Abstract
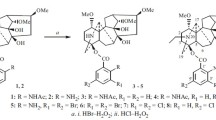
The reaction of [3 + 2] cycloaddition of a lappaconitine-based nitrone with N,N-diethylallylamine is accompanied by the formation of two minor products, unusual for the reaction of this type, containing the pyrrole cycle. The structures of the products have been established from the data of 1H and 13C NMR spectra. The mechanism of their formation is proposed.
Similar content being viewed by others
REFERENCES
Sokolov, S.F., Vestn. Aritmol., 2011, no. 64, p. 60–70.
Wang, F.-P., Chen, Q.-H., and Liu, X.-Y., Nat. Prod. Rep., 2010, vol. 27, pp. 529–570. https://doi.org/10.1039/b916679c
Imada, Y., Chem. Rev., 2019, vol. 119, no. 7, pp. 4684–4716. https://doi.org/10.1021/acs.chemrev.8b00476
Tsyrlina, E.M., Spirikhin, L.V., and Yunusov, M.S., Chem. Nat. Compd., 2016, vol. 52, no. 2, pp. 280–284. https://doi.org/10.1007/s10600-016-1614-7
Tsyrlina, E.M., Gabbasov, T.M., Spirikhin, L.V., and Yunusov, M.S., Chem. Nat. Compd., 2018, vol. 54, no. 3, pp. 515–519. https://doi.org/10.1007/s10600-018-2393-0
Gabbasov, T.M., Tsyrlina, E.M., Spirikhin, L.V., and Yunusov, M.S., Chem. Nat. Compd., 2018, vol. 54, no. 6, pp. 1205–1207. https://doi.org/10.1007/s10600-018-2598-2
Berthet, M., Cheviet, T., Dujardin, G., Parrot, I., and Martinez, J., Chem. Rev., 2016, vol. 116, pp. 15235–15283. https://doi.org/10.1021/acs.chemrev.6b00543
ACKNOWLEDGMENTS
This work was performed using equipment of the “Khimiya” Shared Facility Center, Institute of Chemistry, Ufa Federal Research Center, RAS, and the “Agidel’” Regional Shared Facility Center, Ufa Federal Research Center, RAS.
Funding
This work was performed under the State assignment no. АААА–А20–120012090026–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by I. Kudryavtsev
Rights and permissions
About this article
Cite this article
Gabbasov, T.M., Tsyrlina, E.M. & Yunusov, M.S. Unexpected Products of the Reaction of Lappaconitine-Based Nitrone with N,N-Diethylallylamine. Dokl Chem 495, 175–177 (2020). https://doi.org/10.1134/S0012500820110038
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0012500820110038