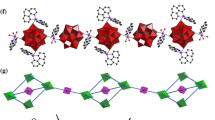
Abstract
Three Keggin polyoxotungstate inorganic–organic hybrid compounds have been synthesized under hydrothermal conditions by using H4SiW12O40 as starting materials: [{CuII(2,2′-bipy)2}2SiW12O40]·2H2O (1), [{CuII(phen)2}2SiW12O40] (2) and [{CuI(2,2′-bipy)2}4SiW12O40] (3) (2,2′-bipy = 2,2′-bipyridine, phen = 1,10′-phenanthroline) and structurally characterized by elemental analysis, IR spectra, TG analysis and X-ray single-crystal diffraction. Three compounds are all based on [SiW12O40]4− α-Keggin type building block and synthesized under the same reaction condition, only different compound 1 and 2 ligand, 1 and 3 pH value. Compound 1 displays a novel example of two-dimensional framework with tetra-supporting α-Keggin polyanions which are linked by four same fragments and it becomes 3D structure due to the existence of hydrogen bonds. Compound 2 shows that the bi-supporting polyoxotungstate molecule consists of a α-Keggin polyoxoanion [SiW12O40]4− and a pair of transition metal complex fragments which are covalently linked to opposite sides of the surface terminal oxygen atoms of WO6 octahedra. Compound 3 consists of α-[SiW12O40]4− polyoxoanions, counter cations [Cu(2,2′-bipy)2]+. The crystal packing is stabilized by H-bonds and weak π–π interactions, leads to a 3D supramolecular architecture. The results show that the pH value of the reaction system plays a crucial role in structural control of self-assembled processes. The thermal stabilities of the compounds are discussed.
Graphical Abstract
Three Keggin polyoxotungstate inorganic–organic hybrid compounds have been synthesized under hydrothermal conditions by using H4SiW12O40 as starting materials: [{CuII(2,2′-bipy)2}2SiW12O40]·2H2O (1), [{CuII(phen)2}2SiW12O40] (2) and [{CuI(2,2′-bipy)2}4SiW12O40] (3) (2,2′-bipy = 2,2′-bipyridine, phen = 1,10′-phenanthroline) and structurally characterized by elemental analysis, IR spectra, TG analysis and X-ray single-crystal diffraction. Three compounds are all based on [SiW12O40]4− α-Keggin type building block and synthesized under the same reaction condition, only different compound 1 and 2 ligand, 1 and 3 pH value.




Similar content being viewed by others
References
M. Arefian, M. Mirzaei, H. Eshtiagh-Hosseini, and A. Frontera (2017). Dalton Trans. 46, 6812–6829.
A. Misra, I. F. Castillo, D. P. Müller, C. González, S. Eyssautier-Chuine, A. Ziegler, J. M. de la Fuente, S. G. Mitchell, and C. Streb (2018). Angew. Chem. Int. Ed. 57, 14926–14931.
A. Bijelic, M. Aureliano, and A. Rompel (2019). Angew. Chem. Int. Ed. 58, 2980–2999.
M. Zhao, Y. Fang, L. H. Ma, X. Y. Zhu, L. Jiang, M. X. Li, and Q. X. Han (2020). J. Inorg. Biochem. 210, 111131. https://doi.org/10.1016/j.jinorgbio.2020.111131.
Y. J. Gong, Y. X. Guo, Q. Z. Hu, C. Wang, L. Zang, and L. Yu (2017). ACS Sustainable Chem. Eng. 5, 3650–3658.
Y. Y. Hu, T. T. Zhang, X. Zhang, D. C. Zhao, X. B. Cui, Q. S. Huo, and J. Q. Xu (2016). Dalton Trans. 45, 2562–2573.
H. R. Ghalebi, S. Aber, and A. Karimi (2016). J. Mol. Catal. A Chem. 415, 96–103.
W. L. Zhou, Y. P. Zheng, and J. Peng (2018). J. Solid. State. Chem. 258, 786–791.
Y. Ammari, E. Dhahri, M. Rzaigui, E. K. Hlil, and S. Abid (2016). J. Clust. Sci. 27, 1213–1227.
H. Zhang, X. K. Lin, Y. Yan, and L. X. Wu (2006). Chem. Comm. 44, 4575–4577.
Y. Zhang, X. Bo, A. Nsabimana, A. Munyentwali, M. Li, L. Guo, and C. Han (2015). Biosens. Bioelectron. 66, 191–197.
P. He, W. L. Chen, J. P. Li, H. Zhang, Y. W. Li, and E. B. Wang (2020). Sci. Bull. 65, 35–44.
J. F. Keggin (1933). Nature 131, 908–909.
F. X. Ma, Y. G. Chen, D. M. Shi, F. X. Meng, and H. J. Pang (2008). Trans. Met. Chem. 33, 697–703.
F. X. Ma, Y. G. Chen, H. Y. Yang, X. W. Dong, H. Jiang, and F. Wang (2018). J. Clust. Sci. 30, 123–129.
Z. Z. He, D. Ma, B. R. Cao, X. Q. Li, and Y. Lu (2018). Inorg. Chim. Acta. 471, 316–325.
Dutta, M. Dolai, S. Biswas, A. Dutta, and M. Ali (2020). Polyhedron 176, 114204. https://doi.org/10.1016/j.poly.2019.114204.
Y. Hou, H. J. Pang, L. Zhang, B. N. Li, J. J. Xin, K. Q. Li, H. Y. Ma, X. M. Wang, and L. C. Tan (2020). J. Power Sources 446, 227319. https://doi.org/10.1016/j.jpowsour.2019.227319.
C. L. Pan, J. Q. Xu, Y. Sun, D. Q. Chu, L. Ye, Z. L. Lü, and T. G. Wang (2003). Inorg. Chem. Commun. 6, 233–237.
G. N. Wang, T. T. Chen, C. J. Gómez-García, F. Zhang, M. Y. Zhang, H. Y. Ma, H. J. Pang, X. M. Wang, and L. C. Tan (2020). Small. https://doi.org/10.1002/smll.202001626.
S. Lu, F. X. Meng, D. M. Shi, and Y. G. Chen (2008). Trans. Met. Chem. 33, 353–359.
K. Liu, F. X. Meng, F. X. Ma, and Y. G. Chen (2007). Chem. Res. Chin. Univ. 23, 391–394.
A. X. Tian, J. Ying, J. Peng, J. Q. Sha, H. J. Pang, P. P. Zhang, Y. Chen, M. Zhu, and Z. M. Su (2009). Inorg. Chem. 48, 100–110.
K. Liu, F. X. Meng, and Y. G. Chen (2007). Trans. Met. Chem. 32, 350–354.
C. M. Liu, D. Q. Zhang, M. Xiong, and D. B. Zhu (2002). Chem. Commun. 1416–1417.
Z. G. Han, Y. L. Zhao, J. Peng, H. Y. Ma, Q. Liu, E. B. Wang, N. H. Hu, and H. Q. Jia (2005). Eur. J. Inorg. Chem. 2, 264–271.
C. M. Liu, D. Q. Zhang, C. Y. Xu, and D. B. Zhu (2004). Solid State Sci. 6, 689–696.
L. M. Rodrigguez-Albelo, A. R. Ruiz-Salvador, A. Sampieri, D. W. Lewis, A. Gómez, B. Nohra, P. Mialane, J. Marrot, F. Sécheresse, C. Mellot-Draznieks, R. N. Biboum, B. Keita, L. Nadjo, and A. Dolbecq (2009). J. Am. Chem. Soc. 131, 16078–16087.
M. Mirzaei, H. Eshtiagh-Hosseini, and A. Hassanpoor (2019). Inorg. Chim. Acta. 484, 332–337.
Rigaku, PROCESS-AUTO (Rigaku Corporation, Tokyo, Japan, 1998).
L. J. Farrugia (2012). J. Appl. Crystallogr. 45, 849–854.
G. M. Sheldrick (2015). Acta. Crystallogr. A71, 3–8.
G. M. Sheldrick (2015). Acta. Crystallogr. C71, 3–8.
S. H. Feng and R. R. Xu (2001). Acc. Chem. Res. 34, 239–247.
Z. Y. Shi, X. J. Gu, J. Peng, and Y. H. Chen (2005). J. Solid. State Chem. 178, 1988–1995.
L. F. Chen, M. J. Turo, M. Gembicky, R. A. Reinicke, and A. M. Schimpf (2020). Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.202005627.
X. Y. Zhang, R. Xi, S. L. Yin, X. R. Cao, Y. L. Zhang, L. Lin, R. Chen, and H. Wu (2018). J. Solid State. Chem. 258, 737–743.
Acknowledgements
This work was supported by the Key Construction Disciplines of Biology, Jilin Agricultural Science and Technology University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, FX., Chen, YG., Jiang, JK. et al. Both Rigid Organic Ligands and pH-Controlled Three Keggin-Type Polyoxotungstates Derivates: Synthesis, Crystal Structure. J Clust Sci 33, 707–715 (2022). https://doi.org/10.1007/s10876-021-02001-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02001-y