Abstract
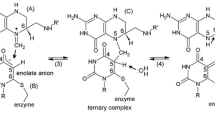
Human thymidylate synthase (hTS) is a 72 kDa homodimeric enzyme responsible for the conversion of deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP), making it the sole source of de novo dTMP in human cells. As a result, hTS is an attractive anti-cancer therapeutic target. Additionally, hTS is known to possess a number of interesting biophysical features, including adoption of active and inactive conformations, positively cooperative substrate binding, half-the-sites activity, and interacting with its own mRNA. The physical mechanisms underlying these properties, and how they may be leveraged to guide therapeutic development, are yet to be fully explored. Here, as a preface to detailed NMR characterization, we present backbone amide and ILVM methyl resonance assignments for hTS in apo and dUMP bound forms. In addition, we present backbone amide resonance assignments for hTS bound to a substrate analog and the native cofactor.



Similar content being viewed by others
Availability of data and material
The chemical shift assignments are deposited in the Biological Magnetic Resonance Data Bank under accession numbers 50520, 50521, and 50519 for apo, dUMP, and diligand bound states. The NMRViewJ software is available at https://nmrfx.org.
Code availability
Files needed for MAGIC analysis of NOESY data are available at https://github.com/NMRsoftware/MAGIC.
References
Bonin JP et al (2019) Positive cooperativity in substrate binding by human thymidylate. Synthase Biophys J 117:1074–1084. https://doi.org/10.1016/j.bpj.2019.08.015
Chen D, Jansson A, Sim D, Larsson A, Nordlund P (2017) Structural analyses of human thymidylate synthase reveal a site that may control conformational switching between active and inactive states. J Biol Chem 292:13449–13458. https://doi.org/10.1074/jbc.M117.787267
Delaglio F, Grzesiek S, Vuister GW, Zhu G, Pfeifer J, Bax A (1995) NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J Biomol NMR 6:277–293. https://doi.org/10.1007/BF00197809
Heinig M, Frishman D (2004) STRIDE: a web server for secondary structure assignment from known atomic coordinates of proteins. Nucleic Acids Res 32:W500–W502. https://doi.org/10.1093/nar/gkh429
Johnson BA (2004) Using NMRView to visualize and analyze the NMR spectra of macromolecules. Methods Mol Biol 278:313–352. https://doi.org/10.1385/1-59259-809-9:313
Monneau YR et al (2017) Automatic methyl assignment in large proteins by the MAGIC algorithm. J Biomol NMR 69:215–227. https://doi.org/10.1007/s10858-017-0149-y
Rossi P, Xia Y, Khanra N, Veglia G, Kalodimos CG (2016) (15)N and (13)C- SOFAST-HMQC editing enhances 3D-NOESY sensitivity in highly deuterated, selectively [(1)H,(13)C]-labeled proteins. J Biomol NMR 66:259–271. https://doi.org/10.1007/s10858-016-0074-5
Salzmann M, Pervushin K, Wider G, Senn H, Wuthrich K (1998) TROSY in triple-resonance experiments: new perspectives for sequential NMR assignment of large proteins. P Natl Acad Sci USA 95:13585–13590. https://doi.org/10.1073/pnas.95.23.13585
Salzmann M, Wider G, Pervushin K, Senn H, Wuthrich K (1999) TROSY-type triple-resonance experiments for sequential NMR assignments of large proteins. J Am Chem Soc 121:844–848. https://doi.org/10.1021/ja9834226
Sapienza PJ, Lee AL (2014) Backbone and ILV methyl resonance assignments of E. coli thymidylate synthase bound to cofactor and a nucleotide analogue. Biomol NMR Assign 8:195–199. https://doi.org/10.1007/s12104-013-9482-6
Sapienza PJ, Falk BT, Lee AL (2015) Bacterial thymidylate synthase binds two molecules of substrate and cofactor without cooperativity. J Am Chem Soc 137:14260–14263. https://doi.org/10.1021/jacs.5b10128
Shen Y, Delaglio F, Cornilescu G, Bax A (2009) TALOS+: a hybrid method for predicting protein backbone torsion angles from NMR chemical shifts. J Biomol NMR 44:213–223. https://doi.org/10.1007/s10858-009-9333-z
Spencer HT, Villafranca JE, Appleman JR (1997) Kinetic scheme for thymidylate synthase from Escherichia coli: determination from measurements of ligand binding, primary and secondary isotope effects, and pre-steady-state. catal Biochem 36:4212–4222. https://doi.org/10.1021/bi961794q
Stroud RM, Finer-Moore JS (2003) Conformational dynamics along an enzymatic reaction pathway: thymidylate synthase. "the movie” Biochemistry 42:239–247. https://doi.org/10.1021/bi020598i
Tai N, Schmitz JC, Liu J, Lin X, Bailly M, Chen TM, Chu E (2004) Translational autoregulation of thymidylate synthase and dihydrofolate reductase. Front Biosci 9:2521–2526. https://doi.org/10.2741/1413
Tugarinov V, Kay LE (2003) Ile, Leu, and Val methyl assignments of the 723-residue malate synthase G using a new labeling strategy and novel NMR methods. J Am Chem Soc 125:13868–13878. https://doi.org/10.1021/ja030345s
Wider G, Dreier L (2006) Measuring protein concentrations by NMR spectroscopy. J Am Chem Soc 128:2571–2576. https://doi.org/10.1021/ja055336t
Acknowledgements
We thank Dr. Amnon Kohen for providing a vector containing the hTS coding sequence. We also thank Dr. Gregory Young and Dr. Stewart Parnham of the UNC Biomolecular NMR Facility for assistance in NMR data collection.
Funding
This work was funded by National Institutes of Health (NIH) grant GM083059 to A.L.L and NIH Grant 5 T32 GM 8570-22 to J.P.B. This work was supported in part by the National Cancer Institute of the NIH under Award Number P30CA016086 Cancer Center Support Grant. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
ALL and JPB: Research was designed; JPB performed and analyzed NMR experiments. JPB. and ALL wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bonin, J.P., Lee, A.L. Backbone and ILVM methyl resonance assignments of human thymidylate synthase in apo and substrate bound forms. Biomol NMR Assign 15, 197–202 (2021). https://doi.org/10.1007/s12104-021-10006-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-021-10006-x