fluid. Pet. Chem. 61, 239–249 (2021). https://doi.org/10.1134/S0965544121020122
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965544121020122