Abstract
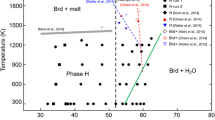
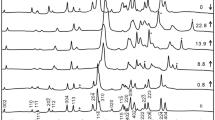
Here, we present new measurements of the phase stability and lattice compressibility of gaspéite (NiCO3) to 50 GPa. Our study is motivated by our interest in understanding high-pressure carbonate behavior. While carbonates have been extensively studied under high-pressure and -temperature conditions, they exhibit different behaviors. We have studied the high-pressure behavior of gaspéite using diamond anvil cells, Raman spectroscopy, and X-ray diffraction. Our experimental data show that gaspéite maintains the calcite structure up to 50 GPa, reverts to its zero-pressure volume on decompression with little hysteresis, and can be fit by a 3rd-order Birch–Murnaghan equation of state. We calculate a bulk modulus (K0T) of 136(4) GPa and a K′ value of 4.6(3). Additionally, we have determined the isothermal Grüneisen parameter for each of the traced Raman modes. These results contribute to growing experimental evidence that suggests some carbonates can be stable at lower mantle conditions. Ultimately, information in this dataset may facilitate predictions of mixing energetics amongst the calcite-structured carbonates, and therefore help determine the role of carbonates in the transition metal geochemistry of the deep Earth.








Similar content being viewed by others
References
Alt JC, Teagle DAH (1999) The uptake of carbon during alteration of the oceanic crust. Geochem Cosmochim Acta 63:1527–1535
Birch F (1947) Finite elastic strain of cubic crystals. Phys Rev 71:809
Born M, Huang K (1954) Dynamical theory of crystal lattices. Clarendon Press, Oxford
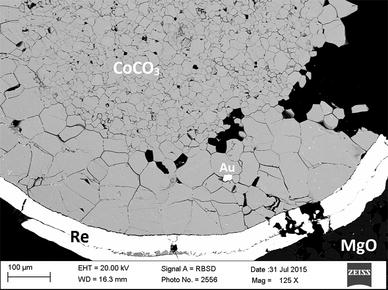
Chariton S, Cerantola V, Ismailova L, Bykova E, Bykov M, Kupenko I, McCammon C, Dubrovinsky L (2017) The high-pressure behavior of spherocobaltite (CoCO3): a single crystal Raman spectroscopy and XRD study. Phys Chem Miner. https://doi.org/10.1007/s00269-017-0902-5
Chulia-Jordan R, Santamaria-Perez D, Ortero-de-la-Roza A, Ruiz-Fuertes J, Marqueno T, Gomis O, MacLeod S, Popescu C (2020) Phase stability of natural Ni0.75Mg0.22Ca0.03CO3 gaspeite mineral at high pressure and temperature. J Phys Chem C 124(36):19781–19792
Dasgupta R, Hirschmann MM (2010) The deep carbon cycle and melting in Earth’s interior. Earth Planet Sci Lett 298:1–13
Farfan G, Wang S, Ma H, Caracas R, Mao W (2012) Bonding and structural changes in siderite at high pressure. Am Miner 97(8–9):1421–1426
Farfan GA, Boulard E, Wang S, Mao WL (2013) Bonding and electronic changes in rhodochrosite at high pressure. Am Miner 98:1817–1823
Farsang S, Facq S, Redfern SAT (2018) Raman modes of carbonate minerals as pressure and temperature gauges up to 6 GPa and 500 C. Am Miner 103:1988–1998
Fiquet G, Guyot F, Itie JP (1994) High pressure X-ray diffraction study of carbonates: MgCO3, CaMg(CO3)2, and CaCO3. Am Miner 79:15–23
Gillet P (1993) Stability of magnesite (MgCO3) at mantle pressure and temperatures conditions: a Raman spectroscopic study. Am Miner 78(11–12):1328–1331
Graf DL (1961) Crystallographic tables for the rhombohedral carbonates. Am Mineral 46(11–12):1283–1316
Holland TJB, Redfern SAT (1997) Unit cell refinement from powder diffraction data: the use of regression diagnostics. Mineral Mag 61:65–77
Isshiki M, Irifune T, Hirose K, Ono S, Ohishi Y, Watanuki T, Nishibori E, Takata M, Sakata M (2004) Stability of magnesite and its high-pressure form in the lowermost mantle. Nature 427:60–63
Klotz S, Chervin JC, Munsch P, Marchand GL (2009) Hydrostatic limits of 11 pressure transmitting media. J Phys D Appl Phys 42:075413. https://doi.org/10.1088/0022-3727/42/7/075413
Kohls DW, Rodda JL (1966) Gaspéite (Ni, Mg, Fe) (CO3), a new carbonate from the Gaspé peninsula, Quebec. Am Miner 51:677–684
Lavina B, Dera P, Down RT, Yang W, Sinogeikin S, Meng Y, Shen G, Schiferl D (2010) Structure of Siderite FeCO3 to 56 GPa and hysteresis of its spin-pairing transition. Phys Rev B 82:0641110
Mao HK, Xu J, Bell PM (1986) Calibration of the ruby pressure gauge to 800 kbar under quasi-hydrostatic conditions. J Geophys Res 91:4673–4676
Merlini M, Hanfland M, Crichton WA (2012) CaCO3-III and CaCO3-VI, high-pressure polymorphs of calcite: possible host structures for carbon in the Earth’s mantle. Earth Planet Sci Lett 333–334:265–271
Merlini M, Hanfland M, Gemmi M (2015) The MnCO3-II high-pressure polymorph of rhodochrosite. Am Miner 100:2625–2629
Momma K, Izumi F (2011) VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J Appl Crystallogr 44:1272–1276
Nickel EH, Clout JFM, Gartrell BJ (1994) Secondary nickel minerals from Widgiemooltha. Mineral Rec 25(4):283
Prescher C, Prakapenka VB (2015) DIOPTAS: a program for reduction of two-dimensional X-ray diffraction data and data exploration. High Press Res 35(3):223–230
Shannon RD (1976) Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst A 32(751):751–767
Thornber MR, Nickel EH (1976) Supergene alteration of sulphides. III. The composition of associated carbonates. Chem Geol 17:45–72
Zhang J, Reeder R (1999) Comparative compressibilities of calcite-structured carbonates: Deviations from empirical relations. Am Miner 84(5–6):861–870
Acknowledgements
This work was supported by the US NSF EAR-1522560. Portions of this work were performed at GeoSoilEnviroCARS (The University of Chicago, Sector 13), Advanced Photon Source (APS), supported by the Argonne National Laboratory. GeoSoilEnviroCARS is supported by the National Science Foundation-Earth Sciences (EAR-1634415) and Department of Energy-GeoSciences (DE-FG02-94ER14466). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357. We would like to thank Eran Greenburg for help at APS beamline 13-ID-D and Martin Kunz for help at ALS beamline 12.2.2. The Advanced Light Source is supported by the Director, Office of Science, Office of Basic Energy Sciences, of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231. We would like to thank Sergey Tkachev at the Advanced Photon Source and Andrew Doran at the Advanced Light Source for gas loading: this was supported by COMPRES under NSF Cooperative Agreement EAR-1661511. We thank Lars Stixrude for helpful discussions and two anonymous reviewers for helpful comments.
Funding
This work was supported by the US NSF EAR-1522560. Portions of this work were performed at GeoSoilEnviroCARS (The University of Chicago, Sector 13), Advanced Photon Source (APS), supported by EAR-1634415, DE-FG02-94ER14466, and DE-AC02-06CH11357. Portions of this works were also performed at the Advanced Light Source supported by DE-AC02-05CH11231 and EAR-1661511.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest or competing interests in any of the material discussed in this article.
Availability of data and material
Any additional data not presented directly in this work can be requested by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sawchuk, K., Kamat, R., McGuire, C. et al. An X-ray diffraction and Raman spectroscopic study of the high-pressure behavior of gaspéite (Ni0.73Mg0.27CO3). Phys Chem Minerals 48, 7 (2021). https://doi.org/10.1007/s00269-020-01133-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00269-020-01133-3