Abstract
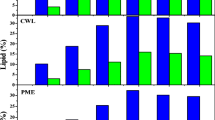
Lignocellulosic wastes are abundant and considered as the important feedstock. It is a value-added product by using microorganisms. Yet little is known regarding usage of lignocellulosic biomass by anoxygenic photosynthetic bacteria although they provide various applications and their biomass are rich in essential nutrients. This limits applications between lignocellulosic wastes and phototrophic bacteria. We examined growth and valuable products of photosynthetic bacterium Rhodopseudomonas faecalis PA2 cultivated in different lignocellulosic waste suspension under light condition. Suspension of rice straw, bagasse, coconut meal, soybean meal, corncob, fiber of palmyra palm peel, and spent coffee ground were prepared by filtration or boiling to be used as sole substrates without additional nutrients. Here we show that the selected strain could grow in the lignocellulosic waste suspension without heat pretreatment. Soybean meal showed the highest biomass, carbohydrate, lipid, carotenoid and bacteriochlorophyll productivities of 0.71 g/L/day, 350, 9.61, 33.11 and 212.54 mg/L/day, respectively, whereas coconut meal was the best substrate for protein productivity (138.61 mg/L/day). Rice straw and fiber of palmyra palm peel seem to be interesting and utilization of fiber of palmyra palm peel in microbial cultivation is reported for the first time. Our results demonstrate that lignocellulosic material suspension can be used as the promising substrates to produce valuable biomass from anoxygenic photosynthetic bacteria without pretreatment and secondary solid waste. We anticipate our investigation to be a starting point for further simultaneous value-added product production by anoxygenic photosynthetic bacteria as well as lignocellulosic waste utilization.
Graphic Abstract


Similar content being viewed by others
References
Lokhande, A., Ingale, S.L., Lee, S.H., Kim, J.S., Lohakare, J.D., Chae, B.J., Kwon, I.K.: The effects of Rhodobacter capsulatus KCTC-2583 on cholesterol metabolism, egg production and quality parameters during the late laying periods in hens. Asian-Australas J Anim Sci. 26, 831–837 (2013)
Liu, J., Xia, B., Du, X., Zeng, T., Liu, Y., Chen, L., Lu, L., Li, C.: Effects of water supplemented with Bacillus subtilis and photosynthetic bacteria on egg production, egg quality, serum immunoglobulins and digestive enzyme activity of ducks. J. Appl. Anim. Res. 46, 322–326 (2018)
Delamare-Deboutteville, J., Batstone, D.J., Kawasaki, M., Stegman, S., Salini, M., Tabrett, S., Smullen, R., Barnes, A.C., Hulsen, T.: Mixed culture purple phototrophic bacteria is an effective fishmeal replacement in aquaculture. Water Res. X. 4, 100031 (2019)
Loo, P.L., Vikineswary, S., Chong, V.C.: Nutritional value and production of three species of purple non-sulfur bacteria grown in palm oil mill effluent and their application in rotifer culture. Aquac. Nutr. 19, 895–907 (2013)
Chiu, K., Liu, W.: Dietary administration of the extract of Rhodobacter sphaeroides WL-APD911 enhances the growth performance and innate immune responses of seawater red tilapia (Oreochromis mossambicus × Oreochromis niloticus). Aquaculture. 418–419, 32–38 (2014)
Saejung, C., Chaiyarat, A., Sa-noamuang, L.: Effects of algae, yeast and photosynthetic bacteria diets on survival and growth performance in the fairy shrimp, Streptocephalus sirindhornae (Branchiopoda, Anostraca). Crustaceana. 91, 1505–1522 (2018)
Nunkaew, T., Kantachote, D., Nitoda, T., Kanzaki, H.: The use of rice straw broth as an appropriate medium to isolate purple nonsulfur bacteria from paddy fields. Electron. J. Biotechnol. 15 (2012). https://doi.org/10.2225/vol15-issue6-fulltext-8
Patthawaro, S., Lomthaisong, K., Saejung, C.: Bioconversion of agro-industrial waste to value-added product lycopene by photosynthetic bacterium Rhodopseudomonas faecalis and its carotenoid composition. Waste Biomass Valor. (2019). https://doi.org/10.1007/s12649-018-00571-z
Patthawaro, S., Saejung, C.: Production of single cell protein from manure as animal feed by using photosynthetic bacteria. Microbiol. Open. (2019). https://doi.org/10.1002/mbo3.913
Saejung, C., Ampornpat, W.: Production and nutritional performance of carotenoid-producing photosynthetic bacterium Rhodopseudomonas faecalis PA2 grown in domestic wastewater intended for animal feed production. Waste Biomass Valor. 10, 299–310 (2019)
Saejung, C., Puensungnern, L.: Evaluation of molasses-based medium as a low cost medium for carotenoids and fatty acid production by photosynthetic bacteria. Waste Biomass Valor. 11, 143–152 (2020)
Saejung, C., Salasook, P.: Recycling of sugar industry wastewater for single-cell protein production with supplemental carotenoids. Environ. Technol. 41, 59–70 (2020)
Saratale, G.D., Kshirsagar, S.D., Saratale, R.G., Govindwar, S.P., Oh, M.K.: Fermentative hydrogen production using sorghum husk as a biomass feedstock and process optimization. Biotechnol. Bioprocess Eng. 20, 733–743 (2015)
Ravindran, R., Jaiswal, A.K.: Microbial enzyme production using lignocellulosic food industry wastes as feedstock: a review. Bioengineering (Basel). 3(30) (2016)
Alessi, A.M., Bird, S.M., Oates, N.C., Li, Y., Dowle, E.H., de Azevedo, E.R., Bennett, J.P., Polikarpov, I., Young, J.P.W., McQueen-Mason, S.J., Bruce, N.C.: Defining functional diversity for lignocellulose degradation in a microbial community using multi-omics studies. Biotechnol. Biofuels. 11, 166 (2018)
Santek, M.I., Beluhan, S., Santek, B.: Production of microbial lipids from lignocellulosic biomass. In: Nageswara-Rao, M., Soneji, J.R. (eds.) Advances in Biofuels and Bioenergy. IntechOpen, London (2018). https://doi.org/10.5772/intechopen.74013
Kanokkanjana, K., Garivait, V.: Alternative rice straw management practices to reduce field open burning in Thailand. Int. J. Environ. Sci. Dev. 4, 119–123 (2013)
Saejung, C., Apaiwong, P.: Enhancement of carotenoid production in the new carotenoid-producing photosynthetic bacterium Rhodopseudomonas faecalis PA2. Biotechnol. Bioprocess Eng. 20, 701–707 (2015)
Sheng, G.P., Yu, H.Q., Yu, Z.: Extraction of extracellular polymeric substances from the photosynthetic bacterium Rhodopseudomonas acidophila. Appl. Microbiol. Biotechnol. 67, 125–130 (2005)
DuBois, M., Gilles, K.A., Hamilton, J.K., Rebers, P.A., Smith, F.: Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356 (1956)
Kwon, D.Y., Rhee, J.S.: A simple and rapid colorimetric method for determination of free fatty acids for lipase assay. J. Am. Oil Chem. Soc. 63, 89–92 (1986)
Shen, D., Xiao, R., Gu, S., Zhang, H.: The overview of thermal decomposition of cellulose in lignocellulosic biomass, cellulose. In: van de Ven, T., Kadla, J. (eds.) Biomass Conversion. IntechOpen, London (2013). https://doi.org/10.5772/51883
Li, S., Zhu, D., Li, K., Yang, Y., Lei, Z., Zhang, Z.: Soybean curd residue: composition, utilization, and related limiting factors. ISRN Ind. Eng. (2013). https://doi.org/10.1155/2013/423590
Jia, X., Chen, M., Wan, J., Su, H., He, C.: Review on the extraction, characterization and application of soybean polysaccharide. RSC Adv. 5, 73525–73534 (2015)
Getha, K., Vikineswary, S., Chong, V.C.: Isolation and growth of the phototrophic bacterium Rhodopseudomonas palustris strain B1 in sago-starch-processing wastewater. World J. Microbiol. Biotechnol. 14, 505–511 (1998)
Ashokkumar, V., Salam, Z., Tiwari, O.N., Chinnasamy, S., Mohammed, S., Ani, F.N.: An integrated approach for biodiesel and bioethanol production from Scenedesmus bijugatus cultivated in a vertical tubular photobioreactor. Energy Convers. Manag. 101, 778–786 (2015)
Tourang, M., Baghdadi, M., Torang, A., Sarkhosh, S.: Optimization of carbohydrate productivity of Spirulina microalgae as a potential feedstock for bioethanol production. Int. J. Environ. Sci. Technol. 16, 1303–1318 (2019)
Rosa, G.M., Moraes, L., Souza, M.R.A.Z., Costa, J.A.V.: Spirulina cultivation with a CO2 absorbent: influence on growth parameters and macromolecule production. Bioresour. Technol. 200, 528–534 (2016)
Alhattab, M., Kermanshahi-Pour, A., Brooks, M.S.L.: Microalgae disruption techniques for product recovery: influence of cell wall composition. J. Appl. Phycol. 31, 61–88 (2019)
Silva, C.E.F., Sforza, E.: Carbohydrate productivity in continuous reactor under nitrogen limitation: effect of light and residence time on nutrient uptake in Chlorella vulgaris. Process Biochem. 51, 2112–2118 (2016)
Ho, S.H., Kondo, A., Hasunuma, T., Chang, J.S.: Engineering strategies for improving the CO2 fixation and carbohydrate productivity of Scenedesmus obliquus CNW-N used for bioethanol fermentation. Bioresour. Technol. 143, 163–171 (2013)
Cheng, D., Li, D., Yuan, Y., Zhou, L., Li, X., Wu, T., Wang, L., Zhao, Q., Wei, W., Sun, Y.: Improving carbohydrate and starch accumulation in Chlorella sp. AE10 by a novel two-stage process with cell dilution. Biotechnol. Biofuels. 10, 75 (2017)
Saejung, C., Thammaratana, T.: Biomass recovery during municipal wastewater treatment using photosynthetic bacteria and prospect of production of single cell protein for feedstuff. Environ. Technol. 37, 3055–3061 (2016)
Ng, S.P., Tan, C.P., Lai, O.M., Long, K., Mirhosseini, H.: Extraction and characterization of dietary fiber from coconut residue. J. Food Agric. Environ. 8, 172–177 (2010)
Oladosu, Y., Rafii, M.Y., Abdullah, N., Magaji, U., Hussin, G., Ramli, A., Miah, G.: Fermentation quality and additives: a case of rice straw silage. Biomed. Res. Int. (2016). https://doi.org/10.1155/2016/7985167
Mukherjee, R., Chakraborty, R., Dutta, A.: Role of fermentation in improving nutritional quality of soybean meal—a review. Asian Australas. J. Anim. Sci. 29, 1523–1529 (2016)
Higuchi-Takeuchi, M., Morisaki, K., Toyooka, K., Numata, K.: Synthesis of high-molecular-weight polyhydroxyalkanoates by marine photosynthetic purple bacteria. PLoS One. (2016). https://doi.org/10.1371/journal.pone.0160981
Padovani, G., Carlozzia, P., Seggianib, M., Cinellib, P., Vitolob, S., Lazzeri, A.: PHB-rich biomass and bioH2 production by means of photosynthetic microorganisms. Chem. Eng. Trans. 49, 55–60 (2016)
Pierce, E.C., LaFayette, P.R., Ortega, M.A., Joyce, B.L., Kopsell, D.A., Parrott, W.A.: Ketocarotenoid production in soybean seeds through metabolic engineering. PLoS One. 10, e0138196 (2015)
Vincent, U., Serano, F., Von Holst, C.: Development and validation of a multi-analyte method for the regulatory control of carotenoids used as feed additives in fish and poultry feed. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 34, 1285–1297 (2017)
Pasarin, D., Rovinaru, C.: Sources of carotenoids and their uses as animal feed additives-a review. Sci. Papers Ser. D, Anim. Sci. 61, 68–73 (2018)
Lucas, A., Morales, J., Velando, A.: Differential effects of specific carotenoids on oxidative damage and immune response of gull chicks. J. Exp. Biol. 217, 1253–1262 (2014)
Zaheer, K.: Hen egg carotenoids (lutein and zeaxanthin) and nutritional impacts on human health: a review. CYTA-J. Food. 15, 474–487 (2017)
Swamy, B., Samia, M., Boncodin, R., Marundan, S., Rebong, D.B., Ordonio, R.L., Miranda, R.T., Rebong, A., Alibuyog, A.Y., Adeva, C.C., Reinke, R., MacKenzie, D.J.: Compositional analysis of genetically engineered GR2E “Golden Rice” in comparison to that of conventional rice. J. Agric. Food Chem. 67, 7986–7994 (2019)
Oren, A.: Characterization of pigments of prokaryotes and their use in taxonomy and classification. In: Rainey, F., Oren, A. (eds.) Methods in Microbiology, vol. 38, pp. 261–282. Elsevier, Amsterdam (2011)
Paengkoum, P.: Utilization of concentrate supplements containing varying levels of coconut meal by Thai native Anglo-Nubian goats. Livestock Res. Rural Devel. 23, 1–7 (2011)
Abeysekara, T.S., Atapattu, N.S.B.M.: Effects of dietary coconut oil meal with or without an enzyme mixture on laying performance and physical parameters of eggs of Japanese quail (Coturnix coturnix). Trop. Agric. Res. 7, 414–419 (2016)
Thiruchitrambalam, M., Logesh, M., Shanmugam, D., Muthukumar, S.: The physical, chemical properties of untreated and chemically treated Palmyra palm leaf fibres. Int. J. Eng. Technol. 7, 582–585 (2018)
Saha, B.C., Qureshi, N., Kennedy, G.J., Cotta, M.A.: Biological pretreatment of corn Stover with white-rot fungus for improved enzymatic hydrolysis. Int. Biodeterior. Biodegradation. 109, 29–35 (2016)
Kucharska, K., Rybarczyk, P., Holowacz, I., Lukajtis, R., Glinka, M., Kaminski, M.: Pretreatment of lignocellulosic materials as substrates for fermentation processes. Molecules. 23, 2937 (2018)
Amin, F.R., Khalid, H., Zhang, H., Rahman, S., Zhang, R., Liu, G., Chen, C.: Pretreatment methods of lignocellulosic biomass for anaerobic digestion. AMB Express. 7, 72 (2017)
Acknowledgements
This work was supported by Thailand Science Research and Innovation (TSRI) and Office of the Higher Education Commission (Grant No. MRG6280016) and Faculty of Science, Khon Kaen University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saejung, C., Sanusan, W. Valorization of Lignocellulosic Wastes and Nutrient Recovery by Anoxygenic Photosynthetic Bacteria. Waste Biomass Valor 12, 4835–4844 (2021). https://doi.org/10.1007/s12649-021-01351-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-021-01351-y