Abstract
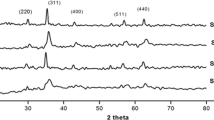
Hydrophobic cobalt ferrite nanoparticles (CoFe2O4) (HCF NPs) were synthesized with the size of 14–27 nm by a simple chemical coprecipitation method in the presence of sodium dodecyl sulfate (SDS) surfactant. Then the decoration of the surface of graphite powder with hydrophobic cobalt ferrite nanoparticles was carried out using the sonication method. The result of the sonication method revealed that hydrophobic CoFe2O4 nanoparticles were attached to the graphite powder via shared hydrophobic interaction and caused in nanoscale CoFe2O4-graphite nanocomposites (HCFG). The broadband at 550 nm from the UV-Vis. spectroscopy confirmed the formation of cobalt ferrite nanoparticles. The functional groups and metal oxide peaks of the synthesized HCFG were characterized using FTIR spectroscopy. SEM, AFM, and TEM results established the surface morphology and size of the nanocomposites. The iron, oxygen, and carbon contents and the crystalline nature of the nanocomposites were confirmed using EDAX and XRD results. The magnetic measurement value of HCFG nanocomposites was 15 emu/g. This method offers a simple, fast, and low-cost method for the synthesis of hydrophobic cobalt ferrite-graphite nanocomposites.










Similar content being viewed by others
References
Amiri, S., Shokrollahi, H.: The role of cobalt ferrite magnetic nanoparticles in medical science. Mater Sci Eng C. 33, 1–8 (2013). https://doi.org/10.1016/j.msec.2012.09.003
Srinivasan, S.Y., Paknikar, K.M., Bodas, D., Gajbhiye, V.: Applications of cobalt ferrite nanoparticles in biomedical nanotechnology. Nanomedicine. 13, 1221–1238 (2018). https://doi.org/10.2217/nnm-2017-0379
Amiri, M., Eskandari, K., Salavati-Niasari, M.: Magnetically retrievable ferrite nanoparticles in the catalysis application. Adv Colloid Interf Sci. 271, 101982 (2019). https://doi.org/10.1016/j.cis.2019.07.003
Hunyek, A., Sirisathitkul, C., Mahaphap, C., Boonyang, U., Tangwatanakul, W.: Sago starch: chelating agent in sol-gel synthesis of cobalt ferrite nanoparticles. J Aust Ceram Soc. 53, 173–176 (2017). https://doi.org/10.1007/s41779-017-0022-1
Abbasian, A.R., Lorfasaei, Z., Shayesteh, M., Afarani, M.S.: Synthesis of cobalt ferrite colloidal nanoparticle clusters by ultrasonic-assisted solvothermal process. J Aust Ceram Soc. (2020). https://doi.org/10.1007/s41779-020-00456-2
Allaedini, G., Masrinda, S., Payam, T.: Magnetic properties of cobalt ferrite synthesized by hydrothermal method. Int Nano Lett. 5, 183–186 (2015). https://doi.org/10.1007/s40089-015-0153-8
Vestal, C.R., Zhang, Z.J.: Synthesis of CoCrFeO 4 Nanoparticles using microemulsion methods and size-dependent studies of their magnetic properties, pp. 3817–3822 (2002)
Thomas, J., Thomas, N., Girgsdies, F., Beherns, M., Huang, X., Sudheesh, V.D., Sebastian, V.: Synthesis of cobalt ferrite nanoparticles by constant pH co-precipitation and their high catalytic activity in CO oxidation. New J Chem. 41, 7356–7363 (2017). https://doi.org/10.1039/c7nj00558j
Mazarío, E., Herrasti, P., Morales, M.P., Menéndez, N.: Synthesis and characterization of CoFe 2O 4 ferrite nanoparticles obtained by an electrochemical method. Nanotechnology. 23, (2012). https://doi.org/10.1088/0957-4484/23/35/355708
Goswami, P.P., Choudhury, H.A., Chakma, S., Moholkar, V.S.: Sonochemical synthesis of cobalt ferrite nanoparticles. Int J Chem Eng. 2013, (2013). https://doi.org/10.1155/2013/934234
Akhtar, S., Khan, Q., Anwar, S., Ali, G., Maqbool, M., Khan, M., Karim, S., Gao, L.: A comparative study of the toxicity of polyethylene glycol coated cobalt ferrite nanospheres and nanoparticles. Nanoscale Res Lett. 14, 386 (2019). https://doi.org/10.1186/s11671-019-3202-9
Lickmichand, M., Shaji, C.S., Valarmathi, N., Benjamin, A.S., Arun Kumar, R.K., Nayak, S., Saraswathy, R., Sumathi, S., Arunai Nambi Raj, N.: In vitro biocompatibility and hyperthermia studies on synthesized cobalt ferrite nanoparticles encapsulated with polyethylene glycol for biomedical applications. Mater Today: Proc. 15, 252 (2019)
Nam, P.H., Lu, L.T., Linh, P.H., Manh, D.H., Tam, T.T., Phuc, N.X., Phong, P.T., Lee, I.J.: Polymer-coated cobalt ferrite nanoparticles: synthesis, characterization, and toxicity for hyperthermaia applications. New J Chem. 42, 14530 (2018). https://doi.org/10.1039/C8NJ01701H
Salunkhea, A.B., Khota, V.M., Thorata, N.D., Phadatarea, M.R., Sathishb, C.I., Dhawaleb, D.S., Pawar, S.H.: Polyvinyl alcohol functionalized cobalt ferrite nanoparticles for biomedical applications. Appl Surf Sci. 264, 598 (2013). https://doi.org/10.1016/j.apsusc.2012.10.073
Peeples, B., Goornavar, V., Peeples, C., Spence, D., Parker, V., Bell, C., Biswal, D., Ramesh, G.T., Pradhan, A.K.: Structural, stability, magnetic, and toxicity studies of nanocrystalline iron oxide and cobalt ferrites for biomedical applications. J Nanopart Res. 16, 2290 (2014). https://doi.org/10.1007/s11051-014-2290-9
Laznev, K., Tzerkovsky, D., Kekalo, K., Zhavnerko, G., Agabekov, V.: Iron-cobalt ferrite nanoparticles-biocompatibility and distribution after administration to rat. IEEE Trans Magn. 49, 425 (2013). https://doi.org/10.1109/TMAG.2012.2222020
Bohara, B.A., Thorat, N.D., Yadava, H.M., Pawar, S.H.: One-step synthesis of uniform and biocompatible amine functionalized cobalt ferrite nanoparticles: a potential carrier for biomedical applications. New J Chem. 38, 2979 (2014). https://doi.org/10.1039/C4NJ00344F
Sanpo, N., Tharajak, J., Li, Y., Bernt, C.C., Wen, C., Wang, J.: Biocompatibility of transition metal-substituted cobalt ferrite nanoparticles. J Nanopart Res. 16, 2510 (2014). https://doi.org/10.1007/s11051-014-2510-3
Phong, P.T., Phuc, N.X., Nam, P.H., Chien, N.V., Dung, D.D., Linh, P.H.: Size-controlled heating ability of CoFe2O4 nanoparticles for hyperthermia applications. Phys B Condens Matter. 531, 30 (2018)
Karthikeyan, P., Vigneshwaran, S., Preethi, J., Meenakshi, S.: Diamond & related materials preparation of novel cobalt ferrite coated-porous carbon composite by simple chemical co-precipitation method and their mechanistic performance. Diam Relat Mater. 108, 107922 (2020). https://doi.org/10.1016/j.diamond.2020.107922
Vijaya, K., Shanmugapriya, S., Surendran, S., Chan, S., Kalai, R.: Journal of colloid and interface science facile hydrothermal synthesis of carbon-coated cobalt ferrite spherical nanoparticles as a potential negative electrode for flexible supercapattery. J Colloid Interface Sci. 513, 480–488 (2018). https://doi.org/10.1016/j.jcis.2017.11.054
Lizundia, E., Rincón-Iglesias, M., Lanceros-Méndez, S.: Combining cobalt ferrite and graphite with cellulose nanocrystals for magnetically active and electrically conducting mesoporous nanohybrids. Carbohydr Polym. 236, 116001 (2020). https://doi.org/10.1016/j.carbpol.2020.116001
Ivanov, A.V., Pavlova, J.A., Kalachev, I.L., Maksimova, N.V., Dzhevakov, P.B., Malakho, A.P.: ScienceDirect preparation of magnetic sorbent based on exfoliated graphite with different content of cobalt ferrite. Mater Today Proc. 5, 26010–26017 (2018). https://doi.org/10.1016/j.matpr.2018.08.021
Li, L., Xiang, C., Qian, H., Hao, B., Chen, K., Qiao, R.: Expanded graphite/cobalt ferrite/polyaniline ternary composites: fabrication, properties, and potential applications, pp. 2683–2690 (2011). https://doi.org/10.1557/jmr.2011.317
Alyami, M., Khashab, M., Costa, P.M.F.J.: Cobalt ferrite supported on reduced graphene oxide as a T 2 contrast agent for magnetic resonance imaging †, pp. 6299–6309 (2019). https://doi.org/10.1039/c8ra09476d
Wang, G., Ma, Y., Wei, Z., Qi, M.: Development of multifunctional cobalt ferrite / graphene oxide nanocomposites for magnetic resonance imaging and controlled drug delivery. Chem Eng J. 289, 150–160 (2016). https://doi.org/10.1016/j.cej.2015.12.072
Thombare, B.R., Gavhane, D.S., Lole, G.S., Bankar, P.K., Dusane, P.R., Kolhe, P.S., Khupse, N.D., Choudhary, R.J., Phase, D.M., Devan, R.S., Sonawane, K.M., More, M.A., Patil, S.I.: Cobalt ferrite decorated multiwalled carbon nanotubes as the electrode for efficient field electron emission. Phys E Low-Dimensional Syst Nanostructures. 121, 114131 (2020). https://doi.org/10.1016/j.physe.2020.114131
Möller, L., Thauer, E., Ottmann, A., Deeg, L., Ghunaim, R., Hampel, S., Klingeler, R.: CoFe2O4-filled carbon nanotubes as anode material for lithium-ion batteries. J Alloys Compd. 834, (2020). https://doi.org/10.1016/j.jallcom.2020.155018
Sayahi, H., Mohsenzadeh, F., Darabi, H.R., Aghapoor, K.: Facile and economical fabrication of magnetite/graphite nanocomposites for supercapacitor electrodes with significantly extended potential window. J Alloys Compd. 778, 633–642 (2019). https://doi.org/10.1016/j.jallcom.2018.11.186
Zheng, Y., Tian, Y., Zhang, H., Guo, Q., Luo, J.: Microwave rapid synthesis of NiO/Ni3S2@graphite nanocomposites for supercapacitor applications. Inorg Chem Commun. 110, 107596 (2019). https://doi.org/10.1016/j.inoche.2019.107596
Xu, T., Lin, N., Cai, W., Yi, Z., Zhou, J., Han, Y., Zhu, Y., Qian, Y.: Stabilizing Si/graphite composites with Cu and: in situ synthesized carbon nanotubes for high-performance Li-ion battery anodes. Inorg Chem Front. 5, 1463–1469 (2018). https://doi.org/10.1039/c8qi00173a
Kim, H., Nguyen, Q.H., Kim, I.T., Hur, J.: Scalable synthesis of high-performance molybdenum diselenide-graphite nanocomposite anodes for lithium-ion batteries. Appl Surf Sci. 481, 1196–1205 (2019). https://doi.org/10.1016/j.apsusc.2019.03.165
Mohammed, H.A., Rashid, S.A., Abu Bakar, M.H., Ahmad Anas, S.B., Mahdi, M.A., Yaacob, M.H.: Fabrication and characterizations of a novel etched-tapered single mode optical fiber ammonia sensors integrating PANI/GNF nanocomposite. Sensors Actuators B Chem. 287, 71–77 (2019). https://doi.org/10.1016/j.snb.2019.01.115
Ponvel, K.M., Lee, D.G., Woo, E.J., Ahn, I.S., Lee, C.H.: Immobilization of lipase on surface modified magnetic nanoparticles using alkyl benzenesulfonate. Korean J Chem Eng. 26, 127–130 (2009). https://doi.org/10.1007/s11814-009-0019-8
Uran, S., Alhani, A., Silva, C.: Study of ultraviolet-visible light absorbance of exfoliated graphite forms. AIP Adv. 7, (2017). https://doi.org/10.1063/1.4979607
Vinosha, P.A., Immaculate, G., Mary, N., Mahalakshmi, K., Mely, L.A., Das, S.J.: Study on cobalt ferrite nanoparticles synthesized by co-precipitation technique for photo-fenton application. Mech. Mater. Sci. Eng. MMSE Journal. Open Access www.mmse.xyz. 2412–5954 (2017). https://doi.org/10.2412/mmse.36.49.466
Loan, N.T.T., Lan, N.T.H., Hand, N.T.T., Hai, N.Q., Anh, D.T.T., Hau, V.T., Tan, L.V., Tran, T.V.: CoFe2O4 nanomarterial: effect of annealing temperature on characterization, magnetic, photocatalytic, and photo-Fenton properties. Processes. 7, 885 (2019). https://doi.org/10.3390/pr7120885
Ravindra, A.V., Chandrika, M., Rajesh, C., Kollu, P., Ju, S., Ramarao, S.D.: Simple synthesis, structural and optical properties of cobalt ferrite nanoparticles. Eur Phys J Plus. 134, 296 (2019). https://doi.org/10.1140/epjp/i2019-12690-2
Sharma, D., Khare, N.: Tailoring the optical bandgap and magnetization of cobalt ferrite thin films through controlled zinc doping. AIP Adv. 6, 085005 (2016). https://doi.org/10.1063/1.4960989
Kumar, V., Singh, V., Umrao, S., Parashar, V., Abraham, S., Singh, A.K., Nath, G., Saxena, P.S., Srivastava, A.: Facile, rapid and upscaled synthesis of green luminescent functional graphene quantum dots for bioimaging. RSC Adv. 4, 21101 (2014). https://doi.org/10.1039/C4RA01735H
Safi, R., Ghasemi, A., Shoja-Razavi, R., Tavousi, M.: The role of pH on the particle size and magnetic consequence of cobalt ferrite. J Magn Magn Mater. 396, 288–294 (2015). https://doi.org/10.1016/j.jmmm.2015.08.022
Goodarz Naseri, M., Saion, E.B., Abbastabar Ahangar, H., Shaari, A.H., Hashim, M.: Simple synthesis and characterization of cobalt ferrite nanoparticles by a thermal treatment method. J Nanomater. 2010, (2010). https://doi.org/10.1155/2010/907686
Hosseini, S.M., Sohrabnejad, S., Nabiyouni, G., Jashni, E., Van der Bruggen, B., Ahmadi, A.: Magnetic cation exchange membrane incorporated with cobalt ferrite nanoparticles for chromium ions removal via electrodialysis. J Membr Sci. 583, 292–300 (2019). https://doi.org/10.1016/j.memsci.2019.04.069
Verde, E.L., Landi, G.T., Gomes, J.A., Sousa, M.H., Bakuzis, A.F.: Magnetic hyperthermia investigation of cobalt ferrite nanoparticles: comparison between experiment, linear response theory, and dynamic hysteresis simulations. J Appl Phys. 111, 123902 (2012). https://doi.org/10.1063/1.4729271
Jiang, Y., Liu, X., Li, J., Zhou, L., Yang, X., Huang, Y.: Enhanced electrochemical oxidation of p-nitrophenol using single-walled carbon nanotubes/silver nanowires hybrids modified electrodes. J Nanosci Nanotechnol. 15, 6078 (2015). https://doi.org/10.1166/jnn.2015.10275
Arvinte, A., Mahosenaho, M., Pinteala, M., Sesay, A.-M., Virtanen, V.: Electrochemical oxidation of p-nitrophenol using graphene-modified electrodes, and a comparison to the performance of MWNT-based electrodes. Microchim Acta. 174, 337 (2011). https://doi.org/10.1007/s00604-011-0628-x
Saadati, F., Ghahramani, F., Shayani-jam, H., Piri, F., Yaftian, M.R.: Synthesis and characterization of nanostructure molecularly imprinted polyaniline/graphene oxide composite as a highly selective electrochemical sensor for detection of p -nitrophenol. J Taiwan Inst Chem Eng. 86, 231 (2018). https://doi.org/10.1016/j.jtice.2018.02.019
Vinoth, S., Sampathkumar, P., Giribabu, K., Pandikumar, A.: Ultrasonically assisted synthesis of barium stannate incorporated graphitic carbon nitride nanocomposite and its analytical performance in electrochemical sensing of 4-nitrophenol. Ultrason Sonochem. 62, 104855 (2020). https://doi.org/10.1016/j.ultsonch.2019.104855
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rathimeena, M., Ponvel, K.M. & Chavali, M. Simple and efficient synthesis of graphite nanocomposites with hydrophobic cobalt ferrite nanoparticles. J Aust Ceram Soc 57, 555–565 (2021). https://doi.org/10.1007/s41779-020-00552-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41779-020-00552-3