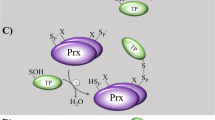
Abstract
The malaria-causing parasite Plasmodium falciparum is a severe threat to human health across the globe. This parasite alone causes the highest morbidity and mortality than any other species of Plasmodium. The parasites dynamically multiply in the erythrocytes of the vertebrate hosts, a large number of reactive oxygen species that damage biological macromolecules are produced in the cell during parasite growth. To relieve this intense oxidative stress, the parasite employs an NADPH-dependent thioredoxin and glutathione system that acts as an antioxidant and maintains redox status in the parasite. The mutual interaction of both redox proteins is involved in various biological functions and the survival of the erythrocytic stage of the parasite. Since the Plasmodium species is deficient in catalase and classical glutathione peroxidase, so their redox balance relies on a complex set of five peroxiredoxins, differentially positioned in the cytosol, mitochondria, apicoplast, and nucleus with partly overlapping substrate preferences. Moreover, Plasmodium falciparum possesses a set of members belonging to the thioredoxin superfamily, such as three thioredoxins, two thioredoxin-like proteins, one dithiol, three monocysteine glutaredoxins, and one redox-active plasmoredoxin with largely redundant functions. This review paper aims to discuss and encapsulate the biological function and current knowledge of the functional redox network of Plasmodium falciparum.



Similar content being viewed by others
References
Andricopulo AD, Akoachere MB, Krogh R, Nickel C, McLeish MJ, Kenyon GL, Arscott D, Williams CH, Davioud-Charvet E, Becker K (2006) Specific inhibitors of Plasmodium falciparum thioredoxin reductase as potential antimalarial agents. Bioorganic Med Chem Lett 16:2283–2292. https://doi.org/10.1016/j.bmcl.2006.01.027
Atamna H, Ginsburg H (1993) Origin of reactive oxygen species in erythrocytes infected with Plasmodium falciparum. Mol Biochem Parasitol 61:231–241. https://doi.org/10.1016/0166-6851(93)90069-A
Atkinson HJ, Babbitt PC (2009) An atlas of the thioredoxin fold class reveals the complexity of function-enabling adaptations. PLoS Comput Biol 5:e1000541. https://doi.org/10.1371/journal.pcbi.1000541
Barrand MA, Winterberg M, Ng F, Nguyen M, Kirk K, Hladky SB (2012) Glutathione export from human erythrocytes and Plasmodium falciparum malaria parasites. Biochem J 448:389–400. https://doi.org/10.1042/BJ20121050
Becker K, Koncarevic SHN (2005) Oxidative stress and antioxidant defense in malarial parasites. In: Sherman I (ed) Molecular approaches to malaria. ASM Press, Washington, pp 365–383
Becker K, Kanzok SM, Iozef R, Fischer M, Schirmer RH, Rahlfs S (2003) Plasmoredoxin, a novel redox-active protein unique for malarial parasites. Eur J Biochem 270:1057–1064. https://doi.org/10.1046/j.1432-1033.2003.03495.x
Becker K, Tilley L, Vennerstrom JL, Roberts D, Rogerson S, Ginsburg H (2004) Oxidative stress in malaria parasite-infected erythrocytes: host-parasite interactions. Int J Parasitol 34:163–189. https://doi.org/10.1016/j.ijpara.2003.09.011
Boucher IW, McMillan PJ, Gabrielsen M, Akerman SE, Brannigan JA, Schnick C, Brzozowski AM, Wilkinson AJ, Müller S (2006) Structural and biochemical characterization of a mitochondrial peroxiredoxin from Plasmodium falciparum. Mol Microbiol 61:948–959. https://doi.org/10.1111/j.1365-2958.2006.05303.x
Boumis G, Giardina G, Angelucci F, Bellelli A, Brunori M, Dimastrogiovanni D, Saccoccia F, Miele AE (2012) Crystal structure of Plasmodium falciparum thioredoxin reductase, a validated drug target. Biochem Biophys Res Commun 425:806–811. https://doi.org/10.1016/j.bbrc.2012.07.156
Buchholz K, Schirmer RH, Eubel JK, Akoachere MB, Dandekar T, Becker K, Gromer S (2008) Interactions of methylene blue with human disulfide reductases and their orthologues from Plasmodium falciparum. Antimicrob Agents Chemother 52:183–191. https://doi.org/10.1128/AAC.00773-07
Buchholz K, Putrianti ED, Rahlfs S, Schirmer RH, Becker K, Matuschewski K (2010) Molecular genetics evidence for the in vivo roles of the two major NADPH-dependent disulfide reductases in the malaria parasite. J Biol Chem 285:37388–37395. https://doi.org/10.1074/jbc.M110.123323
Chaudhari R, Sharma S, Patankar S (2017) Glutathione and thioredoxin systems of the malaria parasite Plasmodium falciparum: partners in crime? Biochem Biophys Res Commun 488:95–100. https://doi.org/10.1016/j.bbrc.2017.05.015
Davioud-Charvet E, McLeish MJ, Veine DM, Giegel D, Arscott LD, Andricopulo AD, Becker K, Müller S, Schirmer RH, Williams CH, Kenyon GL (2003) Mechanism-based inactivation of Thioredoxin Reductase from Plasmodium falciparum by Mannich bases. Implication for Cytotoxicity Biochemistry 42:13319–13330. https://doi.org/10.1021/bi0353629
De Koning-ward TF, Gilson PR, Boddey JA, Rug M, Smith BJ, Papenfuss AT, Sanders PR, Lundie RJ, Maier AG, Cowman AF, Crabb BS (2009) A novel protein export machine in malaria parasites. Nature 459:945–949. https://doi.org/10.1038/nature08104.A
Deponte M, Becker K, Rahlfs S (2005) Plasmodium falciparum glutaredoxin-like proteins. Biol Chem 386:33–40. https://doi.org/10.1515/BC.2005.005
Deponte M, Rahlfs S, Becker K (2007) Peroxiredoxin systems of protozoal parasites. Subcell Biochem 44:219–229. https://doi.org/10.1007/978-1-4020-6051-9_10
Färber PM, Becker K, Müller S, Schirmer RH, Franklin RM (1996) Molecular cloning and characterization of a putative glutathione reductase gene, the PfGR2 gene, from Plasmodium falciparum. Eur J Biochem 239:655–661. https://doi.org/10.1111/j.1432-1033.1996.0655u.x
Färber PM, Arscott LD, Williams CH, Becker K, Schirmer RH (1998) Recombinant Plasmodium falciparum glutathione reductase is inhibited by the antimalarial dye methylene blue. FEBS Lett 422:311–314. https://doi.org/10.1016/S0014-5793(98)00031-3
Fritz-Wolf K, Becker A, Rahlfs S, Harwaldt P, Schirmer RH, Kabsch W, Becker K (2003) X-ray structure of glutathione S-transferase from the malarial parasite Plasmodium falciparum. Proc Natl Acad Sci U S A 100:13821–13826. https://doi.org/10.1073/pnas.2333763100
Fritz-Wolf K, Jortzik E, Stumpf M, Preuss J, Iozef R, Rahlfs S, Becker K (2013) Crystal structure of the Plasmodium falciparum thioredoxin reductase-thioredoxin complex. J Mol Biol 425:3446–3460. https://doi.org/10.1016/j.jmb.2013.06.037
Gilberger TW, Schirmer RH, Walter RD, Müller S (2000) Deletion of the parasite-specific insertions and mutation of the catalytic triad in glutathione reductase from chloroquine-sensitive Plasmodium falciparum 3D7. Mol Biochem Parasitol 107:169–179. https://doi.org/10.1016/S0166-6851(00)00188-2
Guttmann P. EP (1891) No title. Ueber die wirkung des methylenblau bei malaria Berlin Klin Wochenschr 28:953–956
Jang HH, Lee KO, Chi YH, Jung BG, Park SK, Park JH, Lee JR, Lee SS, Moon JC, Yun JW, Choi YO (2004) Two enzymes in one: two yeast peroxiredoxins display oxidative stress-dependent switching from a peroxidase to a molecular chaperone function. Cell 117:625–635. https://doi.org/10.1016/j.cell.2004.05.002
Jortzik E, Becker K (2012) Thioredoxin and glutathione systems in Plasmodium falciparum. Int J Med Microbiol 302:187–94
Jortzik E, Fritz-Wolf K, Sturm N, Hipp M, Rahlfs S, Becker K (2010) Redox regulation of plasmodium falciparum ornithine δ-aminotransferase. J Mol Biol 402:445–459. https://doi.org/10.1016/j.jmb.2010.07.039
Kanzok SM, Schirmer RH, Türbachova I, Iozef R, Becker K (2000) The thioredoxin system of the malaria parasite Plasmodium falciparum. J Biol Chem 275:40180–40186. https://doi.org/10.1074/jbc.M007633200
Kasozi D, Mohring F, Rahlfs S, Meyer AJ, Becker K (2013) Real-time imaging of the intracellular glutathione redox potential in the malaria parasite Plasmodium falciparum. PLoS Pathog 9:1–18. https://doi.org/10.1371/journal.ppat.1003782
Kawazu SI, Tsuji N, Hatabu T, Kawai S, Matsumoto Y, Kano S (2000) Molecular cloning and characterization of a peroxiredoxin from the human malaria parasite Plasmodium falciparum. Mol Biochem Parasitol 109:165–169. https://doi.org/10.1016/S0166-6851(00)00243-7
Kawazu SI, Ikenoue N, Takemae H, Komaki-Yasuda K, Kano S (2005) Roles of 1-Cys peroxiredoxin in haem detoxification in the human malaria parasite Plasmodium falciparum. FEBS J 272:1784–1791. https://doi.org/10.1111/j.1742-4658.2005.04611.x
Kawazu S, Takemae H, Komaki-Yasuda K, Kano S (2010) Target proteins of the cytosolic thioredoxin in Plasmodium falciparum. Parasitol Int 59:298–302. https://doi.org/10.1016/j.parint.2010.03.005
Kehr S, Sturm N, Rahlfs S, Przyborski JM, Becker K (2010) Compartmentation of redox metabolism in malaria parasites. PLoS Pathog 6. https://doi.org/10.1371/journal.ppat.1001242
Kehr S, Jortzik E, Delahunty C, Yates JR III, Rahlfs S, Becker K (2011) Protein S-glutathionylation in malaria parasites. Antioxidants Redox Signal 15:2855–2865. https://doi.org/10.1089/ars.2011.4029
Klomsiri C, Karplus PA, Poole LB (2011) Cysteine-based redox switches in enzymes. Antioxidants Redox Signal 14:1065–1077. https://doi.org/10.1089/ars.2010.3376
Kocaoğlu E, Talaz O, Çavdar H, Şentürk M, Supuran CT, Ekinci D (2019) Determination of the inhibitory effects of N-methylpyrrole derivatives on glutathione reductase enzyme. J Enzyme Inhib Med Chem 34:51–54. https://doi.org/10.1080/14756366.2018.1520228
Komaki-Yasuda K, Kawazu SI, Kano S (2003) Disruption of the Plasmodium falciparum 2-Cys peroxiredoxin gene renders parasites hypersensitive to reactive oxygen and nitrogen species. FEBS Lett 547:140–144. https://doi.org/10.1016/S0014-5793(03)00694-X
Koncarevic S, Rohrbach P, Deponte M, Krohne G, Prieto JH, Yates J, Rahlfs S, Becker K (2009) The malarial parasite Plasmodium falciparum imports the human protein peroxiredoxin 2 for peroxide detoxification. Proc Natl Acad Sci U S A 106:13323–13328. https://doi.org/10.1073/pnas.0905387106
Kooistra RL, David R, Ruiz AC, Powers SW, Haselton KJ, Kiernan K, Blagborough AM, Solamen L, Olsen KW, Putonti C, Kanzok SM (2018) Characterization of a protozoan Phosducin-like protein-3 (PhLP-3) reveals conserved redox activity. PLoS One 13:1–21. https://doi.org/10.1371/journal.pone.0209699
Krnajski Z, Walter RD, Müller S (2001) Isolation and functional analysis of two thioredoxin peroxidases (peroxiredoxins) from Plasmodium falciparum. Mol Biochem Parasitol 113:303–308. https://doi.org/10.1016/S0166-6851(01)00219-5
Lee S, Kim SM, Lee RT (2013) Thioredoxin and thioredoxin target proteins: from molecular mechanisms to functional significance. Antioxidants Redox Signal 18:1165–1207. https://doi.org/10.1089/ars.2011.4322
Lüersen K, Walter RD, Müller S (1999) The putative γ-glutamylcysteine synthetase from Plasmodium falciparum contains large insertions and a variable tandem repeat. Mol Biochem Parasitol 98:131–142. https://doi.org/10.1016/S0166-6851(98)00161-3
Lüersen K, Walter RD, Müller S (2000) Plasmodium falciparum-infected red blood cells depend on a functional glutathione de novo synthesis attributable to an enhanced loss of glutathione. Biochem J 346:545–552. https://doi.org/10.1042/0264-6021:3460545
Maroziene A, Lesanavičius M, Davioud-Charvet E, Aliverti A, Grellier P, Šarlauskas J, Čėnas N (2019) Antiplasmodial activity of nitroaromatic compounds: correlation with their reduction potential and inhibitory action on plasmodium falciparum glutathione reductase. Molecules. 24. https://doi.org/10.3390/molecules24244509
Martin JL (1995) Thioredoxin -a fold for all reasons. Structure 3:245–250. https://doi.org/10.1016/S0969-2126(01)00154-X
McMillan PJ, Stimmler LM, Foth BJ, McFadden GI, Müller S (2005) The human malaria parasite Plasmodium falciparum possesses two distinct dihydrolipoamide dehydrogenases. Mol Microbiol 55:27–38. https://doi.org/10.1111/j.1365-2958.2004.04398.x
Meierjohann S, Walter RD, Müller S (2002) Glutathione synthetase from Plasmodium falciparum. Biochem J 363:833–838. https://doi.org/10.1042/0264-6021:3630833
Miller LH, Baruch DI, M K, D OK (2002) The pathogenic basis of malaria. Nature 415:673–679
Mohring F, Pretzel J, Jortzik E, Becker K (2014) The redox Systems of Plasmodium falciparum and Plasmodium vivax: comparison, in silico analyses and inhibitor studies. Curr Med Chem 21:1728–1756
Mol IBJ, Embo IBJ, Topics C, Immunol M (1991) Interleukin Ibeta and thioredoxin are secreted through a novel pathway of secretion 1:
Molina-Cruz A, DeJong RJ, Charles B, Gupta L, Kumar S, Jaramillo-Gutierrez G, Barillas-Mury C (2008) Reactive oxygen species modulate Anopheles gambiae immunity against bacteria and Plasmodium. J Biol Chem 283:3217–3223. https://doi.org/10.1074/jbc.M705873200
Mueller S, Riedel HD, Stremmel W (1997) Direct evidence for catalase as the predominant H2O2-removing enzyme in human erythrocytes. Blood 90:4973–4978. https://doi.org/10.1182/blood.v90.12.4973.4973_4973_4978
Müller S (2004) Redox and antioxidant systems of the malaria parasite Plasmodium falciparum. Mol Microbiol 53:1291–1305. https://doi.org/10.1111/j.1365-2958.2004.04257.x
Müller S, Liebau E, Walter RD, Krauth-Siegel RL (2003) Thiol-based redox metabolism of protozoan parasites. Trends Parasitol 19:320–328. https://doi.org/10.1016/S1471-4922(03)00141-7
Munte CE, Becker K, Schirmer RH, Kalbitzer HR (2005) 1H, 13C, and 15N sequence-specific resonance assignment and secondary structure of Plasmodium falciparum thioredoxin [11]. J Biomol NMR 32:340. https://doi.org/10.1007/s10858-005-0458-4
Nickel C, Trujillo M, Rahlfs S, Deponte M, Radi R, Becker K (2005) Plasmodium falciparum 2-Cys peroxiredoxin reacts with plasmoredoxin and peroxynitrite. Biol Chem 386:1129–1136. https://doi.org/10.1515/BC.2005.129
Nickel C, Rahlfs S, Deponte M, Koncarevic S, Becker K (2006) Thioredoxin networks in the malarial parasite Plasmodium falciparum. Antioxid Redox Signal 7–8:1227–1239
Olszewski KL, Mather MW, Morrisey JM, Garcia BA, Vaidya AB, Rabinowitz JD, Llinás M (2010) Branched tricarboxylic acid metabolism in Plasmodium falciparum. Nature 466:774–778
Pal C, Bandyopadhyay U (2012) Redox-Active Antiparasitic Drugs 17:. https://doi.org/10.1089/ars.2011.4436
Pastrana-Mena R, Dinglasan RR, Franke-Fayard B, Vega-Rodríguez J, Fuentes-Caraballo M, Baerga-Ortiz A, Coppens I, Jacobs-Lorena M, Janse CJ, Serrano AE (2010) Glutathione reductase-null malaria parasites have normal blood stage growth but arrest during development in the mosquito. J Biol Chem 285:27045–27056. https://doi.org/10.1074/jbc.M110.122275
Patzewitz EM, Wong EH, Müller S (2012) Dissecting the role of glutathione biosynthesis in Plasmodium falciparum. Mol Microbiol 83:304–318. https://doi.org/10.1111/j.1365-2958.2011.07933.x
Putonti C, Quach B, Kooistra RL, Kanzok SM (2013) The evolution and putative function of phosducin-like proteins in the malaria parasite Plasmodium. Infect Genet Evol 13:49–55. https://doi.org/10.1016/j.meegid.2012.08.023
Rahlfs S, Fischer M, Becker K (2001) Plasmodium falciparum possesses a classical Glutaredoxin and a second, Glutaredoxin-like protein with a PICOT homology domain. J Biol Chem 276:37133–37140. https://doi.org/10.1074/jbc.M105524200
Rahlfs S, Nickel C, Deponte M, Schirmer RH, Becker K (2003) Plasmodium falciparum thioredoxins and glutaredoxins as central players in redox metabolism. Redox Rep 8:246–250. https://doi.org/10.1179/135100003225002844
Rhee SG, Woo HA (2011) Multiple functions of peroxiredoxins: peroxidases, sensors and regulators of the intracellular messenger H2O2, and protein chaperones. Antioxidants Redox Signal 15:781–794. https://doi.org/10.1089/ars.2010.3393
Richard D, Bartfai R, Volz J, Ralph SA, Muller S, Stunnenberg HG, Cowman AF (2011) A genome-wide chromatin-associated nuclear peroxiredoxin from the malaria parasite Plasmodium falciparum. J Biol Chem 286:11746–11755. https://doi.org/10.1074/jbc.M110.198499
Sarma GN, Savvides SN, Becker K, Schirmer M, Schirmer RH, Karplus PA (2003) Glutathione reductase of the malarial parasite Plasmodium falciparum: crystal structure and inhibitor development. J Mol Biol 328:893–907. https://doi.org/10.1016/S0022-2836(03)00347-4
Savvides SN, Scheiwein M, Böhme CC, Arteel GE, Karplus PA, Becker K, Schirmer RH (2002) Crystal structure of the antioxidant enzyme glutathione reductase inactivated by peroxynitrite. J Biol Chem 277:2779–2784. https://doi.org/10.1074/jbc.M108190200
Sen CK (1999) Glutathione homeostasis in response to exercise training and nutritional supplements. Mol Cell Biochem 196:31–42. https://doi.org/10.1023/A:1006910011048
Sharma A, Sharma A, Dixit S, Sharma A (2011) Structural insights into thioredoxin-2: a component of malaria parasite protein secretion machinery. Sci Rep 1:1–9. https://doi.org/10.1038/srep00179
Sienkiewicz N, Daher W, Dive D, Wrenger C, Viscogliosi E, Wintjens R, Jouin H, Capron M, Müller S, Khalife J (2004) Identification of a mitochondrial superoxide dismutase with an unusual targeting sequence in Plasmodium falciparum. Mol Biochem Parasitol 137:121–132. https://doi.org/10.1016/j.molbiopara.2004.05.005
Storm J, Perner J, Aparicio I, Patzewitz EM, Olszewski K, Llinas M, Engel PC, Muller S (2011) Plasmodium falciparum glutamate dehydrogenase a is dispensable and not a drug target during erythrocytic development. Malar J. https://doi.org/10.1186/1475-2875-10-193
Sturm N, Hu Y, Zimmermann H, Fritz-Wolf K, Wittlin S, Rahlfs S, Becker K (2009a) Compounds structurally related to ellagic acid show improved antiplasmodial activity. Antimicrob Agents Chemother 53:622–630. https://doi.org/10.1128/AAC.00544-08
Sturm N, Jortzik E, Mailu BM, Koncarevic S, Deponte M, Forchhammer K, Rahlfs S, Becker K (2009b) Identification of proteins targeted by the thioredoxin superfamily in Plasmodium falciparum PLoS Pathog 5:. https://doi.org/10.1371/journal.ppat.1000383
Tiwari S, Wadhawan M, Singh N, Rathaur S (2015) Effect of CDNB on filarial thioredoxin reductase : a proteomic and biochemical approach. J Proteome 113:435–446. https://doi.org/10.1016/j.jprot.2014.10.007
Tiwari NK, Reynolds PJ, Calderón AI (2016) Preliminary LC-MS based screening for inhibitors of Plasmodium falciparum Thioredoxin Reductase (PfTrxR) among a set of Antimalarials from the malaria box. Molecules. 21. https://doi.org/10.3390/molecules21040424
Tiwari S, Wadhawan M, Yadav S, Rathaur S (2018) Role of thioredoxin and glutathione reductase redox system in bovine filarial parasite Setaria cervi. Adv Medico veterinary Parasitol: an Indian Prospective, Panima Publishing Corporation, New Delhi, 20: 392–416.
Urig S, Becker K (2006) On the potential of thioredoxin reductase inhibitors for cancer therapy. Semin Cancer Biol 16:452–465. https://doi.org/10.1016/j.semcancer.2006.09.004
Vega-Rodríguez J, Franke-Fayard B, Dinglasan RR, Anse CJ, Pastrana-Mena R, Waters AP, Coppens I, Rodríguez-Orengo JF, Jacobs-Lorena M, Serrano AE (2009) The glutathione biosynthetic pathway of Plasmodium is essential for mosquito transmission. PLoS Pathog 5:16–18. https://doi.org/10.1371/journal.ppat.1000302
Wang W, Huang P, Jiang N, Lu H, Zhang D, Wang D, Zhang K, Wahlgren M, Chen Q (2018) A thioredoxin homologous protein of Plasmodium falciparum participates in erythrocyte invasion. Infect Immun 86:1–11. https://doi.org/10.1128/IAI.00289-18
Werner C, Stubbs MT, Krauth-Siegel RL, Klebe G (2005) The crystal structure of Plasmodium falciparum glutamate dehydrogenase, a putative target for novel antimalarial drugs. J Mol Biol 349(3):597–607
Williams CH, David Arscott L, Müller S, Lennon BW, Ludwig ML, Wang PF, Veine DM, Becker K, Schirmer RH (2000) Thioredoxin reductase: two modes of catalysis have evolved. Eur J Biochem 267:6110–6117. https://doi.org/10.1046/j.1432-1327.2000.01702.x
Wood ZA, Poole LB, Karplus PA (2003) Peroxiredoxin evolution and the regulation of hydrogen peroxide signaling. Science (80- ) 300:650–653. https://doi.org/10.1126/science.1080405
Yano K, Komaki-Yasuda K, Kobayashi T, Takemae H, Kita K, Kano S, Kawazu SI (2005) Expression of mRNAs and proteins for peroxiredoxins in Plasmodium falciparum erythrocytic stage. Parasitol Int 54:35–41. https://doi.org/10.1016/j.parint.2004.08.005
Yano K, Otsuki H, Arai M, Komaki-Yasuda K, Tsuboi T, Torii M, Kano S, Kawazu SI (2008) Disruption of the Plasmodium berghei 2-Cys peroxiredoxin TPx-1 gene hinders the sporozoite development in the vector mosquito. Mol Biochem Parasitol 159:142–145. https://doi.org/10.1016/j.molbiopara.2008.03.002
Acknowledgments
The authors thank the Department of Science and Technology and the Indian Council of Medical Research for fellowship.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
The Indian Council of Medical Research (ICMR), India provide the financial assistance.
Additional information
Section Editor: Tobili Sam-Yellowe
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tiwari, S., Sharma, N., Sharma, G.P. et al. Redox interactome in malaria parasite Plasmodium falciparum. Parasitol Res 120, 423–434 (2021). https://doi.org/10.1007/s00436-021-07051-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07051-9