Abstract
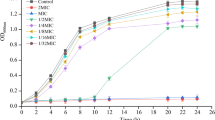
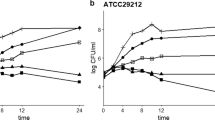
As a primary cause of food contamination and human diseases, Salmonella Typhimurium can easily form a biofilm that is difficult to remove from food surfaces, and often causes significant invisible threats to food safety. Although berberine has been widely used as an anti-infective drug in traditional medicine, some basic principles underlying its mechanism, especially the interaction between berberine and type I fimbriae genes, has not been verified yet. In this study, two strains of major fimbrial gene mutants (ΔfimA and ΔfimH) were constructed to demonstrate the possible action of berberine on type I fimbriae genes. The broth microdilution method was used to determine the antibacterial activity of berberine against selected strains (WT, ΔfimA, and ΔfimH). Cell agglutination experiments revealed that the number of S. Typhimurium type I fimbriae reduced after berberine treatment, which was consistent with transmission electron microscopy results. Quantitative real-time PCR experiments also confirmed that berberine reduced fimA gene expression, indicating a certain interaction between berberine and fimA gene. Furthermore, confocal laser scanning microscopy imaging of biofilm clearly revealed that berberine prevents biofilm formation by reducing the number of type I fimbriae. Overall, it is well speculated for us that berberine could be an excellent combating-biofilm drug in clinical microbiology and food preservation.
Key points
• Reduce the number of fimbriae.
• Berberine targeting fimA.
• Effective biofilm inhibitor.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Andino A, Hanning I (2015) Salmonella enterica: survival, colonization, and virulence differences among serovars. Sci World J 2015:520179–520179. https://doi.org/10.1155/2015/520179
Aswathanarayan JB, Vittal RR (2018) Inhibition of biofilm formation and quorum sensing mediated phenotypes by berberine in Pseudomonas aeruginosa and Salmonella Typhimurium. RSC Adv 8(63):36133–36141. https://doi.org/10.1039/C8RA06413J
Baugh S, Ekanayaka AS, Piddock LJV, Webber MA (2012) Loss of or inhibition of all multidrug resistance efflux pumps of Salmonella enterica serovar Typhimurium results in impaired ability to form a biofilm. J Antimicrob Chemoth 67(10):2409–2417. https://doi.org/10.1093/jac/dks228
Boberek JM, Stach J, Good L (2010) Genetic evidence for inhibition of bacterial division protein FtsZ by berberine. PLoS One 5(10):e13745–e13745. https://doi.org/10.1371/journal.pone.0013745
Chen L, Bu Q, Xu H, Liu Y, She P, Tan R, Wu Y (2016) The effect of berberine hydrochloride on Enterococcus faecalis biofilm formation and dispersion in vitro. Microbiol Res 186-187:44–51. https://doi.org/10.1016/j.micres.2016.03.003
Chu M, Ding R, Z-y C, M-b Z, X-y L, Xie S-h, Y-j Z, Wang Y-d (2014) Role of berberine in anti-bacterial as a high-affinity LPS antagonist binding to TLR4/MD-2 receptor. BMC Complem Altern M 14:89–89. https://doi.org/10.1186/1472-6882-14-89
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97(12):6640–6645. https://doi.org/10.1073/pnas.120163297
Eran Z, Akçelik M, Yazıcı BC, Özcengiz G, Akçelik N (2020) Regulation of biofilm formation by marT in Salmonella Typhimurium. Mol Biol Rep 47(7):5041–5050. https://doi.org/10.1007/s11033-020-05573-6
Fàbrega A, Vila J (2013) Salmonella enterica serovar Typhimurium skills to succeed in the host: virulence and regulation. Clin Microbiol Rev 26(2):308–341. https://doi.org/10.1128/CMR.00066-12
Flemming H-C, Wingender J, Szewzyk U, Steinberg P, Rice SA, Kjelleberg S (2016) Biofilms: an emergent form of bacterial life. Nat Rev Microbiol 14(9):563–575. https://doi.org/10.1038/nrmicro.2016.94
Habtemariam S (2020) Berberine pharmacology and the gut microbiota: a hidden therapeutic link. Pharmacol Res 155:104722. https://doi.org/10.1016/j.phrs.2020.104722
Hospenthal MK, Costa TRD, Waksman G (2017) A comprehensive guide to pilus biogenesis in gram-negative bacteria. Nat Rev Microbiol 15(6):365–379. https://doi.org/10.1038/nrmicro.2017.40
Imenshahidi M, Hosseinzadeh H (2016) Berberis vulgaris and berberine: an update review. Phytother Res 30(11):1745–1764. https://doi.org/10.1002/ptr.5693
Karaosmanoglu K, Sayar NA, Kurnaz IA, Akbulut BS (2013) Assessment of berberine as a multi-target antimicrobial: a multi-omics study for drug discovery and repositioning. OMICS: OMICS 18(1):42–53. https://doi.org/10.1089/omi.2013.0100
Koo H, Allan RN, Howlin RP, Stoodley P, Hall-Stoodley L (2017) Targeting microbial biofilms: current and prospective therapeutic strategies. Nat Rev Microbiol 15(12):740–755. https://doi.org/10.1038/nrmicro.2017.99
Korhonen TK (1979) Yeast cell agglutination by purified enterobacterial pili. FEMS Microbiol Lett 6(6):421–425. https://doi.org/10.1111/j.1574-6968.1979.tb03756.x
Korhonen TK, Lounatmaa K, Ranta H, Kuusi N (1980) Characterization of type 1 pili of Salmonella Typhimurium LT2. J Bacteriol 144(2):800–805. https://doi.org/10.1128/JB.144.2.800-805.1980
Kuan N-L, Yeh K-S (2019) Arginine within a specific motif near the N-terminal of FimY is critical for the maximal production of type 1 fimbriae in Salmonella enterica serovar Typhimurium. MICROBIOLOGYOPEN 8(9):e00846–e00846. https://doi.org/10.1002/mbo3.846
Kuehn B (2019) Multidrug-resistant Salmonella. JAMA 322(14):1344–1344. https://doi.org/10.1001/jama.2019.15309
Kumar A, Ekavali CK, Mukherjee M, Pottabathini R, Dhull DK (2015) Current knowledge and pharmacological profile of berberine: an update. Eur J Pharmacol 761:288–297. https://doi.org/10.1016/j.ejphar.2015.05.068
Ledeboer NA, Frye JG, McClelland M, Jones BD (2006) Salmonella enterica serovar Typhimurium requires the Lpf, Pef, and Tafi fimbriae for biofilm formation on HEp-2 tissue culture cells and chicken intestinal epithelium. Infect Immun 74(6):3156–3169. https://doi.org/10.1128/IAI.01428-05
Liu Q, Niu H, Zhang W, Mu H, Sun C, Duan J (2015) Synergy among thymol, eugenol, berberine, cinnamaldehyde and streptomycin against planktonic and biofilm-associated food-borne pathogens. Lett Appl Microbiol 60(5):421–430. https://doi.org/10.1111/lam.12401
Liu Y, Liu X, Zhang N, Yin M, Dong J, Zeng Q, Mao G, Song D, Liu L, Deng H (2020) Berberine diminishes cancer cell PD-L1 expression and facilitates antitumor immunity via inhibiting the deubiquitination activity of CSN5. Acta Pharm Sin B 10:2299–2312. https://doi.org/10.1016/j.apsb.2020.06.014
Lukaszczyk M, Pradhan B, Remaut H (2019) The biosynthesis and structures of bacterial pili. In: Kuhn A (ed) Bacterial cell walls and membranes. Springer International Publishing, Cham, pp 369–413
Meng X, Meng X, Wang J, Wang H, Zhu C, Ni J, Zhu G (2019) Small non-coding RNA STnc640 regulates expression of fimA fimbrial gene and virulence of Salmonella enterica serovar Enteritidis. BMC Vet Res 15(1):319–319. https://doi.org/10.1186/s12917-019-2066-7
Michael GB, Schwarz S (2016) Antimicrobial resistance in zoonotic nontyphoidal Salmonella: an alarming trend? Clin Microbiol Infec 22(12):968–974. https://doi.org/10.1016/j.cmi.2016.07.033
Proft T, Baker EN (2008) Pili in gram-negative and gram-positive bacteria — structure, assembly and their role in disease. Cell Mol Life Sci 66(4):613–635. https://doi.org/10.1007/s00018-008-8477-4
Roy R, Tiwari M, Donelli G, Tiwari V (2018) Strategies for combating bacterial biofilms: a focus on anti-biofilm agents and their mechanisms of action. VIRULENCE 9(1):522–554. https://doi.org/10.1080/21505594.2017.1313372
Shi C, Li M, Muhammad I, Ma X, Chang Y, Li R, Li C, He J, Liu F (2018) Combination of berberine and ciprofloxacin reduces multi-resistant Salmonella strain biofilm formation by depressing mRNA expressions of luxS, rpoE, and ompR. J Vet Sci 19(6):808–816. https://doi.org/10.4142/jvs.2018.19.6.808
Stapels DAC, Hill PWS, Westermann AJ, Fisher RA, Thurston TL, Saliba A-E, Blommestein I, Vogel J, Helaine S (2018) Salmonella persisters undermine host immune defenses during antibiotic treatment. Science 362(6419):1156–1160. https://doi.org/10.1126/science.aat7148
Steenackers H, Hermans K, Vanderleyden J, De Keersmaecker SCJ (2012) Salmonella biofilms: an overview on occurrence, structure, regulation and eradication. Food Res Int 45(2):502–531. https://doi.org/10.1016/j.foodres.2011.01.038
Sun D, Abraham SN, Beachey EH (1988) Influence of berberine sulfate on synthesis and expression of pap fimbrial adhesin in uropathogenic Escherichia coli. Antimicrob Agents CH 32(8):1274–1277. https://doi.org/10.1128/aac.32.8.1274
Uchiya K-I, Kamimura Y, Jusakon A, Nikai T (2019) Salmonella fimbrial protein FimH is involved in expression of proinflammatory cytokines in a toll-like receptor 4-dependent manner. Infect Immun 87(3):e00881–e00818. https://doi.org/10.1128/IAI.00881-18
Wang K-C, Hsu Y-H, Huang Y-N, Yeh K-S (2012) A previously uncharacterized gene stm0551 plays a repressive role in the regulation of type 1 fimbriae in Salmonella enterica serotype Typhimurium. BMC Microbiol 12:111–111. https://doi.org/10.1186/1471-2180-12-111
Wolfmeier H, Pletzer D, Mansour SC, Hancock REW (2018) New perspectives in biofilm eradication. ACS Infect DIS 4(2):93–106. https://doi.org/10.1021/acsinfecdis.7b00170
Yang Y, Li J, Yin Y, Guo D, Jin T, Guan N, Shi Y, Xu Y, Liang S, Xia X, Shi C (2019) Antibiofilm activity of coenzyme Q0 against Salmonella Typhimurium and its effect on adhesion–invasion and survival–replication. Appl Microbiol Biot 103(20):8545–8557. https://doi.org/10.1007/s00253-019-10095-8
Zeiner SA, Dwyer BE, Clegg S (2012) FimA, FimF, and FimH are necessary for assembly of type 1 fimbriae on Salmonella enterica serovar Typhimurium. Infect Immun 80(9):3289–3296. https://doi.org/10.1128/IAI.00331-12
Zeng L, Zhang L, Wang P, Meng G (2017) Structural basis of host recognition and biofilm formation by Salmonella Saf pili. Elife 6:e28619. https://doi.org/10.7554/eLife.28619
Funding
This work was supported by the National Natural Science Foundation of China (31770109) and (21775036).
Author information
Authors and Affiliations
Contributions
CX designed, supervised the experiments, analyzed the results, revised the first draft, and prepared the last draft of the manuscript. LD performed part of the experiments, contributed significantly to analysis and manuscript preparation, and revised the last draft of the manuscript. FH and FW performed part of the experiments, analyzed the data, and participated in the first draft of the manuscript. DH analyzed the data and helped perform the analysis with constructive discussions. MY collaborated in the design of the experiments and revised different versions of the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no conflict of interest.
Additional information
This paper is our original work. It has not been submitted elsewhere, and it is not under consideration in any other Journal.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, C., Wang, F., Huang, F. et al. Targeting effect of berberine on type I fimbriae of Salmonella Typhimurium and its effective inhibition of biofilm. Appl Microbiol Biotechnol 105, 1563–1573 (2021). https://doi.org/10.1007/s00253-021-11116-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11116-1