Abstract
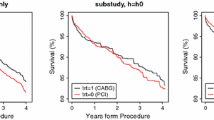
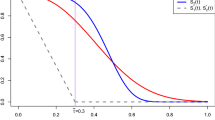
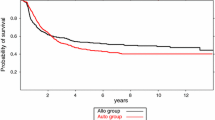
For equivalence trials with survival outcomes, a popular testing approach is the elegant test for equivalence of two survival functions suggested by Wellek (Biometrics 49: 877–881, 1993). This test evaluates whether or not the difference between the true survival curves is practically irrelevant by specifying an equivalence margin on the hazard ratio under the proportional hazards assumption. However, this approach is based on extrapolating the behavior of the survival curves to the whole time axis, whereas in practice survival times are only observed until the end of follow-up. We propose a modification of Welleks test that only addresses equivalence until end of follow-up and derive the large sample properties of this test. Another issue is the proportional hazards assumption which may not be realistic. If this assumption is violated, one may severely misjudge the actual treatment effect with a hazard ratio quantification and wrongly declare equivalence. We suggest a non-parametric test for assessing survival equivalence within the follow-up period. We derive the large sample properties of this test and provide an approximation to the limiting distribution under some mild assumptions on the functional form of the difference between the two survival curves. Both suggestions are investigated by simulation and applied to a clinical trial on survival of gastric cancer patients.



Similar content being viewed by others
References
Andersen PK, Borgan Ø, Gill RD, Keiding N (1993) Statistical models based on counting processes. Springer, New York
Borgan Ø, Liestøl K (1990) A note on confidence intervals and bands for the survival function based on transformations. Scand J Stat 17:35–41
Com-Nogue C, Rodary C, Patte C (1993) How to establish equivalence when data are censored: a randomized trial of treatments fo b non hodkin lymphoma. Stat Med 12:1353–1364
Dette H, Möllenhoff K, Volgushev S, Bretz F (2018) Equivalence of regression curves. J Am Stat Assoc 113:711–729
Fleming TR, O’Fallon JR, O’Brien PC, Harrington DP (1980) Modified Kolmogorov–Smirnov test procedures with application to arbitrarily right-censored tests. Biometrics 36:607–625
Freitag G, Lange S, Munk A (2006) Non-parametric assessment of non-inferiority with censored data. Stat Med 25:1201–1217
Koti KM (2013) New tests for assessing non-inferiority and equivalence from survival data. Open J Stat 3:55–64
Martinez E, Sinha D, Wang W, Lipsitz S, Chappel S (2017) Tests for equivalence of two survival functions: alternative to the tests under proportional hazards. Stat Methods Med Res 26:75–87
Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DA, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, Chewaskulyong B, Jiang H, Duffield EL, Watkins CL, Armour AA, Fukuoka M (2009) Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 361:947–957
Parzen MJ, Wei LJ, Ying Z (1997) Simultaneous confidence intervals for the difference of two survival functions. Scand J Stat 24:309–314
Schoenfeld D (1983) Sample-size formula for the proportional hazards regression model. Biometrics 39:499–503
Shao J, Tu D (1995) The jacknife and bootstrap. Springer, New York
The Gastrointestinal Tumor Study Group (1982) A comparison of combination chemotherapy and combined modality therapy for locally advanced gastric carcinoma. Cancer 49:1771–1777
Uno H, Wittes J, Fu H, Solomon S, Claggett B, Tian L, Cai T, Pfeffer MA, Evans SR, Wei LJ (2015) Alternatives to hazard ratios for comparing the efficacy or safety of therapies in noninferiority studies. Ann Intern Med 163:127–134
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, for the STROBE Initiative (2008) Strengthening the reporting of observational studies in epidemiology (strobe) statement: guidelines for reporting observational studies. J Clin Epidemiol 61:344–349
Wellek S (1993) A log-rank test for equivalence of two survivor functions. Biometrics 49:877–881
Westlake WJ (1972) Use of confidence intervals in analysis of comparative bioavailability trials. J Pharm Sci 61:1340–1341
Zhao L, Claggett B, Tian L, Uno H, Pfeffer MA, Solomon SD, Trippa L, Wei L-J, (2016) On the restricted mean survival time curve in survival analysis. Biometrics 72:215–221
Acknowledgements
We thank the Associate editor and the two reviewers for their valuable suggestions and comments that substantially improved earlier versions of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
A Proof of Proposition 1
When \(\theta \ne 1\) a straightforward application of the Delta Theorem shows that
where \(\mathcal {Z}=(\mathcal {Z}_{1},\mathcal {Z}_{2})\) is normally distributed with mean 0 and \(var(\mathcal {Z}_{1})=\sigma _{1}^{2}\), \(var(\mathcal {Z}_{2})=\sigma _{2}^{2}\), and
Now define \(\hat{P}\) as the indicator of \( \hat{S}_{0}(\tau ) > \hat{\theta }^{\frac{1}{1-\hat{\theta }}}\), that is
Then \(\hat{m}_{\tau }\) may be rewritten as:
When \(S_{0}(\tau )<\theta ^{\frac{1}{1-\theta }}\) we have that \(m_{\tau }=m_{1}(\tau )\) and that \(\hat{P}\) converges in probability to 0. Consequently
and thus in this situation
When \(S_{0}(\tau )>\theta ^{\frac{1}{1-\theta }}\) we have that \(m_{\tau }=m_{2}(\tau )\) and that \(\hat{P}\) converges in probability to 1. Consequently
and thus in this situation
When \(S_{0}(\tau )=\theta ^{\frac{1}{1-\theta }}\) we have that \(m_{\tau }=m_{1}(\tau )=m_{2}(\tau )\). Moreover a straightforward calculation shows that \(\sigma _{1}^{2}=\sigma _{2}^{2}\) and that \(cor(\mathcal {Z}_{1},\mathcal {Z}_{2})=1\). Accordingly, since \(\mathcal {Z}_{1}-\mathcal {Z}_{2}=0\), we may conclude by the Continuous Mapping Theorem that:
Consequently
and thus in this situation
This proves the first claim of Proposition 1.
To prove that \(\hat{\xi }^{2}\) converges in probability to \(\xi ^{2}\) note that \(\hat{\sigma }_{1}^{2}\) and \(\hat{\sigma }_{2}^{2}\) converge in probability to \(\sigma _{1}^{2}\) and \(\sigma _{2}^{2}\). Also note that
The proof now follows along the same lines as the proof of the first claim of Proposition 1.
B Proof of Proposition 2
The claim that
can be verified by copying the proof of Theorem 4.1 in Dette et al. (2018). For the special case of \(\mathcal {E}^{\pm }(f)=\{t^{*}\}\) we have that
From which it follows directly that
Now assume that \(\mathcal {E}^{\pm }(f)=\mathcal {E}^{+}(f)=\{t^{*}\}\) and let \(t^{\star }_{n}\in \mathcal {E}^{\pm }(f_{n})\). We proceed by showing that
By the continuous mapping theorem and the weak convergence of \(G_{n}\) we have that
Accordingly
It now follows from
that
From this we deduce that since \(\mathcal {E}^{\pm }(f)=\mathcal {E}^{+}(f)=\{t^{*}\}\) and consequently \(\sup _{t\in [0,\tau ]}\{-f(t)\}<\sup _{t\in [0,\tau ]}f(t)\) it holds that
Since \(t^{\star }_{n}\in \mathcal {E}^{+}(f_{n})\) when \(\sup _{t\in [0,\tau ]}\{-f_{n}(t)\}<\sup _{t\in [0,\tau ]}f_{n}(t)\) we conclude that (7) holds.
Next we note that as a direct consequence of (8) we have
from which it follows that
When \(t^{\star }_{n}\in \mathcal {E}^{+}(f_{n})\) and \(t^{\star }_{n}\in \mathcal {E}^{\pm }(f_{n})\) we have per definition that
Combining the above we conclude that
Now note that by the definition of \(t^{\star }\) we have \(f(t)<f(t^{\star })\) for all \(t\in [0,\tau ]\) with \(t\ne t^{\star }\). Hence continuity of f and compactness of \([0,\tau ]\) implies that
Accordingly for any \(\delta >0\) the exists a \(\varepsilon >0\) so that
Since the right hand side of the above inequality converges to zero as \(n\rightarrow \infty \) we conclude that:
This claim is shown by similar arguments when \(\mathcal {E}^{\pm }(f)=\mathcal {E}^{-}(f)=\{t^{*}\}\).
Finally it follows from the continuity of \(\sigma _{0}^{2}(t)\) and \(\sigma _{1}^{2}(t)\) combined with the uniform convergence in probability of their respective estimators that
This concludes the proof of Proposition 2.
C Formulas for asymptotic variances
Let \(T_{i}\), \(C_{i}\), and \(Z_{i}\) denote the failure time, censoring time, and treatment arm of the ith subject, respectively. Define the counting process and at risk process of the ith individual as:
Then with
it follows directly from the derivations in Chapter VII.2.2 of Andersen et al. (1993) that consistent estimates of \(\sigma ^2\), \(\omega ^2\), and \(\rho \) are given by:
Now let
Then it follows directly from Theorem IV.3.2. in Chapter IV.3.2 of Andersen et al. (1993) that for \(t\le \tau \):
Rights and permissions
About this article
Cite this article
Furberg, J.K., Pipper, C.B. & Scheike, T. Testing equivalence of survival before but not after end of follow-up. Lifetime Data Anal 27, 216–243 (2021). https://doi.org/10.1007/s10985-021-09517-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10985-021-09517-5