Abstract
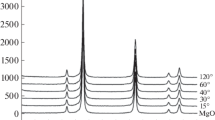
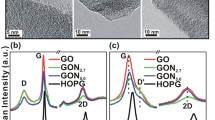
A method for synthesis of nitrogen-doped graphene (N-graphene) with high specific surface area has been developed. In this method, magnesium oxide is used as a template where a carbon layer modified with nitrogen atoms is deposited. Nitrogen–carbon–mineral composites (N–C/MgO) containing from 5 to 27 wt % of nitrogen-doped carbon have been synthesized. After dissolving magnesium oxide, N-graphene with a specific surface area of 1300–1400 m2 g–1 has been synthesized for the first time. The synthesis of N-graphene has been characterized by electron microscopy, X-ray diffraction, and X-ray photoelectron spectroscopy.








Similar content being viewed by others
REFERENCES
Novoselov, K.S., Geim, A.K., Morozov, S.V., Jiang, D., Zhang, Y., Dubonos, S.V., Grigorieva, I.V., and Firsov, A.A., Science, 2004, vol. 306, no. 5696, pp. 666–669. https://doi.org/10.1126/science.1102896
Ning, G., Fan, Z., Wang, G., Gao, J., Qian, W., and Wei, F., Chem. Commun., 2011, vol. 47, no. 21, pp. 5976–5978. https://doi.org/10.1039/c1cc11159k
Bulushev, D.A., Zacharska, M., Shlyakhova, E.V., Chuvilin, A.L., Guo, Y., Beloshapkin, S., Okotrub, A.V., and Bulusheva, L.G., ACS Catal., 2016, vol. 6, no. 2, pp. 681–691. https://doi.org/10.1021/acscatal.5b02381
Chesnokov, V.V., Kriventsov, V.V., Malykhin, S.E., Chichkan, A.S., and Podyacheva, O.Yu., J. Struct. Chem., 2018, vol. 59, no. 4, pp. 876–882. https://doi.org/10.1134/S0022476618040133
Chesnokov, V.V., Kriventsov, V.V., Malykhin, S.E., Svintsitskiy, D.A., Podyacheva, O.Y., Lisitsyn, A.S., and Richards, R.M., Diamond Relat. Mater., 2018, vol. 89, pp. 67–73. https://doi.org/10.1016/j.diamond.2018.08.007
Chesnokov, V.V., Chichkan, A.S., Bedilo, A.F., and Shuvarakova, E.I., Fullerenes Nanotubes Carbon Nanostruct., 2020, vol. 28, no. 5, pp. 402–406. https://doi.org/10.1080/1536383x.2019.1695249
Chesnokov, V.V., Chichkan, A.S., Bedilo, A.F., Shuvarakova, E.I., and Parmon, V.N., Dokl. Phys. Chem., 2019, vol. 488, pp. 154–157. https://doi.org/10.1134/S0012501619100038
Qu, L., Liu, Y., Baek, J.-B., and Dai, L., ACS Nano, 2010, vol. 4, no. 3, pp. 1321–1326. https://doi.org/10.1021/nn901850u
Zhao, L., He, R., Rim, K.T., Schiros, T., Kim, K.S., Zhou, H., Gutierrez, Ch., Chockalingam, S.P., Arguello, C.J., Palova, L., and Nordlund, D., Science, 2011, vol. 333, no. 6045, pp. 999–1003. https://doi.org/10.1126/science.1208759.
Moon, J., An, J., Sim, U., Cho, S.-P., Kang, J.H., Chung, C., Seo, J.-H., Lee, J., Nam, K.T., and Hong, B.H., Adv. Mater., 2014, vol. 26, pp. 3501–3505. https://doi.org/10.1002/adma.201306287
Rybin, M., Pereyaslavtsev, A., Vasilieva, T., Myasnikov, V., Sokolov, I., Pavlova, A., Obraztsova, E., Khomich, A., Ralchenko, V., and Obraztsova, E., Carbon, 2016, vol. 96, pp. 196–202. https://doi.org/10.1016/j.carbon.2015.09.056
Deng, D., Pan, X., Yu, L., Cui, Y., Jiang, Y., Qi, J., Li, W.-X., Fu, Q., Ma, X., Xue, Q., Sun, G., and Bao, X., Chem. Mater., 2011, vol. 23, no. 5, pp. 1188–1193. https://doi.org/10.1021/cm102666r
Reddy, A.L.M., Srivastava, A., Gowda, S.R., Gullapalli, H., Dubey, M., and Ajayan, P.M., ACS Nano, 2010, vol. 4, no. 11, pp. 6337–6342. https://doi.org/10.1021/nn101926g
Zhao, J., Zhu, G., Huang, W., He, Z., Feng, X., Ma, Y., Dong, X., Fan, Q., Wang, L., Hu, Z., Lu, Y., and Huang, W., J. Mater. Chem., 2012, vol. 22, no. 37, pp. 19679–19683. https://doi.org/10.1039/c2jm33209d
Wang, H., Maiyalagan, T., and Wang, X., ACS Catal., 2012, vol. 2, no. 5, pp. 781–794. https://doi.org/10.1021/cs200652y
Jin, Z., Yao, J., Kittrell, C., and Tour, J.M., ACS Na, vol. 5, no. 5, pp. 4112–4117. https://doi.org/10.1021/nn200766e
Pan, W., He, M., Zhang, L., Hou, Y., and Chen, C., Nanomaterials, 2019, vol. 9, no. 5, p. 798. https://doi.org/10.3390/nano9050798
Buyanov, R.A. Zakoksovanie katalizatorov (Carbonization of Catalysts). Moscow: Nauka, 1983.
Gor, G.Yu., Thommes, M., Cychosz, K.A., and Neimark, A.V., Carbon, 2012, vol. 50, pp. 1583–1590. https://doi.org/10.1016/j.carbon.2011.11.037
Thommes, M., Kaneko, K., Neimark, A.V., Olivier, J.P., Rodriguez-Reinoso, F., Rouquerol, J., and Sing, K.S.W., Pure Appl. Chem., 2015, vol. 87, pp. 1051–1069. https://doi.org/10.1515/pac-2014-1117
Chesnokov, V.V., Chichkan, A.S., Paukshtis, E.A., Svintsitskii, D.A., Ismagilov, Z.R., and Parmon, V.N., Dokl. Phys. Chem., 2017, vol. 476, part 2, pp. 186–189. https://doi.org/10.7868/S0869565217290126
Moulder, J.F., Stickle, W.F., Sobol, P.E., and Bomben, K.D., Handbook of X-ray Photoelectron Spectroscopy, Perkin-Elmer Corp., Eden Prairie, Minnesota, USA, 1992.
Yang, H., Kannappan, S., Pandian, A.S., Jang, J-H., Lee, Y.S., and Lu, W., J. Power Sources, 2015, vol. 284, pp. 146–153. https://doi.org/10.1016/j.jpowsour.2015.03.015
Arrigo, R., Schuster, M.E., Xie, Z., Yi, Y., Wowsnick, G., Sun, L.L., Hermann, K.E., Friedrich, M., Kast, P., Havecker, M., Knop-Gericke, A., and Schlogl, R., ACS Catal., 2015, vol. 5, pp. 2740–2753. https://doi.org/10.1021/acscatal.5b00094
ACKNOWLEDGMENTS
Electron microscopic studies were performed using the equipment of the Shared Use Center “National Center for Catalyst Research,” Boreskov Institute of Catalysis, SB RAS, Novosibirsk.
Funding
The work was supported by the Russian Science Foundation (project no.17-73-30032).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by G. Kirakosyan
Rights and permissions
About this article
Cite this article
Chesnokov, V.V., Chichkan, A.S., Svintsitskiy, D.A. et al. A Method for Synthesis of Nitrogen-Doped Graphene with High Specific Surface Area. Dokl Phys Chem 495, 159–165 (2020). https://doi.org/10.1134/S0012501620110019
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0012501620110019