Abstract
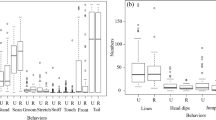
The Trivers–Willard hypothesis (TWH) posits that maternal care will be biased in favor of the sex that provides the greatest fitness returns per unit of investment, depending on maternal physical condition. Our aim was to examine the TWH in mantled howler monkeys living at Los Tuxtlas (Veracruz, Mexico). The biological attributes of mantled howler monkeys (Alouatta palliata) meet the assumptions of TWH better than those of other explanations, so we expected that females in better physical condition should bias maternal care toward sons, whereas mothers in worse physical condition should bias care toward daughters. Between December 2017 and March 2019, we studied mother–infant interactions in 20 dyads with focal-animal sampling and continuous recording (N = 204 h). We performed genetic analysis to determine offspring sex (N = 7 daughters and 13 sons) and measured C-peptide in urine samples of mothers to assess their physical condition (N = 46 samples). Mothers in better physical condition spent less time in contact with their sons but more time in contact with their daughters. For proximity behavior, mothers in better physical condition spent more time near their sons and less time near their daughters. These results suggest a bias in maternal care towards daughters, contrary to our predictions. In light of current models of maternal investment, our results support that mothers obtain higher fitness returns through daughters.



Similar content being viewed by others
References
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49:227–266. https://doi.org/10.1163/156853974X00534
Altmann J (1980) Baboon mothers and infants. University of Chicago Press, Chicago
Altmann M, Altmann J (1991) Models of status-correlated bias in offspring sex ratio. Am Nat 137:542–555. https://doi.org/10.1086/285182
Arroyo-Rodríguez V, Serio-Silva J, carlos, Álamo-García J, Ordano M (2007) Exploring immature-to-mother social distances in Mexican mantled howler monkeys at Los Tuxtlas, Mexico. Am J Primatol 69:173–181. https://doi.org/10.1002/ajp.20344
Arroyo-Rodríguez V, Dias P, Cristóbal-Azkarate J (2011) Group size and foraging effort in mantled howlers at Los Tuxtlas, Mexico: a preliminary test of the ecological-constraints model. In: Gama-Campillo L, Pozo-Montuy G, Contreras-Sánchez W, Arriaga-Weiss S (eds) Perspectivas en primatología mexicana. Universidad Juárez Autónoma de Tabasco, Villahermosa, pp 103–116
Balcells CD, Veà Baró JJ (2009) Developmental stages in the howler monkey, subspecies Alouatta palliata mexicana: a new classification using age-sex categories. Neotrop Primates 16:1–8. https://doi.org/10.1896/044.016.0101
Baldwin JD, Baldwin JI (1973) Interactions between adult female and infant howling monkeys (Alouatta palliata). Folia Primatol 20:27–71. https://doi.org/10.1159/000155566
Barrett L, Dunbar RIM, Dunbar P (1995) Mother-infant contact as contingent behaviour in gelada baboons. Anim Behav 49(3):805–810
Bentley-Condit VK (2003) Sex differences in captive olive baboon behavior during the first 14 days of life. Int J Primatol 24:1093–1112. https://doi.org/10.1023/A:1026232413614
Bereczkei T, Dunbar RIM (1997) Female-biased reproductive strategies in a Hungarian Gypsy population. Proc R Soc Lond B Biol Sci 264:17–22. https://doi.org/10.1098/rspb.1997.0003
Borgstede M (2019) Is there a Trivers-Willard effect for parental investment? Modelling evolutionarily stable strategies using a matrix population model with nonlinear mating. Theor Popul Biol 130:74–82. https://doi.org/10.1016/j.tpb.2019.10.001
Brown GR, Dixson AF (2000) The development of behavioural sex differences in infant rhesus macaques ( Macaca mulatta). Primates 41:63–77
Brown GR (2001) Sex-biased investment in nonhuman primates: can Trivers and Willard’s theory be tested? Anim Behav 61:683–694. https://doi.org/10.1006/anbe.2000.1659
Cameron EZ (2004) Facultative adjustment of mammalian sex ratios in support of the Trivers-Willard hypothesis: evidence for a mechanism. Proc R Soc Lond B Biol Sci 271:1723–1728. https://doi.org/10.1098/rspb.2004.2773
Cameron EZ, Linklater WL (2000) Individual mares bias investment in sons and daughters in relation to their condition. Anim Behav 60:359–367. https://doi.org/10.1006/anbe.2000.1480
Cameron EZ, Linklater WL (2002) Sex bias in studies of sex bias: the value of daughters to mothers in poor condition. Anim Behav 63:F5–F8. https://doi.org/10.1006/anbe.2001.1902
Cano-Huertes B, Rangel-Negrín A, Coyohua-Fuentes A et al (2017) Reproductive energetics of female mantled howlers (Alouatta palliata). Int J Primatol 38:942–961. https://doi.org/10.1007/s10764-017-9990-9
Castillo-Campos G, Laborde D (2004) La vegetación. In: Guevera S, Laborde D, Sánchez-Ríos G (eds) Los Tuxtlas. El paisaje de la Sierra, Instituto de Ecología, A.C., Xalapa, pp 231–265
Charnov EL (1982) The theory of sex allocation. Princeton University Press, Princeton, NJ
Chism J (1986) Development and mother-infant relations among captive patas monkeys. Int J Primatol 7:49–81
Clancey E, Byers JA (2014) The definition and measurement of individual condition in evolutionary studies. Ethology 120:845–854. https://doi.org/10.1111/eth.12272
Clark AB (1978) Sex ratio and local resource competition in a prosimian primate. Science 201:163–165. https://doi.org/10.1126/science.201.4351.163
Clarke MR, Glander KE (1984) Female reproductive success in a group of free-ranging howling monkeys (Alouatta palliata) in Costa Rica. In: Small MF (ed) Female primates: studies by women primatologists. Liss, New York, A.R, pp 111–126
Clarke MR, Zucker EL, Ford RT, Harrison RM (2007) Behavior and endocrine concentrations do not distinguish sex in monomorphic juvenile howlers (Alouatta palliata). Am J Primatol 69:477–484. https://doi.org/10.1002/ajp.20354
Clutton-Brock TH (1991) The evolution of parental care. Princeton university press, Princeton NJ
Clutton-Brock TH, Harvey PH, Rudder B (1977) Sexual dimorphism, socionomic sex ratio and body weight in primates. Nature 269:797–800. https://doi.org/10.1038/269797a0
Clutton-Brock TH, Albon SD, Gibson RM, Guinness FE (1979) The logical stag: adaptive aspects of fighting in red deer (Cervus elaphus L.). Anim Behav 27:211–225. https://doi.org/10.1016/0003-3472(79)90141-6
Connour JR, Glander KE (2020) Sexual dimorphism and growth in Alouatta palliata based on 20+ years of field data. Am J Phys Anthropol. https://doi.org/10.1002/ajpa.24055 (n/a)
Cristóbal-Azkarate JC, Dunn JC, Balcells CD, Baró JV (2017) A demographic history of a population of howler monkeys (Alouatta palliata) living in a fragmented landscape in Mexico. PeerJ 5:e3547. https://doi.org/10.7717/peerj.3547
Cronk L (1989) Low socioeconomic status and female-biased parental investment: the mukogodo example. Am Anthropol 91:414–429. https://doi.org/10.1525/aa.1989.91.2.02a00090
Cronk L (2007) Boy or girl: gender preferences from a Darwinian point of view. Reprod Biomed Online 15:23–32. https://doi.org/10.1016/S1472-6483(10)60546-9
De Jong T, Klinkhamer P (2005) Evolutionary ecology of plant reproductive strategies. Cambridge university press, Cambridge
Di Fiore A (2005) A rapid genetic method for sex assignment in non-human primates. Conserv Genet 6:1053–1058. https://doi.org/10.1007/s10592-005-9086-5
Dias PAD, Rangel-Negrin A, Veà JJ, Canales-Espinosa D (2010) Coalitions and male–male behavior in Alouatta palliata. Primates 51:91. https://doi.org/10.1007/s10329-009-0170-1
Dias PAD, Coyohua-Fuentes A, Canales-Espinosa D, Rangel-Negrín A (2016) Factors influencing the reproductive success of female black howlers (Alouatta pigra). Int J Primatol 6:638–655. https://doi.org/10.1007/s10764-016-9928-7
Dias P, Cano-Huertes B, Coyohua-Fuentes A et al (2018) Maternal condition and maternal investment during lactation in mantled howler monkeys. Am J Phys Anthropol 167:178–184. https://doi.org/10.1002/ajpa.23626
Dias PAD, Domínguez ILM, Negrín AR (2020) Factors influencing infant sex ratio in howler monkeys (Alouatta spp.): a literature review and analysis. Am J Phy Anthropol 172:48–57. https://doi.org/10.1002/ajpa.24035
Douhard M (2017) Offspring sex ratio in mammals and the Trivers-Willard hypothesis: in pursuit of unambiguous evidence. BioEssays 39:1700043. https://doi.org/10.1002/bies.201700043
Emery Thompson M (2017) Energetics of feeding, social behavior, and life history in non-human primates. Horm Behav 91:84–96. https://doi.org/10.1016/j.yhbeh.2016.08.009
Emery Thompson M, Knott CD (2008) Urinary C-peptide of insulin as a non-invasive marker of energy balance in wild orangutans. Horm Behav 53:526–535. https://doi.org/10.1016/j.yhbeh.2007.12.005
Fairbanks LA, Hinde K (2013) Behavioral response of mothers and infants to variation in maternal condition: adaptation, compensation, and resilience. In: Clancy KBH, Hinde K, Rutherford JN (eds) Building babies: primate development in proximate and ultimate perspective. Springer, New York, NY, pp 281–302
Fairbanks LA, Mcguire MT (1995) Maternal condition and the quality of maternal care in vervet monkeys. Behaviour 132:733–754. https://doi.org/10.1163/156853995X00126
Fisher RA (1930) The genetical theory of natural selection. Oxford university press, Oxford
Förster S, Cords M (2004) Development of mother-infant relationships and infant behavior in wild blue monkeys (Cercopithecus mitis stuhlmanni). In: Glens ME, Cords M (eds) The guenons: diversity and adaptation in African monkeys. Springer, Boston, MA, pp 245–272
Froehlich JW, Thorington RW, Otis JS (1981) The demography of Howler Monkeys (Alouatta palliata) on Barro Colorado Island, Panamá. Int J Primatol 2:207–236. https://doi.org/10.1007/BF02739331
Fujita M, Roth E, Lo Y-J et al (2012) In poor families, mothers’ milk is richer for daughters than sons: a test of Trivers-Willard hypothesis in agropastoral settlements in Northern Kenya. Am J Phys Anthropol 149:52–59. https://doi.org/10.1002/ajpa.22092
Geary MPP, Pringle PJ, Rodeck CH et al (2003) Sexual dimorphism in the growth hormone and insulin-like growth factor axis at birth. J Clin Endocrinol Metab 88:3708–3714. https://doi.org/10.1210/jc.2002-022006
Hamilton WD (1967) Extraordinary sex ratios. Science 156:477–488. https://doi.org/10.1126/science.156.3774.477
Hewison AJM, Gaillard J-M (1999) Successful sons or advantaged daughters? The Trivers-Willard model and sex-biased maternal investment in ungulates. Trends Ecol Evol 14:229–234. https://doi.org/10.1016/S0169-5347(99)01592-X
Hinde K (2009) Richer milk for sons but more milk for daughters: sex-biased investment during lactation varies with maternal life history in rhesus macaques. Am J Hum Biol 21:512–519. https://doi.org/10.1002/ajhb.20917
Johnson RL, Southwick CH (1987) Ecological constraints on the development of infant independence in rhesus. Am J Primatol 13(2):103–118
Kanai N, Fujii T, Saito K, Tokoyama T (1994) Rapid and simple method for preparation of genomic DNA from easily obtainable clotted blood. J Clin Pathol 47:1043–1044. https://doi.org/10.1136/jcp.47.11.1043
Keller MC, Nesse RM, Hofferth S (2001) The Trivers-Willard hypothesis of parental investment: no effect in the contemporary United States. Evol Hum Behav 22:343–360. https://doi.org/10.1016/S1090-5138(01)00075-7
Koban T, Brent L, Evan S (2003) The influence of temperature on the behavior of captive mother-infant baboons. Behaviour 140(2):209–224
Labov JB, William Huck U, Vaswani P, Lisk RD (1986) Sex ratio manipulation and decreased growth of male offspring of undernourished golden hamsters (Mesocricetus auratus). Behav Ecol Sociobiol 18:241–249. https://doi.org/10.1007/BF00300000
Le Boeuf BJ (1974) Male-male competition and reproductive success in elephant seals. Integr Comp Biol 14:163–176. https://doi.org/10.1093/icb/14.1.163
Leimar O (1996) Life-history analysis of the Trivers and Willard sex-ratio problem. Behav Ecol 7:316–325. https://doi.org/10.1093/beheco/7.3.316
Lonsdorf EV (2017) Sex differences in nonhuman primate behavioral development. J Neurosci Res 95:213–221. https://doi.org/10.1002/jnr.23862
Maynard Smith J (1980) A new theory of sexual investment. Behav Ecol Sociobiol 7:247–251. https://doi.org/10.1007/BF00299371
McClure PA (1981) Sex-biased litter reduction in food-restricted wood rats (Neotoma floridana). Science 211:1058–1060. https://doi.org/10.1126/science.211.4486.1058
Milton K, Nolin DA, Ellis K et al (2016) Genetic, spatial, and social relationships among adults in a group of howler monkeys (Alouatta palliata) from Barro Colorado Island, Panama. Primates 57:253–265. https://doi.org/10.1007/s10329-016-0523-5
Nguyen N, Gesquiere L, Alberts SC, Altmann J (2012) Sex differences in the mother–neonate relationship in wild baboons: social, experiential and hormonal correlates. Anim Behav 83:891–903. https://doi.org/10.1016/j.anbehav.2012.01.003
Nidiffer MD, Cortés-Ortiz L (2015) Intragroup genetic relatedness in two howler monkey species (Alouatta pigra and A. palliata): implications for understanding social systems and dispersal. Am J Primatol 77:1333–1345. https://doi.org/10.1002/ajp.22487
Ono KA, Boness DJ (1996) Sexual dimorphism in sea lion pups: differential maternal investment, or sex-specific differences in energy allocation? Behav Ecol Sociobiol 38:31–41. https://doi.org/10.1007/s002650050214
Perret M (1990) Influence of social factors on sex ratio at birth, maternal investment and young survival in a prosimian primate. Behav Ecol Sociobiol 27:447–454. https://doi.org/10.1007/BF00164072
Perret M (1996) Manipulation of sex ratio at birth by urinary cues in a prosimian primate. Behav Ecol Sociobiol 38:259–266. https://doi.org/10.1007/s002650050241
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.Rproject.org/
Raguet-Schofield M, Pavé R (2015) An ontogenetic framework for Alouatta: infant development and evaluating models of life history. In: Kowalewski MM, Garber PA, Cortés-Ortiz L et al (eds) Howler monkeys: adaptive radiation, systematics, and morphology. Springer, New York, NY, pp 289–316
Rangel-Negrín A, Coyohua-Fuentes A, Chavira-Ramírez DR et al (2018) Energetic constraints on the reproduction of female mantled howlers. Am J Primatol 80:e22925. https://doi.org/10.1002/ajp.22925
Reiter J, Stinson NL, Le Boeuf BJ (1978) Northern elephant seal development: the transition from weaning to nutritional independence. Behav Ecol Sociobiol 3:337–367. https://doi.org/10.1007/BF00303199
Remick D (1992) Measuring the costs of reproduction. Trends Ecol Evol 7:42–45. https://doi.org/10.1016/0169-5347(92)90104-J
Rosenblum LA, Sunderland G (1982) Feeding ecology and mother-infant relations. In: Hoffman LW, Gandelman RJ, Schiffman HR (eds) Parenting: its causes and consequences. Erlbaum, NJ, pp 75–110
Ross C (2001) Park or Ride? Evolution of Infant Carrying in Primates. Int J Primatol 22:749–771
Schino G, D’Amato FR, Troisi A (1995) Mother-infant relationships in Japanese macaques: sources of inter-individual variation. Anim Behav 49:151–158. https://doi.org/10.1016/0003-3472(95)80162-6
Sikes RS (1995) Maternal response to resource limitations in eastern woodrats. Anim Behav 49:1551–1558. https://doi.org/10.1016/0003-3472(95)90076-4
Silk JB (1980) Kidnapping and female competition among captive bonnet macaques. Primates 21:100–110
Silk JB, Brown GR (2004) Sex ratios in primate groups. In: Kappeler PM, van Schaik CP (eds) Sexual selection in primates: new and comparative perspectives. Cambridge University Press, Cambridge, pp 253–265
Soto M, Gama M (1997) Climas. In: González-Soriano E, Dirzo R, Vogt R (eds) Historia natural de Los Tuxtlas. UNAM and CONABIO, Mexico City, pp 7–23
Treves A (2000) Theory and method in studies of vigilance and aggregation. Anim Behav 60:711–722
Trivers RL (1972) Parental investment and sexual selection. In: Campbell B (ed) Sexual selection and the descent of man. Aldine, Chicago, pp 1871–1971
Trivers RL, Willard DE (1973) Natural selection of parental ability to vary the sex ratio of offspring. Science 179:90–92. https://doi.org/10.1126/science.179.4068.90
Veller C, Haig D, Nowak MA (2016) The Trivers-Willard hypothesis: sex ratio or investment? Proc R Soc Lond B Biol Sci 283:20160126. https://doi.org/10.1098/rspb.2016.0126
Wang E, Milton K (2003) Intragroup social relationships of male Alouatta palliata on Barro Colorado Island, Republic of Panama. Int J Primatol 24:1227–1243. https://doi.org/10.1023/B:IJOP.0000005989.29238.ce
Wright SL, Crawford CB, Anderson JL (1988) Allocation of reproductive effort in Mus domesticus: responses of offspring sex ratio and quality to social density and food availability. Behav Ecol Sociobiol 23:357–365. https://doi.org/10.1007/BF00303709
Xu R (2003) Measuring explained variation in linear mixed effects models. Stat Med 22:3527–3541. https://doi.org/10.1002/sim.1572
Young RL, Badyaev AV (2004) Evolution of sex-biased maternal effects in birds: I. Sex-specific resource allocation among simultaneously growing oocytes. J Evol Biol 17:1355–1366. https://doi.org/10.1111/j.1420-9101.2004.00762.x
Acknowledgements
We thank P. Cruz-Miros, Don J. Landa, and several volunteers for their help during fieldwork. We thank La Flor de Catemaco S.A. de C.V., Gen. J.A. González de la Fuente, and various landowners at Balzapote for granting access to their properties. Ing. J.L. Ponce Puente facilitated our work at La Flor de Catemaco, and A. Molina provided vital administrative support to our project. We thank F. Aureli, S. Torre, J.C. Bicca Marques (Associate Editor), and two anonymous reviewers for providing invaluable feedback to the work. This study was supported by Consejo Nacional de Ciencia y Tecnología (grants 866671/629266 and ciencia básica 254217). This study was noninvasive and complied with the guidelines of the Primate Society of Japan. Research protocols were approved by the Secretaria de Medio Ambiente y Recursos Naturales (permits SGPA/DGVS/0381/17) and adhered to the legal requirements of the Mexican law (NOM-059-SEMARNAT-2010). A. de la Torre would like to extend her sincere gratitude to Dario de la Torre and Mary Paz Herrera for support. P.A.D. Dias and A. Rangel-Negrín thank Mariana and Fernando for constant support and inspiration to study primate behavior.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
de la Torre, A., Coyohua Fuentes, A., Rangel Negrín, A. et al. Maternal care according to offspring sex and maternal physical condition in mantled howler monkeys (Alouatta palliata). Primates 62, 379–388 (2021). https://doi.org/10.1007/s10329-020-00883-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-020-00883-6