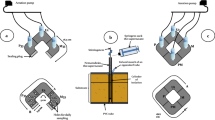
Abstract
Numerous studies have focused on the response of meiofauna after exposure to polycyclic aromatic hydrocarbons (PAHs), but none has been devoted to their uptake into nematode body compartments. The present study monitored PAH uptake by Oncholaimus campylocercoides which was selected after 40 days in the laboratory through original protocols from natural sediments collected in the Old Harbor of Bizerte, Tunisia. To achieve the mono-species level, a grain size magnification was applied by gradually adding a biosubstrate made from either the crushed shells of Mytilus galloprovincialis or minced leaves of Posidonia oceanica. After selection, O. campylocercoides was cultured and fed with earthworm powder (560 mg.l-1). Thereafter, it was exposed for 3 weeks to phenanthrene and chrysene (38, 116, and 348 ppb). Fluorescence microscopy revealed higher intensities of PAHs at the spicules, mouths, and pharynges compared with the other organs considered. Moreover, the buccal fluorescence showed a significant correlation with that measured in the biosubstrate made with shells of M. galloprovincialis.





Similar content being viewed by others
References
Alkio M, Tabuchi TM, Wang X, Cólon-Carmona A (2005) Stress responses to polycyclic aromatic hydrocarbons in Arabidopsis include growth inhibition and hypersensitive response-like symptoms. J Exp Bot 56:2983–2994
Allouche M, Nasri A, Harrath AH, Mansour L, Beyrem H, Boufahja F (2020a) Experimental selection of Marylynnia puncticaudata (Cyatholaimidae, Nematoda) and effects of organic enrichment. Environ Sci Pollut Res DOI. https://doi.org/10.1007/s11356-020-11050-1
Allouche M, Hamdi I, Nasri A, Harrath AH, Mansour L, Beyrem H, Boufahja F (2020b) Laboratory bioassay exploring the effects of anti-aging skincare products on free-living marine nematodes: case study of collagen. Environ Sci Pollut Res 27:11403–11412
Armenteros M, Pérez-García JA, Ruiz-Abierno A, Díaz-Asencio L, Helguera Y, Vincx M, Decraemer W (2010) Effects of organic enrichment on nematode assemblages in a microcosm experiment. Marine Environmental Research 70(5):374–382
Barrett J (1982) Metabolic responses to anabiosis in the fourth-stage juveniles of Ditylenchus dipsaci (Nematoda). Proc R Soc Lond B 216:159–177
Bates PA, Dokurni P, Freemont PS, Sternberg MJE (1998) Conformational analysis of the first observed non-proline cis-peptide bond occurring within the complementarity determining region (CDR) of an antibody. J Mol Biol 284:549–555
Ben Saïd O, Louati H, Soltani A, Mahmoudi E, Cravo-Laureau C, Pringault O, Duran R, Aissa P (2012) Etude in vitro de l’impact de sédiments artificiellement contaminés par l’anthracène: effets sur les bactéries indigènes et les nématodes libres marins. Can J Civ Eng 39(5):556–564
Bettaieb F, Khiari R, Dufresne A, Mhenni MF, Putaux JL, Boufi S (2015) Nanofibrillar cellulose from Posidonia oceanica: Properties and morphological features. Industrial Crops and Products 72(15):97–106
Bezerra TN, Decraemer W, Eisendle-Flöckner U, Hodda M, Holovachov O (2019) Leduc D, iljutin D, Mokievsky V, Peña Santiago R, Sharma J, Smol N, Tchesunov A, Venekey V, Zeng Z, Vanreusel A. World Database of Nematodes, Nemys available at: http://nemys.ugent.be/
Blaxter ML, De Ley P, Garey GR, Liu LX, Scheldeman P, Vierstraete A, Vanfleteren JR, Mackey LR, Dorris M, Frisse LM, Vida JT, Thomas WK (1998) A molecular evolutionary framework for the phylum Nematoda. Nature 392:71–75
Boufahja F, Hedfi A, Amorri J, Aïssa P, Beyrem H, Mahmoudi E (2011) Examination of the bioindicator potential of Oncholaimus campylocercoides (Nematoda, Oncholaimidae) from Bizerte bay (Tunisia). Ecol Indic 11:1139–1148
Boufahja F, Semprucci F (2015) Stress-induced selection of a single species from an entire meiobenthic nematode assemblage: is this possible using iron enrichment and does pre-exposure affect the ease of the process? Environ Sci Poll Res 22:1979–1998
Boufahja F, Semprucci F, Beyrem H (2016) An experimental protocol to select nematode species from an entire community using progressive sedimentary enrichment. Ecol Indic 60:292–309
Caldwell RL, Caprioli RM (2005) Tissue profiling by mass spectrometry: a review of methodology and applications. Mol Cell Proteomics. 4:394–401
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Clarke KR, Gorley RN (2001) PRIMER v5: User manual/tutorial. PRIMER-E: Plymouth, UK 91p
Clarke KR, Warwick RM (2001) Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd edn. PRIMER-E, Ltd., Plymouth Marine Laboratory, Plymouth
Cobb NA (1931) The use of live nemas (Metoncholaimus pristiurus) in zoological courses in schools and colleges. Science 74:489–490
Cox GN (1992) Molecular and biochemical aspects of nematode collagens. J Parasitol 78:1–15
De Lange HJ, Sperber V, Peeters ET (2006) Avoidance of polycyclic aromatic hydrocarbon-contaminated sediments by the freshwater invertebrates Gammarus pulex and Asellus aquaticus. Environ Toxicol Chem 25:452–457
Dutta K, Ghosh D, Nazmi A, Kumawat KL, Basu A (2010) A common carcinogen benzo[a]pyrene causes neuronal death in mouse via microglial activation. PloS ONE 5(4):e9984
Guo Y, Somerfield PJ, Warwick RM, Zhang Z (2001) Large-scale patterns in the community structure and biodiversity of free living nematodes in the Bohai Sea. China. J Mar Biol Ass UK 81:755–763
Hedfi A, Mahmoudi E, Boufahja F, Beyrem H, Aïssa P (2007) Effects of Increasing Levels of Nickel Contamination on Structure of Offshore Nematode Communities in Experimental Microcosms. Bull Environ Contam Toxicol 79:345–349
Ho NFH, Geary TG, Raub TJ, Barsuhn CL, Thompson DP (1990) Biophysical transport properties of the cuticle of Ascaris suum. Mol Biochem Parasitol 41(2):153–165
Jonker MTO (2008) Absorption of polycyclic aromatic hydrocarbons to cellulose. Chemosphere 70(5):778–782
Juario JV (1975) Nematodes species composition and seasonal fluctuation of sublittoral meiofauna community in the German Bight. Veröff Inst Meeresf Bremerhaven 15:283–337
Louati H, Ben Said O, Soltani A, Got P, Mahmoudi E, Cravo-Laureau C, Duran R, Aissa P, Pringault O (2013) The roles of biological interactions and pollutant contamination in shaping microbial benthic community structure. Chemosphere 93(10):2535–2546
Louati H, Said OB, Soltani A, Got P, Cravo-Laureau C, Duran R, Aissa P, Pringault O, Mahmoudi E (2014) Biostimulation as an attractive technique to reduce phenanthrene toxicity for meiofauna and bacteria in lagoon sediment. Environ Sci Poll Res 21:3670–3679
Mahmoudi E, Essid N, Beyrem H, Hedfi A, Boufahja F, Vtiello P, Aissa P (2005) Effects of hydrocarbon contamination on a free living marine nematode community: Results from microcosm experiments. Mar Poll Bull 50:1197–1204
Massol F, Gravel D, Mouquet N, Cadotte MW, Fukami T, Leibold MA (2011) Linking community and ecosystem dynamics through spatial ecology. Ecol Lett 14:313–323
Moens T, Bouillon S, Gallucci F (2005) Dual stable isotope abundances unravel trophic position of estuarine nematodes. J Mar Biol Assoc UK 85:1401–1407
Moens T, Vincx M (1997) Observations on the feeding ecology of estuarine nematodes. J Mar Biol Assoc UK 77:211–227
Muir L, Lee YC (1970) Glycopeptides from earthworm cuticle collagen. J Biol Chem 245(3):502–509
Pathiratne A, Hemachandra CK, Pathiratne KAS (2010) Assessment of bile fluorescence patterns in a tropical fish, Nile Tilapia (Oreochromis niloticus) exposed to naphthalene, phenanthrene, pyrene and chrysene using fixed wavelength fluorescence and synchronous fluorescence spectrometry. Bull Environ Contam Toxicol 84:554–558
Platt HM, Warwick RM (1983) Free-living marine nematodes. Part I. British Enoploids. Cambridge University, London 307 p
Platt HM, Warwick RM (1988) Free-living marine nematodes. Part II. British Chromadorids. Synopsis of the British fauna (New Series). No. 38, E.J. Brill/W.Backhuys, Leiden
Pearson TH, Rosenberg R (1978) Macrobenthic Succession in Relation to Organic Enrichment and Pollution of the Marine Environment. Oceanogr Mar Biol 16:229–311
Pellerone FI, Archer SK, Behm CA, Grant WN, Lacey MJ, Somerville AC (2003) Trehalose metabolism genes in Caenorhabditis elegans and filarial nematodes. Int J Parasitol 11:1195–1206
Riemann F, Schrage M (1978) The mucus-trap hypothesis on feeding of aquatic nematodes and implications for biodegradation and sediment texture. Oecologia 34:75–88
Reyzer ML, Caprioli RM (2007) MALDI-MS-based imaging of small molecules and proteins in tissues. Curr Opin Chem Biol 11:29–35
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Schratzberger M, Rees HL, Boyd SE (2000a) Effects of simulated deposition of dredged material on structure of nematode assemblages - the role of burial. Mar Biol 136:519–530
Schratzberger M, Rees HL, Boyd SE (2000b) Effects of simulated deposition of dredged material on structure of nematode assemblages - the role of contamination. Mar Biol 137:613–622
Schratzberger M, Whomersley P, Warr K, Bolam SG, Rees HL (2004) Colonisation of various types of sediment by estuarine nematodes via lateral infaunal migration: a laboratory study. Mar Biol 145:69–78
Seinhorst JW (1959) A rapid method for the transfer of nematodes from fixative to anhydrous glycerine. Nematologica 4:67–69
Sun M, Liu K, Zhao Y, Tian D, Ye M, Liu M, Jiao J, Jiang X (2017) Effects of Bacterial-Feeding Nematode Grazing and Tea Saponin Addition on the Enhanced Bioremediation of Pyrene-Contaminated Soil Using Polycyclic Aromatic Hydrocarbon-Degrading Bacterial Strain. Pedosphere 27(6):1062–1072
Takahashi K, Kozuka T, Anegawa A, Nagatani A, Mimura T (2015) Development and Application of a High-Resolution Imaging Mass Spectrometer for the Study of Plant Tissues. Plant Cell Physiol 56:1329–1338
Ukalska-Jaruga A, Smreczak B (2020) The Impact of Organic Matter on Polycyclic Aromatic Hydrocarbon (PAH) Availability and Persistence in Soils. Molecules 25(11):2470
Vitiello P, Dinet A (1979) Définition et échantillonnage du méiobenthos. Rapp Comm Int Mer Médit 25:279–283
Wang P, Zhu Y, Lu H, Zhang Y (2010) Visualizing localizations and movement of anthracene in Kandelia candel (L.) Druce leaves by fluorescence microscopy. J Coast Res 26:549–554
Warwick RM, Platt HM, Somerfield PJ (1998) Free-living marine nematodes. Part III. British monohysterids. Synopsis of British fauna (new series) No. 53, Field Studies Council, Shrewsbury.ne nematodes
Warwick RM, Price R (1979) Ecological and metabolic studies on free-living nematodes from an estuarine mud-flat. Estuarine and Coastal Marine Science 9(3):257–271
Wieser W (1960) Benthic studies in Buzzards Bay. II. The meiofauna. Limnol Oceanogr 5:121–137
Wild E, Dent J, Barber JL, Thomas GO, Jones KC (2004) A novel analytical approach for visualizing and tracking organic chemicals in plants. Environ Sci Technol 38:4195–4199
Ye H, Gemperline E, Venkateshwaran M, Chen R, Delaux PM, Howes-Podoll M, Ané JM, Li L (2013) MALDI mass spectrometry-assisted molecular imaging of metabolites during nitrogen fixation in the Medicago truncatula-Sinorhizobium meliloti symbiosis. Plant J 75:130–145
Yin J, Ren W, Yang G, Duan J, Huang X, Fang R, Li C, Li T, Yin Y, Hou Y, Kim SW, Wu G (2016) L-Cysteine metabolism and its nutritional implications. Mol Nutr Food Res 60:134–146
Zhenjun S, Xianchun L, Lihui S, Chunyang S (1997) Earthworm as a potential protein resource. Ecol Food Nutr 36:221–236
Zhou H (2001) Effects of leaf litter addition on meiofaunal colonization of azoic sediments in a subtropical mangrove in Hong Kong. J Exp Mar Biol Ecol 256:99–121
Zrafi I, Safi N, Safi B, Mzoughi N, Aissi A, Ben Abdennebi H, Saidane D (2010) Distribution and Sources of Polycyclic Aromatic Hydrocarbons Around a Petroleum Refinery Rejection Area in Jarzouna-Bizerte (Coastal Tunisia). Soil Sediment Contam. 19:292–306
Acknowledgments
The authors extendtheir appreciation to Researchers Supporting Project number RSP-2021/17, King Saud University, Riyadh, Saudi Arabia. The authors also thank Dr. Mateusz Płóciennik (University of Łódz, Poland) for the valuable assistance during the fluorescence examinations.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
The authors extendtheir appreciation to Researchers Supporting Project number RSP-2021/17, King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Mohamed Allouche: formal analysis, validation, writing (original draft), writing (review and editing). Ahmed Nasri: data curation, investigation. Abdel Halim Harrath: funding acquisition, writing (review and editing). Lamjed Mansour: funding acquisition, writing (review and editing). Saleh Alwasel: funding acquisition, investigation. Hamouda Beyrem: data curation, investigation. Gabriel Plăvan: investigation. Melissa Rohal-Lupher: writing (review and editing). Fehmi Boufahja: conceptualization, formal analysis, funding acquisition, investigation, methodology, supervision, validation, writing (review and editing).
Corresponding author
Ethics declarations
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Bruno Nunes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
(This manuscript represents the views of the authors, and not those of the Texas Water Development Board)
Rights and permissions
About this article
Cite this article
Allouche, M., Nasri, A., Harrath, A.H. et al. Meiobenthic nematode Oncholaimus campylocercoides as a model in laboratory studies: selection, culture, and fluorescence microscopy after exposure to phenanthrene and chrysene. Environ Sci Pollut Res 28, 29484–29497 (2021). https://doi.org/10.1007/s11356-021-12688-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12688-1