Abstract
Introduction
The plasma membrane of a cell undergoes continuous deformations and turning-over of its constituents required for a diverse set of functions. And yet, the membrane retains its steady-state surface tension—without which these functions are adversely affected.
Objectives
In this review, we discuss how the establishment and maintenance of the homeostatic state of the membrane has a major contribution from the actin cytoskeleton. The regulation of tension at the global (or cellular) scale is extensively studied for the past few decades, leading to our understanding of how actin polymerization forces, myosin-II based contractile forces, and ezrin-mediated attachment to the membrane—each have a separate and sometimes multiple possible effects on membrane tension.
Conclusions
Drawing examples from cell motility and blebbing cells, we highlight how the dynamics of the cytoskeleton decides if the steady-state tension has a uniform profile, front-rear gradients, or temporally varying tension profiles in single cells. Non-invasive studies open up new avenues especially allowing the investigations focusing on local regulation of tension—at the plasma membrane and inside cells.



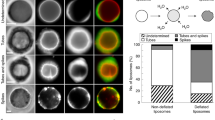
source suffers reflection due to a difference in refractive indices. A camera is used for capturing interference images over time which helps to measure fluctuations and tension. IRM can create maps of tension and requires measurement over 40–100 s. d A cartoon of a migrating cell with front and rear end showing a gradient of membrane tension due to incorporation of FliptR, a molecular probe. The changes can be captured by fluorescence lifetime imaging (FLIM). The probe can detect tension changes because of the fluorescence lifetimes alterations created due to tension planarizing the fluorescent groups. Detecting tension differences by FliptR is a fast process which can used be used all over the cell membrane.

Similar content being viewed by others
References
Morris CE, Homann U (2001) Cell surface area regulation and membrane tension. Membr Biol 179:79–102
Harris H (1999) The birth of the cell. Yale University Press, New Haven and London
Grew N (1682) The anatomy of plants. (W. Rawlins). doi:https://doi.org/10.5962/bhl.title.4
Hooke R (1665) Micrographia: or some physiological descriptions of minute bodies made by magnifying glasses : with observations and inquiries thereupon. Royal Society, London
Schwann T (1839) Mikroskopische Untersuchungen über die Uebereinstimmung in der Struktur und dem Wachsthum der Thiere und Pflanzen. Sander
Stillwell W (2013) Membrane History. in An Introduction to Biological Membranes: From Bilayers to Rafts
Pockels A (1891) Surface tension. Nature 43:437–439
Langmuir I (1917) The constitution and fundamental properties of solids and liquids. II. Liquids. J Am Chem Soc 39:1848–1906
Overton E (1899) Ueber die allgemeinen osmotischen Eigenschaften der Zelle, ihre vermutlichen Ursachen und ihre Bedeutung für die Physiologie. Zürich 64:87–136
Fricke H (1925) The electric capacity of suspensions with special reference to blood. J Gen Physiol 9:137–152
Fricke H, Curtis HJ (1934) Electric impedance of suspensions of yeast cells. Nature 134:102–103
Gorter E, Grendel F (1925) On bimolecular layers of lipoids on the chromocytes of the blood. J Exp Med 41:439–444
Danielli JF, Davson H (1935) A contribution to the theory of permeability of thin films. J Cell Comp Physiol 5:495–508
Robertson JD (1981) Membrane structure. J Cell Biol 91:189–204
Escribá PV (2017) Membrane-lipid therapy: a historical perspective of membrane-targeted therapies—From lipid bilayer structure to the pathophysiological regulation of cells. Biochim Biophys Acta Biomembr 1859:1493–1506
Kalkan KT, Esrefoglu M (2020) The cell membrane: a historical narration. Bezmialem Sci 8:81–88
Lombard J (2014) Once upon a time the cell membranes: 175 years of cell boundary research. Biol Direct 9:1–35
Singer SJ, Nicolson GL (1972) The fluid mosaic model of the structure of cell membranes. Science (80-) 175:720–731
Simons K, Ikonen E (1997) Functional rafts in cell membranes. Nature 387:569–572
Wohlfarth-Bottermann KE (1964) Differentiations of the ground cytoplasm and their significance for the generation of the motive force of ameboid movement. In: Primitive motile systems in cell biology, pp 79–109 (Elsevier, 1964). doi:https://doi.org/10.1016/b978-0-12-395681-1.50013-8
Ervasti JM, Campbell KP (1993) Dystrophin and the membrane skeleton. Curr Opin Cell Biol 5:82–87
Bretscher A (1983) Purification of an 80,000-dalton protein that is a component of the isolated microvillus cytoskeleton, and its localization in nonmuscle cells. J Cell Biol 97:425–432
Tsukita S, Hieda Y, Tsukita S (1989) A new 82-kD barbed end-capping protein (radixin) localized in the cell-to-cell adherens junction: purification and characterization. J Cell Biol 108:2369–2382
Lankes WT, Furthmayr H (1991) Moesin: a member of the protein 4.1-talin-ezrin family of proteins. Proc Natl Acad Sci USA 88:8297–8301
Cohen MH, Turnbull D (1959) Molecular transport in liquids and glasses. J Chem Phys 31:1164–1169
Frye LD, Edidin M (1970) The rapid intermixing of cell surface antigens after formation of mouse-human heterokaryons. J Cell Sci 7:319–335
Axelrod D, Koppel DE, Schlessinger J, Elson E, Webb WW (1976) Mobility measurement by analysis of fluorescence photobleaching recovery kinetics. Biophys J 16:1055–1069
Jékely G (2014) Origin and evolution of the self-organizing cytoskeleton in the network of eukaryotic organelles. Cold Spring Harb Perspect Biol 6
Wegner A (1976) Head to tail polymerization of actin. J Mol Biol 108:139–150
Mullins RD, Heuser JA, Pollard TD (1998) The interaction of Arp2/3 complex with actin: Nucleation, high affinity pointed end capping, and formation of branching networks of filaments. Proc Natl Acad Sci USA 95:6181–6186
Cooper JA, Schafer DA (2000) Control of actin assembly and disassembly at filament ends. Curr Opin Cell Biol 12:97–103
Vignaud T, Blanchoin L, Théry M (2012) Directed cytoskeleton self-organization. Trends Cell Biol 22:671–682
Kühne W (1864) Untersuchungen über das Protoplasma und die Contractilität. (W. Engelmann). doi:https://doi.org/10.5962/bhl.title.46515.
Roth TF, Porter KR (1964) Yolk protein uptake in the oocyte of the mosquito aedes aegyptil. J Cell Biol 20:313–332
Goldstein JL, Anderson RGW, Brown MS (1979) Coated pits, coated vesicles, and receptor-mediated endocytosis. Nature 279:679–685
Ashkin A (1970) Acceleration and trapping of particles by radiation pressure. Phys Rev Lett 24:156–159
Dai J, Sheetz MP (1995) Regulation of endocytosis, exocytosis, and shape by membrane tension. In: Cold Spring Harbor Symposia on Quantitative Biology vol 60:567–571 (Cold Spring Harbor Laboratory Press)
Evans E, Yeung A (1989) Apparent viscosity and cortical tension of blood granulocytes determined by micropipet aspiration. Biophys J 56:151–160
Hochmuth RM (2000) Micropipette aspiration of living cells. J Biomech 33:15–22
Krieg M et al (2008) Tensile forces govern germ-layer organization in zebrafish. Nat Cell Biol 10:429–436
Zilker A, Ziegler M, Sackmann E (1992) Spectral analysis of erythrocyte flickering in the 0.3–4-μm-1 regime by microinterferometry combined with fast image processing. Phys. Rev. A 46:7998–8001
Dai J, Sheetz MP, Wan X, Morris CE (1998) Membrane tension in swelling and shrinking molluscan neurons. J Neurosci 18:6681–6692
Blowers R, Clarkson EM, Maizels M (1951) Flicker phenomenon in human erythrocytes. J Physiol 113:228–239
Colom A et al (2018) A fluorescent membrane tension probe. Nat Chem 10:1118–1125
Nelson DL, Cox MM (2013) Lehninger principles of biochemistry. Winslow, Susan
Nicolson GL (2014) The fluid—mosaic model of membrane structure: still relevant to understanding the structure, function and dynamics of biological membranes after more than 40 years. Biochim Biophys Acta Biomembr 1838:1451–1466
Lodish H et al (2000) Membrane Proteins. in Molecular Cell Biology (W. H. Freeman)
Yang Y, Lee M, Fairn GD (2018) Phospholipid subcellular localization and dynamics. J Biol Chem 293:6230–6240
Raffy S, Teissié J (1999) Control of lipid membrane stability by cholesterol content. Biophys J 76:2072–2080
Houk AR et al (2012) Membrane tension maintains cell polarity by confining signals to the leading edge during neutrophil migration. Cell 148:175–188
Gauthier NC, Fardin MA, Roca-Cusachs P, Sheetz MP, Mogilner A (2011) Temporary increase in plasma membrane tension coordinates the activation of exocytosis and contraction during cell spreading. PNAS 108:14467–14472
Boulant S, Kural C, Zeeh J-C, Ubelmann F, Kirchhausen T (2012) Actin dynamics counteract membrane tension during clathrin- mediated endocytosis. Nat Cell Biol 13:1124–1131
Sukharev S (1999) Mechanosensitive channels in bacteria as membrane tension reporters. FASEB J 13:S55–S61
Denk W, Webb WW (1992) Forward and reverse transduction at the limit of sensitivity studied by correlating electrical and mechanical fluctuations in frog saccular hair cells. Hear Res 60:89–102
Charras GT, Williams BA, Sims SM, Horton MA (2004) Estimating the sensitivity of mechanosensitive ion channels to membrane strain and tension. Biophys J 87:2870–2884
Zhao H, Pykäläinen A, Lappalainen P (2011) I-BAR domain proteins: Linking actin and plasma membrane dynamics. Curr Opin Cell Biol 23:14–21
Sorre B et al (2012) Nature of curvature coupling of amphiphysin with membranes depends on its bound density. Proc Natl Acad Sci USA 109:173–178
Peleg B, Disanza A, Scita G, Gov N (2011) Propagating cell-membrane waves driven by curved activators of actin polymerization. PLoS One 6
Scherfeld D, Kahya N, Schwille P (2003) Lipid dynamics and domain formation in model membranes composed of ternary mixtures of unsaturated and satur1ated phosphatidylcholines and cholesterol. Biophys J 85:3758–3768
Crane JM, Tamm LK (2004) Role of cholesterol in the formation and nature of lipid rafts in planar and spherical model membranes. Biophys J 86:2965–2979
Dinic J, Ashrafzadeh P, Parmryd I (2013) Actin filaments attachment at the plasma membrane in live cells cause the formation of ordered lipid domains. Biochim Biophys Acta - Biomembr 1828:1102–1111
Chichili GR, Rodgers W (2009) Cytoskeleton-membrane interactions in membrane raft structure. Cell Mol Life Sci 66:2319–2328
Stillwell W (2013) An introduction to biological membranes : from bilayers to rafts. Elsevier/Academic Press, New York
Fahey, P. F. et al. Lateral diffusion in planar lipid bilayers. Science (80-. ). 195, 305–306 (1977).
Alberts, B. et al. The Lipid Bilayer. in Molecular Biology of the Cell (eds. Anderson, M. & Granum, S.) (Garland Science, 2002).
Jan Akhunzada, M. et al. Interplay between lipid lateral diffusion, dye concentration and membrane permeability unveiled by a combined spectroscopic and computational study of a model lipid bilayer. Sci. Rep. 9, 1–12 (2019).
Rose M, Hirmiz N, Moran-Mirabal JM, Fradin C (2015) Lipid diffusion in supported lipid bilayers: A comparison between line-scanning fluorescence correlation spectroscopy and single-particle tracking. Membranes (Basel) 5:702–721
Pinkwart K et al (2019) Nanoscale dynamics of cholesterol in the cell membrane. J Biol Chem 294:12599–12609
Ritchie K, Iino R, Fujiwara T, Murase K, Kusumi A (2003) The fence and picket structure of the plasma membrane of live cells as revealed by single molecule techniques. Mol Membr Biol 20:13–18
Suzuki K, Ritchie K, Kajikawa E, Fujiwara T, Kusumi A (2005) Rapid hop diffusion of a G-protein-coupled receptor in the plasma membrane as revealed by single-molecule techniques. Biophys J 88:3659–3680
Svitkina TM (2020) Actin cell cortex: structure and molecular organization. Trends Cell Biol 30:556–565
Phillips R (2013) Kondev, Jane, Theriot, Julie, Gracia. G. H, Physical Biology of Cell
Fritzsche M, Erlenkämper C, Moeendarbary E, Charras G, Kruse K (2016) Actin kinetics shapes cortical network structure and mechanics. Sci Adv 2:1–13
Chugh P et al (2017) Actin cortex architecture regulates cell surface tension. Nat Cell Biol 19:689–697
Shutova, M., Yang, C., Vasiliev, J. M. & Svitkina, T. Functions of nonmuscle myosin ii in assembly of the cellular contractile system. PLoS One 7, (2012).
Murrell M, Oakes PW, Lenz M, Gardel ML (2015) Forcing cells into shape: the mechanics of actomyosin contractility. Nat Rev Mol Cell Biol 16:486–498
Kumar R, Saha S, Sinha B (2019) Cell spread area and traction forces determine myosin-II-based cortex thickness regulation. Biochim Biophys Acta Mol Cell Res. (2019) doi:https://doi.org/10.1016/j.bbamcr.2019.07.011.
Clark AG, Dierkes K, Paluch EK (2013) Monitoring actin cortex thickness in live cells. Biophys J 105:570–580
Ramanathan SP et al (2015) Cdk1-dependent mitotic enrichment of cortical myosin II promotes cell rounding against confinement. Nat Cell Biol 17:148–159
Blanchoin L, Boujemaa-Paterski R, Sykes C, Plastino J (2014) Actin dynamics, architecture, and mechanics in cell motility. Physiol Rev 94:235–263
Bretscher A, Edwards K, Fehon RG (2002) ERM proteins and merlin: integrators at the cell cortex. Nat Rev Mol Cell Biol 3:586–599
Louvet-Vallée S (2000) ERM proteins: From cellular architecture to cell signaling. Biol Cell 92:305–316
Michie, K. A., Bermeister, A., Robertson, N. O., Goodchild, S. C. & Curmi, P. M. G. Two Sides of the Coin: Ezrin/Radixin/Moesin and Merlin Control Membrane Structure and Contact Inhibition. Int. J. Mol. Sci. 20, (2019).
Clucas J, Valderrama F, Bretscher A (2014) ERM proteins in cancer progression. J Cell Sci 127:267–275
Fehon RG, McClatchey AI, Bretscher A (2010) Organizing the cell cortex: the role of ERM proteins. Nat Rev Mol Cell Biol 11:276–287
Motor Proteins. in Cell Biology 623–638 (Elsevier, 2017). doi:https://doi.org/10.1016/B978-0-323-34126-4.00036-0.
Dmitrieff S, Nédélec F (2016) Amplification of actin polymerization forces. J Cell Biol 212:763–766
Apodaca G (2002) Modulation of membrane traffic by mechanical stimuli. Am J Physiol Physiol 282:F179–F190
Masters TA, Pontes B, Viasnoff V, Li Y, Gauthier NC (2013) Plasma membrane tension orchestrates membrane trafficking, cytoskeletal remodeling, and biochemical signaling during phagocytosis. Proc Natl Acad Sci U S A 110:11875–11880
Raucher D, Sheetz MP (2000) Cell Spreading and Lamellipodial Extension Rate Is Regulated by Membrane Tension. J Cell Biol 148:127–136
Diz-Muñoz A, Fletcher DA, Weiner OD (2013) Use the force: Membrane tension as an organizer of cell shape and motility. Trends Cell Biol 23:47–53
Saleem M et al (2015) A balance between membrane elasticity and polymerization energy sets the shape of spherical clathrin coats. Nat. Commun. 6, (2015)
Manneville JB et al (2008) COPI coat assembly occurs on liquid-disordered domains and the associated membrane deformations are limited by membrane tension. Proc Natl Acad Sci USA 105:16946–16951
Goetz R, Lipowsky R (1998) Computer simulations of bilayer membranes: Self-assembly and interfacial tension. J Chem Phys 108:7397–7409
Schmid F (2013) Fluctuations in lipid bilayers: Are they understood? Biophys Rev Lett 8:1–20
Sens P, Plastino J (2015) Membrane tension and cytoskeleton organization in cell motility. J Phys Condens Matter 27:273103
Tinevez J-Y et al (2009) Role of cortical tension in bleb growth. Proc Natl Acad Sci USA 106:18581–18586
Pontes B, Monzo P, Gauthier NC (2017) Membrane tension: a challenging but universal physical parameter in cell biology. Semin Cell Dev Biol 71:30–41
Kozlov MM, Chernomordik LV (2015) Membrane tension and membrane fusion. Curr Opin Struct Biol 33:61–67
Helfrich W (1973) Elastic properties of lipid bilayers elastic properties of lipid bilayers: theory and possible experiments. Z Naturforsch 28:3–7
Rodríguez-García R et al (2015) Direct cytoskeleton forces cause membrane softening in red blood cells. Biophys J 108:2794–2806
Peukes J, Betz T (2014) Direct measurement of the cortical tension during the growth of membrane blebs. Biophys J 107:1810–1820
Biswas A, Alex A, Sinha B (2017) Mapping cell membrane fluctuations reveals their active regulation and transient heterogeneities. Biophys J 113:1768–1781
Gárate F, Pertusa M, Arana Y, Bernal R (2018) Non-invasive neurite mechanics in differentiated PC12 cells. Front Cell Neurosci 12:194
Fournier JB, Ajdari A, Peliti L (2001) Effective-area elasticity and tension of micromanipulated membranes. Phys Rev Lett 86:4970–4973
Betz T, Sykes C (2012) Time resolved membrane fluctuation spectroscopy. Soft Matter 8:5317
Shiba H, Noguchi H, Fournier JB (2016) Monte Carlo study of the frame, fluctuation and internal tensions of fluctuating membranes with fixed area. Soft Matter 12:2373–2380
David F, Leibler S (1991) Vanishing tension of fluctuating membranes. J Phys II(1):959–976
Alert R, Casademunt J, Brugués J, Sens P (2015) Model for probing membrane-cortex adhesion by micropipette aspiration and fluctuation spectroscopy. Biophys J 108:1878–1886
Hetmanski JHR et al (2019) Membrane tension orchestrates rear retraction in matrix-directed cell migration. Dev Cell 51:460-475.e10
Shi Z, Graber ZT, Baumgart T, Stone HA, Cohen AE (2018) Cell membranes resist flow. Cell 175:1769-1779.e13
Lieber AD, Schweitzer Y, Kozlov MM, Keren K (2015) Front-to-rear membrane tension gradient in rapidly moving cells. Biophys J 108:1599–1603
Batchelder EL et al (2011) Membrane tension regulates motility by controlling lamellipodium organization. PNAS 108:11429–11434
Watanabe N, Mitchison TJ (2002) Single-molecule speckle analysis of actin filament turnover in lamellipodia. Science (80-) 295:1083–1086
Vallotton P, Danuser G, Bohnet S, Meister JJ, Verkhovsky AB (2005) Tracking retrograde flow in keratocytes: news from the front. Mol Biol Cell 16:1223–1231
Theriot JA, Mitchison TJ (1991) Actin microfilament dynamics in locomoting cells. Nature 352:126–131
McGrath JL, Tardy Y, Dewey CF, Meister JJ, Hartwig JH (1998) Simultaneous measurements of actin filament turnover, filament fraction, and monomer diffusion in endothelial cells. Biophys J 75:2070–2078
Carlier MF et al (1997) Actin depolymerizing factor (ADF/cofilin) enhances the rate of filament turnover: implication in actin-based motility. J Cell Biol 136:1307–1322
Carlsson AE (2010) Actin dynamics: From nanoscale to microscale. Ann Rev Biophys 39:91–110
Lieber AD, Yehudai-Resheff S, Barnhart EL, Theriot JA, Keren K (2013) Membrane tension in rapidly moving cells is determined by cytoskeletal forces. Curr Biol 23:1409–1417
Schliwa M (1982) Action of cytochalasin d on cytoskeletal networks. J Cell Biol 92:79–91
Sheetz MP (2001) Cell control by membrane-cytoskeleton adhesion. Nat Rev Mol Cell Biol 2:392–396
Betz T, Lenz M, Joanny J-F, Sykes CC (2009) ATP-dependent mechanics of red blood cells. PNAS 106:15320–15325
Raucher D, Sheetz MP (1999) Membrane expansion increases endocytosis rate during mitosis. J Cell Biol 144:497–506
Faris MDEA et al (2009) Membrane tension lowering induced by protein activity. Phys Rev Lett 102:038102
Girard P, Prost J, Bassereau P (2005) Passive or active fluctuations in membranes containing proteins. Phys Rev Lett 94:088102
Charras GT, Coughlin M, Mitchison TJ, Mahadevan L (2008) Life and times of a cellular bleb. Biophys J 94:1836–1853
Kubota HY (1981) Creeping locomotion of the endodermal cells dissociated from gastrulae of the Japanese newt, Cynops pyrrhogaster. Exp Cell Res 133:137–148
Keller HU (2000) Redundancy of lamellipodia in locomoting Walker carcinosarcoma cells. Cell Motil Cytoskeleton 46:247–256
Yoshida K, Soldati T (2006) Dissection of amoeboid movement into two mechanically distinct modes. J Cell Sci 119:3833–3844
Charras G, Paluch E (2008) Blebs lead the way: how to migrate without lamellipodia. Nat Perspect 9:730–736
Turlier H et al (2016) Equilibrium physics breakdown reveals the active nature of red blood cell flickering. Nat Phys 12:513–520
Bretou M et al (2014) Cdc42 controls the dilation of the exocytotic fusion pore by regulating membrane tension. Mol Biol Cell 25:3195–3209
Petrie RJ, Koo H, Yamada KM (2014) Generation of compartmentalized pressure by a nuclear piston governs cell motility in a 3D matrix. Science (80-) 345:1062–1065
Tabdanov E et al (2020) Engineering T cells to enhance 3D migration through structurally and mechanically complex tumor microenvironments. doi:https://doi.org/10.1101/2020.04.21.051615
Diz-Muñoz A et al (2010) Control of directed cell migration in vivo by membrane-to-cortex attachment. PLoS Biol 8
Liu Y et al (2012) Constitutively active ezrin increases membrane tension, slows migration, and impedes endothelial transmigration of lymphocytes in vivo in mice. Blood 119:445–453
Rouven Brückner B, Pietuch A, Nehls S, Rother J, Janshoff A (2015) Ezrin is a major regulator of membrane tension in epithelial cells. Sci Rep 5:14700
Bergert M et al (2019) Cell surface mechanics gate stem cell differentiation. 798918. doi:https://doi.org/10.1101/798918.
Paraschiv A, Lagny TJ, Coudrier E, Bassereau P, Šarić A (2020) Influence of membrane-cortex linkers on the extrusion of membrane tubes. https://doi.org/10.1101/2020.07.28.224741.
Gérard A et al (2014) Detection of rare antigen-presenting cells through T cell-intrinsic meandering motility, mediated by Myo1g. Cell 158:492–505
Nambiar R, Mcconnell RE, Tyska MJ (2009) Control of cell membrane tension by myosin-I. PNAS 106:11972–11977
Thottacherry JJ et al (2018) Mechanochemical feedback control of dynamin independent endocytosis modulates membrane tension in adherent cells. Nat Commun 9:4217
Mercier V et al (2020) Endosomal membrane tension regulates ESCRT-III-dependent intra-lumenal vesicle formation. Nat Cell Biol 22:947–959
Acknowledgements
BS acknowledges support from Wellcome Trust/DBT India Alliance fellowship (Grant Number IA/I/13/1/500885), SERB (Grant Number SERV_CRG_2458), and CEFIPRA (Grant Number 6303-1). The authors are grateful to IISER Kolkata and UGC for providing scholarship to AB and RK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Biswas, A., Kumar, R. & Sinha, B. Membrane Homeostasis: The Role of Actin Cytoskeleton. J Indian Inst Sci 101, 81–95 (2021). https://doi.org/10.1007/s41745-020-00217-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41745-020-00217-x