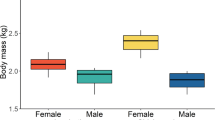
Abstract
Sexual segregation in feeding strategies has been widely reported in seabirds. Most seabirds occupy wide breeding distributional ranges, and dissimilar ecological settings may result in distinct environmental pressures on males and females leading to geographical differences in sexual segregation. Using GPS loggers and stable isotope analysis of whole blood (δ15N, δ13C), we assessed the occurrence of sexual differences in Kelp Gull (Larus dominicanus) foraging trip parameters, habitat use, isotopic niche and diet during the incubation period at three breeding locations in Argentina characterized by different foraging contexts. At Islote Arroyo Jabalí Este, sexual differences were found in trip parameters and habitat use mainly associated with a significantly higher use by males of a refuse dump as foraging site. However, their isotopic niches were similar with both sexes consuming mostly recreational fish waste, suggesting it was mainly obtained by males at the dump and by females at shoreline areas used by fishers. At Punta Tombo, there were no sex differences in trip parameters nor in the main prey consumed consisting of fishery discards, although females used more shoreline areas and males presented a significantly larger isotopic niche width. In contrast, at Isla Vernaci Este, males and females showed similar trip parameters, habitat use, and isotopic niche width and diet composition. Results show geographical differences in the degree of sexual segregation, which may result from differences in the availability of local food resources, particularly anthropogenic food subsidies.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Akaike H (1973) Maximum likelihood identification of Gaussian autoregressive moving average models. Biometrika 60(2):255–265. https://doi.org/10.1093/biomet/60.2.255
Arcos JA, Oro DO, Sol D (2001) Competition between the Yellow-legged Gull Larus cachinnans and Audouin’s Gull Larus audouinii associated with commercial fishing vessels: the influence of season and fishing fleet. Mar Biol 139:807–816. https://doi.org/10.1007/s002270100651
Bates D, Maechler M, Bolker B (2011) lme4: linear mixed-effects models using S4 classes, v. 0.999375-42. https://cran.r-project.org/package=lme4. Accessed 26 June 2019
Bearhop S, Phillips RA, McGill R, Cherel Y, Dawson DA, Croxall JP (2006) Stable isotopes indicate sex-specific and long-term individual foraging specialisation in diving seabirds. Mar Ecol Prog Ser 311:157–164. https://doi.org/10.3354/meps311157
Bertellotti M, Yorio P (1999) Spatial and temporal patterns in the diet of the Kelp Gull in northern Chubut, Patagonia. Condor 101:790–798. https://doi.org/10.2307/1370066
Bertellotti M, Yorio P (2000) Utilization of fishery waste by Kelp Gulls attending coastal trawl and longline vessels in northern Patagonia, Argentina. Ornis Fenn 77:105–115
Bolnick DI, Svanbäck R, Fordyce JM, Yang LH, Davis JM, Hulsey CD, Forister ML (2003) The ecology of individuals: incidence and implications of individual specialization. Am Nat 161:1–28. https://doi.org/10.1086/343878
Calado JG, Paiva VH, Ceia FR, Gomes P, Ramos JA, Velando A (2020) Stable isotopes reveal year-round sexual trophic segregation in four yellow-legged gull colonies. Mar Biol 167:1–11. https://doi.org/10.1007/s00227-020-3676-0
Camphuysen CJ (1995) Herring Gull Larus argentatus and Lesser Black-backed Gull L. fuscus feeding at fishing vessels in the breeding season: competitive scavenging versus efficient flying. Ardea 83:365–380. https://doi.org/10.1086/343878
Camphuysen KCJ, Shamoun-Baranes J, van Loon EE, Bouten W (2015) Sexually distinct foraging strategies in an omnivorous seabird. Mar Biol 162(7):1417–1428. https://doi.org/10.1007/s00227-015-2678-9
Ceia FR, Paiva VH, Fidalgo V, Morais L, Baeta A, Crisóstomo P, Mourato E, Garthe S, Marques JC, Ramos JA (2014) Annual and seasonal consistency in the feeding ecology of an opportunistic species, the Yellow-legged Gull Larus michahellis. Mar Ecol Prog Ser 497:273–284. https://doi.org/10.3354/meps10586
Ceia FR, Ramos JA (2015) Individual specialization in the foraging and feeding strategies of seabirds: a review. Mar Biol 162:1923–1938. https://doi.org/10.1007/s00227-015-2735-4
Cleasby IR, Wakefield ED, Bodey TW, Davies RD, Patrick SC, Newton J, Votier SC, Bearhop S, Hamer KC (2015) Sexual segregation in a wide-ranging marine predator is a consequence of habitat selection. Mar Ecol Prog Ser 518:1–12. https://doi.org/10.3354/meps11112
Cook TR, Cherel Y, Bost C, Tremblay Y (2007) Chick-rearing Crozet shags (Phalacrocorax melanogenis) display sex-specific foraging behaviour. Antarct Sci 19:55–63. https://doi.org/10.1017/S0954102007000089
Coulson R, Coulson G (1993) Diets of the Pacific Gull Larus pacificus and the Kelp Gull Larus dominicanus in Tasmania. Emu Austral Ornithol 93:50–53. https://doi.org/10.1071/MU9930050
Duhem C, Vidal E, Legrand J, Tatoni T (2003a) Opportunistic feeding responses of the Yellow-legged Gull Larus michahellis to accessibility of refuse dumps: the gulls adjust their diet composition and diversity according to refuse dump accessibility. Bird Stud 50:61–67. https://doi.org/10.1080/00063650309461291
Duhem C, Vidal E, Roche P, Legrand J (2003b) Island breeding and continental feeding: How are diet patterns in adult Yellow-legged gulls influenced by landfill accessibility and breeding stages. Ecoscience 10(4):502–508. https://doi.org/10.1080/11956860.2003.11682798
Fieberg J, Kochanny CO (2005) Quantifying home-range overlap: the importance of the utilization distribution. J Wildl Manag 69:1346–1359. https://doi.org/10.2193/0022-541X(2005)69[1346:QHOTIO]2.0.CO;2
Fuirst M, Veit RR, Hahn M, Dheilly N, Thorne LH (2018) Effects of urbanization on the foraging ecology and microbiota of the generalist seabird Larus argentatus. PLoS ONE 13(12):e0209200. https://doi.org/10.1371/journal.pone.0209200
Funes M, Marinao C, Galván DE (2019) Does trawl fisheries affect the diet of fishes? A stable isotope analysis approach. Isot Environ Health Stud 55(4):327–343. https://doi.org/10.1080/10256016.2019.1626381
García E, Bertness M, Alberti J, Silliman B (2011) Crab regulation of cross-ecosystem resource transfer by marine foraging fire ants. Oecologia 166:1111–1119. https://doi.org/10.1007/s00442-011-1952-x
García-Tarrasón M, Becares J, Bateman S, Arcos JM, Lluis J, Sanpera C (2015) Sex-specific foraging behavior in response to fishing activities in a threatened seabird. Ecol Evol 5:2348–2358. https://doi.org/10.1002/ece3.1492
Góngora ME, González-Zevallos D, Pettovello A, Mendía L (2012) Caracterización de las principales pesquerías del golfo San Jorge, Patagonia, Argentina. Lat Am J Aquat Res 40:1–11. https://doi.org/10.3856/vol40-issue1-fulltext-1
González-Solís J, Croxall JP, Wood AG (2000) Foraging partitioning between giant petrels Macronectes spp. and its relationship with breeding population changes at Bird Island. South Ga Mar Biol 204:279–288. https://doi.org/10.3354/meps204279
González-Zevallos D, Yorio P (2006) Seabird use of discards and incidental captures at the Argentine Hake trawl fishery in the Golfo San Jorge, Argentina. Mar Ecol Prog Ser 316:175–183. https://doi.org/10.3354/meps316175
González-Zevallos D, Yorio P (2011) Consumption of discards and interactions between Black-browed Albatrosses (Thalassarche melanophrys) and Kelp Gulls (Larus dominicanus) at trawl fisheries in Golfo San Jorge, Argentina. J Ornithol 152:827–838. https://doi.org/10.1007/s10336-011-0657-6
González-Zevallos D, Marinao C, Yorio P (2017) Importancia de los descartes pesqueros en la dieta de la Gaviota Cocinera (Larus dominicanus) en el golfo San Jorge, Patagonia. Ornitol Neotrop 28:103–111
Greig SA, Coulson JC, Monaghan P (1983) Age-related differences in foraging success in the Herring Gull (Larus argentatus). Anim Behav 31:1237–1243. https://doi.org/10.1016/S0003-3472(83)80030-X
Greig SA, Coulson JC, Monaghan P (1985) Feeding strategies of male and female adult Herring gulls (Larus argentatus). Behaviour 94:41–59. https://doi.org/10.1163/156853985X00262
Halley DJ, Minagawa M, Nieminen M, Gaare E (2008) Preservation in 70% ethanol solution does not affect d13C and d15N values of Reindeer blood samples-relevance for stable isotope studies of diet. Rangifer 28:9–12. https://doi.org/10.7557/2.28.1.146
Hansen JE, Martos P, Madirolas A (2001) Relationship between spatial distribution of the Patagonian stock of Argentine Anchovy, Engraulis anchoita, and sea temperatures during late spring to early summer. Fish Oceanogr 10:193–206
Hobson KA, Clark RG (1992) Assessing avian diets using stable isotopes. I: turnover of 13C in tissues. Condor 94:181–188. https://doi.org/10.2307/1368807
Hobson KA, Gibbs HL, Gloutney ML (1997) Preservation of blood and tissue samples for stable-carbon and stable-nitrogen isotopes analysis. Can J Zool 75:1720–1723. https://doi.org/10.1139/z97-799
Hurvich CM, Tsai CL (1989) Regression and time series model selection in small samples. Biometrika 76:297–307. https://doi.org/10.1093/biomet/76.2.297
Inger R, Bearhop S (2008) Applications of stable isotope analyses to avian ecology. Ibis 150:447–461. https://doi.org/10.1111/j.1474-919X.2008.00839.x
Jackson AL, Inger R, Parnell A, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER—stable isotope Bayesian ellipses in R. J Anim Ecol 80:595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x
Jackson MC, Donohue I, Jackson AL, Britton JR, Harper DM, Grey J (2012) Population-level metrics of trophic structure based on stable isotopes and their application to invasion ecology. PLoS ONE 7:e31757. https://doi.org/10.1371/journal.pone.0031757
Kasinsky T, Suárez N, Marinao C, Yorio P (2018) Kelp Gull (Larus dominicanus) use of alternative feeding habitats at the Bahía San Blas protected area, Argentina. Waterbirds 41:285–294. https://doi.org/10.1675/063.041.0308
Kazama K, Nishizawa B, Tsukamoto S, Gonzalez JE, Kazama MT (2018) Male and female Black-tailed gulls Larus crassirostris feed on the same prey species but use different feeding habitats. J Ornithol 159:923–934. https://doi.org/10.1007/s10336-018-1565-9
Lascelles BG, Taylor PR, Miller MGR, Dias MP, Oppel S, Torres L, Hedd A, Le Corre M, Phillips RA, Shaffer SA, Weimerskirch H, Small C (2016) Applying global criteria to tracking data to define important areas for marine conservation. Divers Distrib 22:422–431. https://doi.org/10.1111/ddi.12411
Lavoie RA, Rail J, Lean DRS (2012) Diet composition of seabirds from Corossol Island, Canada, using direct dietary and stable isotope analyses. Waterbirds 35(3):402–419. https://doi.org/10.1675/063.035.0305
Layman CA, Arrington DA, Montaña CG, Post DM (2007) Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 88:42–48. https://doi.org/10.1007/s00442-006-0630-x
Lewis S, Benvenuti S, Antonia LD, Griffiths R, Money L, Sherratt TN, Wanless S, Hamer KC (2002) Sex-specific foraging behaviour in a monomorphic seabird. Proc R Soc B 269:1687–1693. https://doi.org/10.1098/rspb.2002.2083
Lisnizer N, García Borboroglu P, Yorio P (2011) Spatial and temporal variations in kelp gull population trends in northern Patagonia, Argentina. Emu Austral Ornithol 111:259–267. https://doi.org/10.1071/MU11001
Llompart FM, Colautti DC, Baigun CRM (2012) Assessment of a major shore-based marine recreational fishery in the Southwest Atlantic, Argentina. New Zeal J Mar Fresh 46:57–70. https://doi.org/10.1080/00288330.2011.595420
Ludynia K, Garthe S, Luna-Jorquera G (2005) Seasonal and regional variation in the diet of the Kelp Gull in Northern Chile. Waterbirds 28:359–365. https://doi.org/10.1675/1524-4695(2005)028[0359:SARVIT]2.0.CO;2
Ludynia K, Dehnhard N, Poisbleau M, Demongin L, Masello JF, Voigt CC, Quillfeldt P (2013) Sexual segregation in rockhopper penguins during incubation. Anim Behav 85(1):255–267
Logan M (2011) Biostatistical design and analysis using R: a practical guide. Wiley, Oxford
Marinao C, Yorio P (2011) Use of fishery discards and incidental mortality of seabirds attending coastal shrimp trawlers in Isla Escondida, Patagonia, Argentina. Wilson J Ornithol 123:709–719. https://doi.org/10.1676/11-023.1
Marinao C, Góngora ME, González-Zevallos D, Yorio P (2014) Factors affecting Magellanic Penguin mortality at coastal trawlers in Patagonia, Argentina. Ocean Coast Manag 93:100–105. https://doi.org/10.1016/j.ocecoaman.2014.03.012
Marinao C, Kasinsky T, Suarez N, Yorio P (2018) Contribution of recreational fisheries to the diet of the opportunistic Kelp Gull. Austral Ecol 43:1–15. https://doi.org/10.1111/aec.12627
Monaghan P (1980) Dominance and dispersal between feeding sites in the Herring gull Larus argentatus. Anim Behav 28:521–527
Navarro J, Oro D, Bertolero A, Genovart M, Delgado A, Forero MG (2010) Age and sexual differences in the exploitation of two anthropogenic food resources for an opportunistic seabird. Mar Biol 157:2453–2459. https://doi.org/10.1007/s00227-010-1509-2
Newsome SD, Martinez del Rio C, Bearhop S, Phillips DL (2007) A niche for isotopic ecology. Front Ecol Environ 5:429–436. https://doi.org/10.1890/060150.1
Oro D, Genovart M, Tavecchia G, Fowler MS, Martínez-Abraín A (2013) Ecological and evolutionary implications of food subsidies from humans. Ecol Lett 16:1501–1514. https://doi.org/10.1111/ele.12187
Paiva VH, Ramos JA, Nava C, Neves V, Bried J, Magalhães M (2018) Inter-sexual habitat and isotopic niche segregation of the endangered Monteiro’s storm-petrel during breeding. Zoology 126:29–35. https://doi.org/10.1016/j.zool.2017.12.006
Parnell A, Inger R (2016) Stable isotope mixing models in R with simmr. https://cran.r-project.org/web/packages/simmr/vignettes/simmr.html. Accessed 17 Oct 2019
Petracci PF, LaSala LF, Aguerre G et al (2004) Dieta de la Gaviota cocinera (Larus dominicanus) durante el periodo reproductivo en el estuario de Bahía Blanca, Buenos Aires, Argentina. Hornero 19:23–28
Phillips RA, Xavier JC, Croxall JP (2003) Effects of satellite transmitters on albatrosses and petrels. Auk 120(4):1082–1090. https://doi.org/10.1093/auk/120.4.1082
Phillips RA, McGill RAR, Dawson DA, Bearhop, (2011) Sexual segregation in distribution, diet and trophic level of seabirds: insights from stable isotope analysis. Mar Biol 158(10):2199–2208. https://doi.org/10.1007/s00227-011-1725-4
Phillips DL, Inger R, Bearhop S, Jackson AL, Moore JW, Parnell AC, Semmens BX, Ward EJ (2014) Best practices for use of stable isotope mixing models in food-web studies. Can J Zool 92:823–835. https://doi.org/10.1139/cjz-2014-0127
Pinet P, Jaquemet S, Phillips RA, Le Corre M (2012) Sex-specific foraging strategies throughout the breeding season in a tropical, sexually monomorphic small petrel. Anim Behav 83:979–989. https://doi.org/10.1016/j.anbehav.2012.01.019
Pinheiro J, Bates D, DebRoy S, Sarkar D, Team RDC (2009) nlme: linear and nonlinear mixed effects models. http://cran.r-project.org/web/packages/nlme/index. Accessed 28 June 2019
Pons J (1994) Feeding strategies of male and female Herring gulls during the breeding season under various feeding conditions. Ethol Ecol Evol 6:1–12. https://doi.org/10.1080/08927014.1994.9523003
Post DM, Layman CA, Arrington DA, Takimoto G, Quattrochi J, Montaña CG (2007) Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152:179–189. https://doi.org/10.1007/s00442-006-0630-x
Quintana F, Wilson R, Dell’Arciprete P, Shepard E, Laich AG (2011) Women from Venus, men from Mars: inter-sex foraging differences in the imperial cormorant Phalacrocorax atriceps a colonial seabird. Oikos 120(3):350–358. https://doi.org/10.1111/j.1600-0706.2010.18387.x
Ramírez F, Navarro J, Afan I, Hobson KA, Delgado A, Forero MG (2012) Adapting to a changing world: unraveling the role of man-made habitats as alternative feeding areas for Slender-billed Gull (Chroicocephalus genei). PLoS ONE 7:e47551. https://doi.org/10.1371/journal.pone.0047551
Ramos R, Ramírez F, Sanpera C, Jover L, Ruiz X (2009) Diet of Yellow-legged Gull (Larus michahellis) chicks along the Spanish Western Mediterranean coast: the relevance of refuse dumps. J Ornithol 150(1):265–272. https://doi.org/10.1007/s10336-008-0346-2
R Development Core Team (2018) R-A language and environment for statistical computing. Rev.3.5.1. R Foundation for Statistical Computing, Vienna, Austria. http://r-project.org. Accessed 26 June 2019
Ronconi RA, Steenweg RJ, Taylor PD, Mallory ML (2014) Gull diets reveal dietary partitioning, influences of isotopic signatures on body condition, and ecosystem changes at a remote colony. Mar Ecol Prog Ser 514:247–261. https://doi.org/10.3354/meps10980
Schwemmer P, Garthe S (2008) Regular habitat switch as an important feeding strategy of an opportunistic seabird species at the interface between land and sea. Estuar Coast Shelf S 77:12–22. https://doi.org/10.1016/j.ecss.2007.08.017
Silva MP, Bastida R, Darrieu C (2000) Dieta de la Gaviota Cocinera Larus dominicanus en zonas costeras de la provincia de Buenos Aires, Argentina. Ornitol Neotrop 11:331–339
Smith JA, Mazumder D, Suthers IM, Taylor MD (2013) To fit or not to fit: Evaluating stable isotope mixing models using simulated mixing polygons. Meth Ecol Evol 4:612–618. https://doi.org/10.1111/2041-210X.12048
Steele WK (1992) Diet of Hartlaubs Gull Larus hartlaubii and the Kelp Gull L. dominicanus in the southwestern Cape Province. S Afr Ostrich 63:68–82. https://doi.org/10.1080/00306525.1992.9633952
Storero LP, Botto F, Narvarte MA, Iribarne OO (2016) Influence of maturity condition and habitat type on food resources utilization by Octopus tehuelchus in Atlantic Patagonian coastal ecosystems. Mar Biol 163:1–10. https://doi.org/10.1007/s00227-016-2952-5
Suárez N, Marinao C, Kasinsky T, Yorio P (2014) Distribución reproductiva y abundancia de gaviotas y gaviotines en el Área Natural Protegida Bahía San Blas, Buenos Aires. Hornero 29:29–36
Székely T, Reynolds JD, Figuerola J (2000) Sexual size dimorphism in shorebirds, gulls, and alcids: the influence of sexual and natural selection. Evolution 54:1404–1413. https://doi.org/10.1111/j.0014-3820.2000.tb00572.x
Thomson JA, Heithaus MR, Burkholder DA, Vaudo JJ, Wirsing AJ, Dill LM (2012) Site specialists, diet generalists? Isotopic variation, site fidelity, and foraging by Loggerhead Turtles in Shark Bay, Western Australia. Mar Ecol Prog Ser 453:213–226. https://doi.org/10.3354/meps09637
Torlaschi C, Gandini P, Frere E, Martinez Peck R (2000) Predicting the sex of Kelp Gulls by external measurements. Waterbirds 23:518–520. https://doi.org/10.2307/1522193
Turner TF, Collyer ML, Krabbenhoft TJ (2010) A general hypothesis testing framework for stable isotope ratios in ecological studies. Ecology 91:2227–2233. https://doi.org/10.1890/09-1454.1
Washburn BE, Bernhardt GE, Kutschbach-Brohl L, Chipman RB, Francoeur LC (2013) Foraging ecology of four Gull species at a coastal-urban interface. Condor 115:67–76. https://doi.org/10.1890/09-1454.1
Wearmouth VJ, Sims DW (2008) Sexual segregation in marine fish, reptiles, birds and mammals: behaviour patterns, mechanisms and conservation implications. Adv Mar Biol 54:107–170. https://doi.org/10.1016/S0065-2881(08)00002-3
Weimerskirch H, Shaffer SA, Tremblay Y, Costa DP, Gadenne H, Kato A, Ropert-Coudert Y, Sato K, Aurioles D (2009) Species- and sex-specific differences in foraging behaviour and foraging zones in blue-footed and brown boobies in the Gulf of California. Mar Ecol Prog Ser 391:267–278. https://doi.org/10.3354/meps07981
Welcker J, Steen H, Harding AM, Gabrielsen GW (2009) Sex-specific provisioning behaviour in a monomorphic seabird with a bimodal foraging strategy. Ibis 151(3):502–513. https://doi.org/10.1111/j.1474-919X.2009.00931.x
Wood AG, Naef-Daenzer B, Prince PA, Croxall JP (2000) Quantifying habitat use in satellite tracked pelagic seabirds: application of kernel estimation to Albatross locations. J Avian Biol 31:278–286. https://doi.org/10.1034/j.1600-048X.2000.310302.x
Yorio P, Caille G (1999) Seabird interactions with coastal fisheries in northern Patagonia: use of discards and incidental captures in nets. Waterbirds 22:201–216. https://doi.org/10.2307/1522209
Yorio P, García Borboroglu P (2002) Breeding biology of Kelp Gulls (Larus dominicanus) at Golfo San Jorge, Patagonia, Argentina. Emu 102:257–263. https://doi.org/10.1071/MU00077
Yorio P, Giaccardi M (2002) Urban and fishery waste tips as food sources for birds in northern coastal Patagonia, Argentina. Ornitol Neotrop 13:283–292
Yorio P, Marinao C, Retana MV, Suárez N (2013) Differential use of food resources between the Kelp Gull (Larus dominicanus) and the threatened Olrog’s Gull (L. atlanticus). Ardeola 60:29–44. https://doi.org/10.13157/arla.60.1.2012.29
Yorio P, Suárez N, Kasinsky T, Pollicelli M, Ibarra C, Gatto A (2020) The introduced green crab (Carcinus maenas) as a novel food resource for the opportunistic Kelp Gull (Larus dominicanus) in Argentine Patagonia. Aquat Invasions 15(1):140–159. https://doi.org/10.3391/ai.2020.15.1.10
Zabala S, Bigatti G, Botto F, Iribarne OO, Galván DE (2013) Trophic relationships between a Patagonian gastropod and its epibiotic anemone revealed by using stable isotopes and direct observations. Mar Biol 160:909–919. https://doi.org/10.1007/s00227-012-2143-y
Zalba SM, Nebbia AJ, Fiori SM (2008) Propuesta de Plan de Manejo de la Reserva Natural de Uso Múltiple Bahía San Blas. Universidad Nacional del Sur, Bahía Blanca
Zavalaga CB, Benvenuti S, Dall’Antonia L, Emslie SD (2007) Diving behaviour of Blue-footed boobies Sula nebouxii in northern Perú in relation to sex, body size and prey type. Mar Ecol Prog Ser 336:291–303. https://doi.org/10.3354/meps336291
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank Cynthia Ibarra, Miriam Pollicelli, Ximena Navoa and Juan Carileo for field-work assistance. We also thank Centro para el Estudio de Sistemas Marinos (CCT CONICET-CENPAT) for institutional support. We thank three anonymous reviewers and Susan Elbin for comments on the manuscript.
Funding
This research was funded by the Wildlife Conservation Society. Tatiana Kasinsky was supported by a doctoral scholarship from CONICET. The permits to access the protected areas were provided by the “Dirección de Áreas Naturales Protegidas” of Buenos Aires (Disposición Nº 201/12), “Secretaría de Turismo y Áreas Protegidas” of Chubut (Disposiciones No. 163-SsCyAP/15 and 092-SsCyAP/16) and “Dirección de Fauna y Flora Silvestre” of Chubut (Disposiciones Nº72/15 and 65/16.DFyFS).
Author information
Authors and Affiliations
Contributions
TK, PY and NS contributed to the study conception and design. Data collection was performed by TK, CM and NS, and data analysis was performed by TK and PDA. The first draft of the manuscript was written by TK and PY, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
This research was funded by the Wildlife Conservation Society. All authors declare that they have no conflict of interests.
Ethical approval
Animal handling and blood sampling were permitted under appropriate permits (Disposiciones Nº 201/12-OPDS, 163-SsCyAP/15, 092-SsCyAP/16, Nº 72/15.DFyFSand 65/16.DFyFS). All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Additional information
Responsible editor: V. Paiva.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by J. Calado, M. Huttunen and undisclosed experts.
Supplementary Information
Below is the link to the Supplementary Information.
Rights and permissions
About this article
Cite this article
Kasinsky, T., Yorio, P., Dell’Arciprete, P. et al. Geographical differences in sex-specific foraging behaviour and diet during the breeding season in the opportunistic Kelp Gull (Larus dominicanus). Mar Biol 168, 14 (2021). https://doi.org/10.1007/s00227-020-03812-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-020-03812-9