Abstract
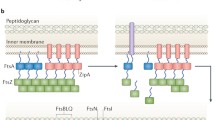
Binary fission of prokaryotic cells depends on a protein called FtsZ that self-assembles into a membrane-associated ring structure (FtsZ-ring) in the early stages of the cell division process. FtsZ is a tubulin homologue, which interacts with many additional proteins contributing to its function forming a ring at the mid-cell, essential for bacterial cell division. Whether the Z-ring is a force-generating machinery or a simple scaffold for organizing all other molecular players is poorly understood. Here, we review briefly the structure, dynamics, and interactions of FtsZ, the Z-ring and its associated proteins and weigh the evidence for and against force production by FtsZ.



Similar content being viewed by others
References
Adams DW, Errington J (2009) Bacterial cell division: assembly, maintenance and disassembly of the Z ring. Nat Rev Microbiol 7(9):642–653
Addinall SG, Bi E, Lutkenhaus J (1996) FtsZ ring formation in fts mutants. J Bacteriol 178(13):3877–3884
Nogales E et al (1998) Tubulin and FtsZ form a distinct family of GTPases. Nat Struct Biol 5(6):451–458
Lowe J, Amos LA (1998) Crystal structure of the bacterial cell-division protein FtsZ. Nature 391(6663):203–206
Buske PJ, Levin PA (2012) Extreme C terminus of bacterial cytoskeletal protein FtsZ plays fundamental role in assembly independent of modulatory proteins. J Biol Chem 287(14):10945–10957
Buske PJ, Levin PA (2013) A flexible C-terminal linker is required for proper FtsZ assembly in vitro and cytokinetic ring formation in vivo. Mol Microbiol 89(2):249–263
Oliva MA, Cordell SC, Lowe J (2004) Structural insights into FtsZ protofilament formation. Nat Struct Mol Biol 11(12):1243–1250
Krol E et al (2012) Bacillus subtilis SepF binds to the C-terminus of FtsZ. PLoS One 7(8):e43293
Coltharp C, Xiao J (2017) Beyond force generation: why is a dynamic ring of FtsZ polymers essential for bacterial cytokinesis? BioEssays 39(1):1–11
Buske PJ et al (2015) An intrinsically disordered linker plays a critical role in bacterial cell division. Semin Cell Dev Biol 37:3–10
Lariviere PJ et al (2018) FzlA, an essential regulator of FtsZ filament curvature, controls constriction rate during Caulobacter division. Mol Microbiol 107(2):180–197
Arumugam S et al (2012) Surface topology engineering of membranes for the mechanical investigation of the tubulin homologue FtsZ. Angew Chem Int Ed Engl 51(47):11858–11862
Li Y et al (2013) FtsZ protofilaments use a hinge-opening mechanism for constrictive force generation. Science 341(6144):392–395
Osawa M, Anderson DE, Erickson HP (2009) Curved FtsZ protofilaments generate bending forces on liposome membranes. EMBO J 28(22):3476–3484
Housman M et al (2016) FtsZ protofilament curvature is the opposite of tubulin rings. Biochemistry 55(29):4085–4091
Du S et al (2018) FtsZ filaments have the opposite kinetic polarity of microtubules. Proc Natl Acad Sci USA 115(42):10768–10773
Yang X et al (2017) GTPase activity-coupled treadmilling of the bacterial tubulin FtsZ organizes septal cell wall synthesis. Science 355(6326):744–747
Chen Y, Erickson HP (2005) Rapid in vitro assembly dynamics and subunit turnover of FtsZ demonstrated by fluorescence resonance energy transfer. J Biol Chem 280(23):22549–22554
Whitley KD et al (2020) FtsZ treadmilling is essential for Z-ring condensation and septal constriction initiation in bacterial cell division. bioRxiv 2020.07.01.182006
Bisson-Filho AW et al (2017) Treadmilling by FtsZ filaments drives peptidoglycan synthesis and bacterial cell division. Science 355(6326):739–743
Arumugam S, Petrasek Z, Schwille P (2014) MinCDE exploits the dynamic nature of FtsZ filaments for its spatial regulation. Proc Natl Acad Sci USA 111(13):E1192–E1200
Marmont LS, Bernhardt TG (2020) A conserved subcomplex within the bacterial cytokinetic ring activates cell wall synthesis by the FtsW-FtsI synthase. Proc Natl Acad Sci USA 117(38):23879–23885
Li Z et al (2007) The structure of FtsZ filaments in vivo suggests a force-generating role in cell division. EMBO J 26(22):4694–4708
Lan G, Wolgemuth CW, Sun SX (2007) Z-ring force and cell shape during division in rod-like bacteria. Proc Natl Acad Sci USA 104(41):16110–16115
Lan G et al (2009) Condensation of FtsZ filaments can drive bacterial cell division. Proc Natl Acad Sci USA 106(1):121–126
Ghosh B, Sain A (2008) Origin of contractile force during cell division of bacteria. Phys Rev Lett 101(17):178101
Surovtsev IV, Morgan JJ, Lindahl PA (2008) Kinetic modeling of the assembly, dynamic steady state, and contraction of the FtsZ ring in prokaryotic cytokinesis. PLoS Comput Biol 4(7):e1000102
Nguyen LT, Oikonomou CM, Jensen GJ (2019) Simulations of proposed mechanisms of FtsZ-driven cell constriction. bioRxiv 737189
Arumugam S, Chwastek G, Schwille P (2011) Protein-membrane interactions: the virtue of minimal systems in systems biology. Wiley Interdiscip Rev Syst Biol Med 3(3):269–280
Osawa M, Erickson HP (2013) Liposome division by a simple bacterial division machinery. Proc Natl Acad Sci USA 110(27):11000–11004
Osawa M, Anderson DE, Erickson HP (2008) Reconstitution of contractile FtsZ rings in liposomes. Science 320(5877):792–794
Godino E et al (2020) Cell-free biogenesis of bacterial division proto-rings that can constrict liposomes. bioRxiv 2020.03.29.009639
Ma X, Ehrhardt DW, Margolin W (1996) Colocalization of cell division proteins FtsZ and FtsA to cytoskeletal structures in living Escherichia coli cells by using green fluorescent protein. Proc Natl Acad Sci USA 93(23):12998–13003
Ma X et al (1997) Interactions between heterologous FtsA and FtsZ proteins at the FtsZ ring. J Bacteriol 179(21):6788–6797
Geissler B, Shiomi D, Margolin W (2007) The ftsA* gain-of-function allele of Escherichia coli and its effects on the stability and dynamics of the Z ring. Microbiology (Reading) 153(Pt 3):814–825
Chen Y et al (2017) ZipA and FtsA* stabilize FtsZ-GDP miniring structures. Sci Rep 7(1):3650
Arjes HA et al (2015) Mutations in the bacterial cell division protein FtsZ highlight the role of GTP binding and longitudinal subunit interactions in assembly and function. BMC Microbiol 15:209
Ramirez-Diaz DA et al (2018) Treadmilling analysis reveals new insights into dynamic FtsZ ring architecture. PLoS Biol 16(5):e2004845
Pichoff S, Du S, Lutkenhaus J (2018) Disruption of divisome assembly rescued by FtsN-FtsA interaction in Escherichia coli. Proc Natl Acad Sci USA 115(29):E6855–E6862
Pichoff S, Lutkenhaus J (2002) Unique and overlapping roles for ZipA and FtsA in septal ring assembly in Escherichia coli. EMBO J 21(4):685–693
Pichoff S, Lutkenhaus J (2007) Identification of a region of FtsA required for interaction with FtsZ. Mol Microbiol 64(4):1129–1138
Pichoff S et al (2012) FtsA mutants impaired for self-interaction bypass ZipA suggesting a model in which FtsA’s self-interaction competes with its ability to recruit downstream division proteins. Mol Microbiol 83(1):151–167
Shiomi D, Margolin W (2007) Dimerization or oligomerization of the actin-like FtsA protein enhances the integrity of the cytokinetic Z ring. Mol Microbiol 66(6):1396–1415
Shiomi D, Margolin W (2008) Compensation for the loss of the conserved membrane targeting sequence of FtsA provides new insights into its function. Mol Microbiol 67(3):558–569
Goley ED et al (2010) Imaging-based identification of a critical regulator of FtsZ protofilament curvature in Caulobacter. Mol Cell 39(6):975–987
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yadu, N., Namboothiri, A. & Arumugam, S. FtsZ: The Force Awakens. J Indian Inst Sci 101, 31–38 (2021). https://doi.org/10.1007/s41745-020-00215-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41745-020-00215-z