Abstract
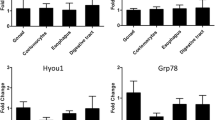
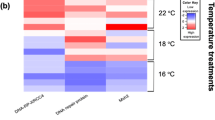
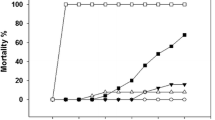
Thermal and saline variations of the Southern Ocean are important signs of climate change which can alter the physiological responses of stenotic species residing at high latitudes. Our study aimed to evaluate the cellular stress response (CSR) of Harpagifer antarcticus subjected to increased ambient temperature and decreased salinity. The fish were distributed in different thermal (2, 5, 8, 11, and 14 °C) and saline (23, 28, and 33 psu) combinations for 10 days. We used qPCR analysis to evaluate the transcription of genes involved in the thermal shock response (HSP70, HSC70, HSP90, and GRP78), ubiquitination (E2, E3, ubiquitin, and CHIP), 26S proteasome complex (PSMA2, PSMB7, and PSMC1), and apoptosis (SMAC/Diablo and BAX) in the liver and gill. The expression profiles were tissue-specific and mainly dependent on temperature rather than salinity in the gill; meanwhile, in the liver, both conditions modulated the expression of these genes. Transcription of markers involved in the heat shock response was much higher in the liver than in the gill and was higher when salinity decreased and the temperature increased. Similarly, the genes involved in the ubiquitination pathway, 26S complex of the proteasome, and the apoptotic pathway showed the same pattern, being mainly induced in the liver rather than in the gill. This is the first study to show that this Antarctic fish can induce the cellular stress response in their tissues when subjected to these thermal/saline combinations.








Similar content being viewed by others
References
Andriashev AP (1965) A general review of the Antarctic fish fauna. Springer Dordr:491–550
Barton BA (2002) Stress in fishes: a diversity of responses with particular reference to changes in circulating corticosteroids. Integr Comp Biol 42:517–525
Bilyk K, Vargas-Chacoff L, Cheng CHC (2018) Evolution in chronic cold: varied loss of cellular response to heat in Antarctic notothenioid fish. BMC Evol Biol 18:1–16
Cárdenas CA, González-aravena M, Santibañez PA (2018) The importance of local settings: within- year variability in seawater temperature at South Bay, Western Antarctic Peninsula. PeerJ 6:e4289
Cheng CH, Yang FF, Liao SA, Miao YT, Ye CX, Wang AL, Tan JW, Chen XY (2015) High temperature induces apoptosis and oxidative stress in pufferfish (Takifugu obscurus) blood cells. J Therm Biol 53:172–179
Ching B, Chen XL, Yong JHA et al (2013) Increases in apoptosis, caspase activity and expression of p53 and bax, and the transition between two types of mitochondrion-rich cells, in the gills of the climbing perch, Anabas testudineus, during a progressive acclimation from freshwater to seawater. Front Physiol:1–21
Ciechanover A (2005) Intracellular protein degradation: from a vague idea thru the lysosome and the ubiquitin – proteasome system and onto human diseases and drug targeting. Angew Chem Int Ed Engl 44:5944–5967
Clark MS, Fraser KPP, Burns G, Peck LS (2008) The HSP70 heat shock response in the Antarctic fish Harpagifer antarcticus. Polar Biol 31:171–180
Constable AJ, Melbourne-thomas J, Corney SP et al (2014) Climate change and Southern Ocean ecosystems I: how changes in physical habitats directly affect marine biota. Glob Chang Biol 20:3004–3025
Dalvi R, Das T, Debnath D et al (2017) Metabolic and cellular stress responses of catfish, Horabagrus brachysoma (Günther) acclimated to increasing temperatures. J Therm Biol 65:32–40
Deane EE, Kelly SP, Luk JCY, Woo NYS (2002) Chronic salinity adaptation modulates hepatic heat shock protein and insulin-like growth factor I expression in black sea bream. Mar Biotechnol 4:193–205
Evans TG, Kültz D (2020) The cellular stress response in fish exposed to salinity fluctuations. J Exp Zool Part A Ecol Integr Physiol 1–15.
Guan Y, Zhang G xia, Zhang S, et al. (2016) The potential role of polyamines in gill epithelial remodeling during extreme hypoosmotic challenges in the Gulf killifish, Fundulus grandis. Comp Biochem Physiol Part - B Biochem Mol Biol 194–195:39–50
Haumann FA, Gruber N, Münnich M, Frenger I, Kern S (2016) Sea-ice transport driving southern ocean salinity and its recent trends. Nature 537:89–92
Hofmann GE, Buckley BA, Airaksinen S, Keen JE, Somero GN (2000) Heat-shock protein expression is absent in the Antarctic fish Trematomus bernacchii (family nototheniidae). J Exp Biol 203:2331–2339
Huang R, Zhang J, Zhu G, He J, Xie J (2017) The core ubiquitin system of mandarin fish, Siniperca chuatsi, can be utilized by infectious spleen and kidney necrosis virus. Fish Shellfish Immunol 70:293–301
Inokuchi M, Kaneko T (2012) Recruitment and degeneration of mitochondrion-rich cells in the gills of Mozambique tilapia Oreochromis mossambicus during adaptation to a hyperosmotic environment. Comp Biochem Physiol - A Mol Integr Physiol 162:245–251
Laurent P, Chevalier C, Wood CM (2006) Appearance of cuboidal cells in relation to salinity in gills of Fundulus heteroclitus, a species exhibiting branchial Na+ but not Cl- uptake in freshwater. Cell Tissue Res 325:481–492
Liston P, Fong WG, Korneluk RG (2003) The inhibitors of apoptosis: there is more to life than Bcl2. Oncogene 22:8568–8580
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔct Method. Methods 25:402–408
Martínez D, Vargas-Lagos C, Saravia J, Oyarzún R, Loncoman C, Pontigo JP, Vargas-Chacoff L (2020) Cellular stress responses of Eleginops maclovinus fish injected with Piscirickettsia salmonis and submitted to thermal stress. Cell Stress Chaperones 25:93–104
Navarro JM, Paschke K, Ortiz A, Vargas-Chacoff L, Pardo LM, Valdivia N (2019) The Antarctic fish Harpagifer antarcticus under current temperatures and salinities and future scenarios of climate change. Progess Oceanogr 174:37–43
Pawlowski J, Kraft AS (2000) Bax-induced apoptotic cell death. Proc Natl Acad Sci U S A 97:529–531
Rasmussen R (2001) Rapid cyclee real-time PCR, Methods and Applications.
Sherman MY, Goldberg AL (2001) Cellular defenses against unfolded proteins: a cell biologist thinks about neurodegenerative diseases. Neuron 29:15–32
Thorne MAS, Burns G, Fraser KPP, Hillyard G, Clark MS (2010) Transcription profiling of acute temperature stress in the Antarctic plunderfish Harpagifer antarcticus. Mar Genomics 3:35–44
Todgham AE, Crombie TA, Hofmann GE (2017) The effect of temperature adaptation on the ubiquitin–proteasome pathway in notothenioid fishes. J Exp Biol 220:369–378
Vargas-Chacoff L, Muñoz JL, Ocampo D et al (2019) The effect of alterations in salinity and temperature on neuroendocrine responses of the Antarctic fish Harpagifer antarcticus. Comp Biochem Physiol -Part A 235:131–137
Wendelaar-Bonga E (1997) The stress response in fish. Physiol Rev 77:591–625
Zhang Y, Chou SD, Murshid A et al (2011) The role of heat shock factors in stress-induced transcription. Methods Mol Biol 787:21–32
Availability of data and material
All relevant data are within the paper and are available with the corresponding author.
Code availability
Not applicable
Funding
This investigation was financed by Fondap-Ideal Grant No. 15150003 and Fondecyt Regular Grant No. 1160877. D. Martínez was funded by the Agencia Nacional de Investigación y Desarrollo (ANID) through a national Postdoctoral scholarship No. 3200418. The Instituto Antártico Chileno (INACH) for logistic support.
Author information
Authors and Affiliations
Contributions
Conceptualization, L.V.CH., K.P., and J.N.; methodology, C.M.K., D.M., and L.V.CH.; investigation, L.V.CH., C.M.K., and D.M.; resources, L.V.CH., J.N., and K.P.; writing-original draft, D.M. and C.M.K.; writing-review and editing, C.M.K. and D.M.; supervision, L.V.CH.; funding acquisition, L.V.CH., K.P., and J.N.
Corresponding authors
Ethics declarations
Ethics approval
The experiment was performed following the standards of the Guide for Care and Use of Laboratory Animals of the National Commission of Science and Technology (CONICYT, Chile) and the Universidad Austral de Chile.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Martínez, D., Moncada-Kopp, C., Paschke, K. et al. Warming and freshening activate the transcription of genes involved in the cellular stress response in Harpagifer antarcticus. Fish Physiol Biochem 47, 533–546 (2021). https://doi.org/10.1007/s10695-021-00931-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00931-y