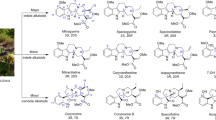
Abstract
Chronic administration of opioids produces physical dependence and opioid-induced hyperalgesia. Users claim the Thai traditional tea “kratom” and component alkaloid mitragynine ameliorate opioid withdrawal without increased sensitivity to pain. Testing these claims, we assessed the combined kratom alkaloid extract (KAE) and two individual alkaloids, mitragynine (MG) and the analog mitragynine pseudoindoxyl (MP), evaluating their ability to produce physical dependence and induce hyperalgesia after chronic administration, and as treatments for withdrawal in morphine-dependent subjects. C57BL/6J mice (n = 10/drug) were administered repeated saline, or graded, escalating doses of morphine (intraperitoneal; i.p.), kratom alkaloid extract (orally, p.o.), mitragynine (p.o.), or MP (subcutaneously, s.c.) for 5 days. Mice treated chronically with morphine, KAE, or mitragynine demonstrated significant drug-induced hyperalgesia by day 5 in a 48 °C warm-water tail-withdrawal test. Mice were then administered naloxone (10 mg/kg, s.c.) and tested for opioid withdrawal signs. Kratom alkaloid extract and the two individual alkaloids demonstrated significantly fewer naloxone-precipitated withdrawal signs than morphine-treated mice. Additional C57BL/6J mice made physically dependent on morphine were then used to test the therapeutic potential of combined KAE, mitragynine, or MP given twice daily over the next 3 days at either a fixed dose or in graded, tapering descending doses. When administered naloxone, mice treated with KAE, mitragynine, or MP under either regimen demonstrated significantly fewer signs of precipitated withdrawal than control mice that continued to receive morphine. In conclusion, while retaining some liabilities, kratom, mitragynine, and mitragynine pseudoindoxyl produced significantly less physical dependence and ameliorated precipitated withdrawal in morphine-dependent animals, suggesting some clinical value.




Similar content being viewed by others
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper. Data related to this manuscript may be requested from the authors.
References
Adkins JE, Boyer EW, Mccurdy CR (2011) Mitragyna speciosa, a psychoactive tree from Southeast Asia with opioid activity. Curr Top Med Chem 11(9):1165–1175. https://doi.org/10.2174/156802611795371305
Balter RE, Dykstra LA (2013) Thermal sensitivity as a measure of spontaneous morphine withdrawal in mice. J Pharmacol Toxicol Methods 67(3):162–168. https://doi.org/10.1016/j.vascn.2013.02.003
Boyer EW, Babu KM, Adkins JE, Mccurdy CR, Halpern JH (2008) Self-treatment of opioid withdrawal using kratom (Mitragynia speciosa korth). Addiction 103(6):1048–1050. https://doi.org/10.1111/j.1360-0443.2008.02209.x
Canestrelli C, Marie N, Noble F (2014) Rewarding or aversive effects of buprenorphine/ naloxone combination (Suboxone) depend on conditioning trial duration. Int J Neuropsychopharmacol 17(9):1367–1373. https://doi.org/10.1017/S146114571400025X
Célèrier E, Rivat C, Jun Y, Laulin J-P, Larcher A, Reynier P, Simonnet G (2000) Long-lasting hyperalgesia induced by fentanyl in rats. Anesthesiology 92(2):465. https://doi.org/10.1097/00000542-200002000-00029
Chakraborty S, Majumdar S (2020) Natural products for the treatment of pain: chemistry and pharmacology of Salvinorin A, Mitragynine, and Collybolide. Biochemistry. https://doi.org/10.1021/acs.biochem.0c00629
Chu LF, Sun J, Clemenson A, Erlendson MJ, Rico T, Cornell E, Obasi H, Sayyid ZN, Encisco EM, Yu J, Gamble JG, Carroll I, Clark JD (2017) Ondansetron does not reduce withdrawal in patients with physical dependence on chronic opioid therapy. J Addict Med 11(5):342–349. https://doi.org/10.1097/ADM.0000000000000321
Chu LF, Rico T, Cornell E, Obasi H, Encisco EM, Vertelney H, Gamble JG, Crawford CW, Sun J, Clemenson A, Erlendson MJ, Okada R, Carroll I, Clark JD (2018) Ondansetron does not prevent physical dependence in patients taking opioid medications chronically for pain control. Drug Alcohol Depend 183:176–183. https://doi.org/10.1016/j.drugalcdep.2017.06.043
Crain SM, Shen K-F (2001) Acute thermal hyperalgesia elicited by low-dose morphine in normal mice is blocked by ultra-low-dose naltrexone, unmasking potent opioid analgesia. Brain Res 888(1):75–82. https://doi.org/10.1016/s0006-8993(00)03010-9
Dehan A, Yassen A, Bijl H, Romberg R, Sarton E, Teppema L, Olofsen E, Danhof M (2005) Comparison of the respiratory effects of intravenous buprenorphine and fentanyl in humans and rats. Brit J Anesth 94(6):825–834. https://doi.org/10.1093/bja/aei145
Devulder J, Bohyn P, Castille F, Laat MD, Rolly G (1996) A case of uncommon withdrawal symptoms after a short period of spinal morphine administration. Pain 64(3):589–591. https://doi.org/10.1016/0304-3959(95)00187-5
Doughty B, Morgenson D, Brooks T (2019) Lofexidine: a newly FDA-approved, nonopioid treatment for opioid withdrawal. Ann Pharmacother 53:747–753. https://doi.org/10.1177/1060028019828954
Dum J, Bläsig J, Herz A (1981) Buprenorphine: demonstration of physical dependence liability. Eur J Pharmacol 70(3):293–300. https://doi.org/10.1016/0014-2999(81)90163-1
Fishman M, Tirado C, Alam D, Gullo K, Clinch T, Gorodetzky CW (2019) Safety and efficacy of lofexidine for medically managed opioid withdrawal: a randomized controlled clinical trial. J Addict Med 13:169–176. https://doi.org/10.1097/ADM.000000000000074
Gold M, Redmond D, Kleber H (1978) Clonidine blocks acute opiate-withdrawal symptoms. Lancet 312(8090):599–602. https://doi.org/10.1016/s0140-6736(78)92823-4
Gowing L, Farrell MF, Ali R, White JM (2014) Alpha2-adrenergic agonists for the management of opioid withdrawal. Cochrane Database Syst Rev 3:CD002024. https://doi.org/10.1002/14651858.cd002024.pub4
Grim TW, Schmid CL, Stahl EL, Pantouli F, Ho J-H, Avevedo-Canabal A, Kennedy NM, Cameron MD, Bannister TD, Bohn LM (2020) A G protein signaling-biased agonist at the μ-opioid receptor reverses morphine tolerance with preventing morphine withdrawal. Neuropsychopharmacology 45(2):416–425. https://doi.org/10.1038/s41386-019-0491-8
Gutridge AM, Robins MT, Cassell RJ, Uprety R, Mores KL, Ko MJ, Pasternak GW, Majumdar S, van Rijn RM (2020) G protein-biased kratom-alkaloids and synthetic carfentanil-amide opioid as potential treatments for alcohol use disorder. Br J Pharmacol 177(7):1497–1513. https://doi.org/10.1111/bph.14913
Harun N, Hassan Z, Navaratnam V, Mansor SM, Shoaib M (2015) Discriminative stimulus properties of mitragynine (kratom) in rats. Psychopharmacology 232(13):2227–2238. https://doi.org/10.1007/s00213-015-3866-5
Hassan Z, Muzaimi M, Navaratnam V, Yusoff NH, Suhaimi FW, Vadivelu R, Vicknasingam BK, Amato D, von Hörsten S, Ismail NI, Jayabalan N (2013) From Kratom to mitragynine and its derivatives: Physiological and behavioural effects related to use, abuse, and addiction. Neurosci Biobehav Rev 37(2):138–151. https://doi.org/10.1016/j.neubiorev.2012.11.012
Hassan R, See CP, Sreenivasan S, Mansor SM, Müller CP, Hassan Z (2020) Mitragynine attenuates morphine withdrawal effects in rats—a comparison with methadone and buprenorphine. Front Psychiatry. https://doi.org/10.3389/fpsyt.2020.00411
Hiranita T, Leon F, Felix JS, Restrepo LF, Reeves ME, Pennington AE, Obeng S, Avery BA, McCurdy CR, McMahon LR, Wilkerson JL (2019) The effects of mitragynine and morphine on schedule-controlled responding and antinociception in rats. Psychopharmacology 236(9):2725–2734. https://doi.org/10.1007/s00213-019-05247-7
Hutcheson DM, Everitt BJ, Robbins TW, Dickinson A (2001) The role of withdrawal in heroin addiction: enhances reward or promotes avoidance? Nat Neurosci 4(9):943–947. https://doi.org/10.1038/nn0901-943
Hutchinson MR, Shavit Y, Grace PM, Rice KC, Maier SF, Watkins LR (2011) Exploring the neuroimmunopharmacology of opioids: an integrative review of mechanisms of central immune signaling and their implications for opioid analgesia. Pharmacol Rev 63(3):772–810. https://doi.org/10.1124/pr.110.004135
Jasinski DR (1985) Clonidine in morphine withdrawal. Arch Gen Psychiatry 42(11):1063. https://doi.org/10.1001/archpsyc.1985.01790340041006
Jhamandas K, Marsala M, Ibuki T, Yaksh T (1996) Spinal amino acid release and precipitated withdrawal in rats chronically infused with spinal morphine. J Neurosci 16(8):2758–2766. https://doi.org/10.1523/jneurosci.16-08-02758.1996
Journigan VB, Mésangeau C, Vyas N, Eans SO, Cutler SJ, McLaughlin JP, Mollereau C, McCurdy CR (2014) Nonpeptide small molecule agonist and antagonist original leads for neuropeptide FF1 and FF2 receptors. J Med Chem 57(21):8903–8927. https://doi.org/10.1021/jm500989n
Kamble SH, León F, King TI, Berthold EC, Lopera-Londoño C, Siva Rama Raju K, Hampson AJ, Sharma A, Avery BA, McMahon LR, McCurdy CR (2020) Metabolism of a kratom alkaloid metabolite in human plasma increases its opioid potency and efficacy. ACS Pharmacol Transl Sci. https://doi.org/10.1021/acsptsci.0c00075
Kamei J, Ohsawa M (1997) Role of noradrenergic functions in the modification of naloxone-precipitated withdrawal jumping in morphine-dependent mice by diabetes. Life Sci 60(15):PL223-228. https://doi.org/10.1016/s0024-3205(97)00074-x
Katz J (1986) Effects of clonidine and morphine on opioid withdrawal in rhesus monkeys. Psychopharmacology. https://doi.org/10.1007/bf00180844
Khor B-S, Jamil MFA, Adenan MI, Shu-Chien AC (2011) Mitragynine attenuates withdrawal syndrome in morphine-withdrawn zebrafish. PLoS ONE. https://doi.org/10.1371/journal.pone.0028340
Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG (2010) Animal research: reporting in vivo experiments: the ARRIVE guidelines. Brit J Pharmacol 160(7):1577–1579
King T, Ossipov MH, Vanderah TW, Porreca F, Lai J (2005) Is paradoxical pain induced by sustained opioid exposure an underlying mechanism of opioid antinociceptive tolerance? Neurosignals 14(4):194–205. https://doi.org/10.1159/000087658
Kleber HD (2007) Pharmacologic treatments for opioid dependence: detoxification and maintenance options. Dialogues Clin Neurosci 9:455–470
Koob GF, Caine S, Parsons L, Markou A, Weiss F (1997) Opponent process model and psychostimulant addiction. Pharmacol Biochem Behav 57(3):513–521. https://doi.org/10.1016/s0091-3057(96)00438-8
Kosten TR, O'Connor PG (2003) Management of drug and alcohol withdrawal. New Engl J Med 348(18):1786–1795
Kosten TR, Baxter LE (2019) Effective management of opioid withdrawal symptoms: a gateway to opioid dependence treatment. Am J Addict 28:55–62. https://doi.org/10.1111/ajad.12862
Kreek MJ, Laforge KS, Butelman E (2002) Pharmacotherapy of addictions. Nat Rev Drug Discov 1(11):710–726. https://doi.org/10.1038/nrd897
Kreek MJ, Borg I, Ducat E, Ray B (2010) Pharmacotherapy in the treatment of addiction: methadone. J Addict Dis 29:200–216. https://doi.org/10.1080/10550881003684798
Kruegel AC, Grundmann O (2018) The medicinal chemistry and neuropharmacology of kratom: a preliminary discussion of a promising medicinal plant and analysis of its potential for abuse. Neuropharmacology 134:108–120. https://doi.org/10.1016/j.neuropharm.2017.08.026
Kruegel AC, Gassaway MM, Kapoor A, Váradi A, Majumdar S, Filizola M, Javitch JA, Sames D (2016) Synthetic and receptor signaling explorations of the mitragyna alkaloids: mitragynine as an atypical molecular framework for opioid receptor modulators. J Am Chem Soc 138(21):6754–6764. https://doi.org/10.1021/jacs.6b00360
Kruegel AC, Uprety R, Grinnell SG, Langreck C, Pekarskaya EA, Le Rouzic V, Ansonoff M, Gassaway MM, Pintar JE, Pasternak GW, Javitch JA, Majumdar S, Sames D (2019) 7-Hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS Cent Sci 5(6):992–1001. https://doi.org/10.1021/acscentsci.9b00141
Laulin J-P, Maurette P, Corcuff J-B, Rivat C, Chauvin M, Simonnet AG (2002) The role of ketamine in preventing fentanyl-induced hyperalgesia and subsequent acute morphine tolerance. Anesth Analg 94(5):1263–1269. https://doi.org/10.1097/00000539-200205000-00040
Li X, Angst MS, Clark JD (2001) Opioid-induced hyperalgesia and incisional pain. Anesth Analg. https://doi.org/10.1097/00000539-200107000-00040
Lipman JJ, Blumenkopf B (1989) Comparison of subjective and objective analgesic effects of intravenous and intrathecal morphine in chronic pain patients by heat beam dolorimetry. Pain 39(3):249–256. https://doi.org/10.1016/0304-3959(89)90037-7
Matsumoto K, Mizowaki M, Suchitra T, Takayama H, Sakai S-I, Aimi N, Watanabe H (1996a) Antinociceptive action of mitragynine in mice: evidence for the involvement of supraspinal opioid receptors. Life Sci 59(14):1149–1155. https://doi.org/10.1016/0024-3205(96)00432-8
Matsumoto K, Mizowaki M, Suchitra T, Murakami Y, Takayama H, Sakai SI, Aimi N, Watanabe H (1996b) Central antinociceptive effects of mitragynine in mice: contribution of descending noradrenergic and serotonergic systems. Eur J Pharmacol 317(1):75–81. https://doi.org/10.1016/s0014-2999(96)00714-5
Meepong R, Sooksawate T (2019) Mitragynine reduced morphine-induced conditioned place preference and withdrawal in rodents. Thai J Pharm Sci 43(1):21–29
Minami M, Satoh M (1995) Molecular biology of the opioid receptors: structures, functions and distributions. Neurosci Res 23(2):121–145. https://doi.org/10.1016/0168-0102(95)00933-k
Miser AW, Chayt KJ, Sandlund JT, Cohen PS, Dothage JA, Miser JS (1986) Narcotic withdrawal syndrome in young adults after the therapeutic use of opiates. Am J Dis Child 140(6):603–604. https://doi.org/10.1001/archpedi.1986.02140200113039
Obeng S, Kamble SH, Reeves ME, Restrepo LF, Patel A, Behnke M, Chear NJ-Y, Ramanathan S, Sharma A, León F, Hiranita T, Avery BA, McMahon LR, McCurdy CR (2020) Investigation of the adrenergic and opioid binding affinities, metabolic stability, plasma protein binding properties, and functional effects of selected indole-based kratom alkaloids. J Med Chem 63(1):433–439
Özdoğan ÜK, Lähdesmäki J, Scheinin M (2003) Influence of prazosin and clonidine on morphine analgesia, tolerance and withdrawal in mice. Eur J Pharmacol 460(2–3):127–134. https://doi.org/10.1016/S0014-2999(02)02961-8
Pinelli A, Trivulzio S, Tomasoni L (1997) Effects of ondansetron administration on opioid withdrawal syndrome observed in rats. Eur J Pharmacol 340(2–3):111–119. https://doi.org/10.1016/s0014-2999(97)01349-6
Sabetghadam A, Ramanathan S, Mansor SM (2010) The evaluation of antinociceptive activity of alkaloid, methanolic, and aqueous extracts of Malaysian Mitragyna speciosa Korth leaves in rats. Pharmacogn Res 2(3):181–185. https://doi.org/10.4103/0974-8490.65514
Shafer SL, Varvel JR (1991) Pharmacokinetics, pharmacodynamics, and rational opioid selection. Anesthesiology 74(1):53–63. https://doi.org/10.1097/00000542-199101000-00010
Shaw-Lutchman TZ, Barrot M, Wallace T, Gilden L, Zachariou V, Impey S, Duman RS, Storm D, Nestler EJ (2002) Regional and cellular mapping of cAMP response element-mediated transcription during Naltrexone-precipitated morphine withdrawal. J Neurosci 22(9):3663–3672
Singh D, Yeou Chear NJ, Narayanan S, Leon F, Sharma A, McCurdy CR, Avery BA, Balasingam V (2020) Patterns and reasons for kratom (Mitragyna speciose) use among current and former opioid poly-drug users. J Ethnopharmacol 249:112462. https://doi.org/10.1016/j.jep.2019.112462
Srivastava AB, Mariani JJ, Levin FR (2020) New directions in the treatment of opioid withdrawal. Lancet 395(10241):1938–1948. https://doi.org/10.1016/S0140-6736(20)30852-7
Sufka KJ, Hughes RA, Giordano J (1991) Effects of selective opiate antagonists on morphine-induced hyperalgesia in domestic fowl. Pharmacol Biochem Behav 38(1):49–54. https://doi.org/10.1016/0091-3057(91)90588-s
Sun HL (1998) Naloxone-precipitated acute opioid withdrawal syndrome after epidural morphine. Anesth Analg 86(3):544–545. https://doi.org/10.1213/00000539-199803000-00019
Toce MS, Chai PR, Burns MM, Boyer EW (2018) Pharmacologic treatment of opioid use disorder: a review of pharmacotherapy, adjuncts, and toxicity. J Med Toxicol 14:306–312. https://doi.org/10.1007/s13181-018-0685-1
Umbricht A, Montoya ID, Hoover DR, Demuth KL, Chiang CT, Preston KL (1999) Naltrexone shortened opioid detoxification with buprenorphine. Drug Alcohol Depend 56:181–190. https://doi.org/10.1016/s0376-8716(99)00033-2
Váradi A, Marrone GF, Palmer TC, Narayan A, Szabó MR, Le Rouzic V, Grinnell SG, Subrath JJ, Warner E, Kalra S, Hunkele A (2016) Mitragynine/corynantheidine pseudoindoxyls as opioid nalgesics with Mu agonism and delta antagonism, which do not recruit β-arrestin-2. J Med Chem 59(18):8381–8397. https://doi.org/10.1021/acs.jmedchem.6b00748
Wakim JH (2012) Alleviating symptoms of withdrawal from an opioid. Pain Ther 1(4):1–6. https://doi.org/10.1007/s40122-012-0004-5
Wilson LL, Harris HM, Eans SO, Brice-Tutt AC, Cirino TJ, Stacy HM, Simons CA, León F, Sharma A, Boyer EW, Avery BA (2020) Lyophilized kratom tea as a therapeutic option for opioid dependence. Drug Alcohol Depend 216:108310. https://doi.org/10.1016/j.drugalcdep.2020.108310
Xin W, Chun W, Ling L, Wei W (2011) Role of melatonin in the prevention of morphine-induced hyperalgesia and spinal glial activation in rats: protein kinase C pathway involved. Int J Neurosci 122(3):154–163. https://doi.org/10.3109/00207454.2011.635828
Yusoff NH, Suhaimi FW, Vadivelu RK, Hassan Z, Rümler A, Rotter A, Amato D, Dringenberg HC, Mansor SM, Navaratnam V, Müller CP (2014) Abuse potential and adverse cognitive effects of mitragynine (kratom). Addict Biol 21(1):98–110. https://doi.org/10.1111/adb.12185
Funding
This research was supported by Grants from the National Institute on Drug Abuse RO1DA046487 and R21/R33 DA045884 (to SM), Cancer Center Support Grant P30 CA008748 from the National Cancer Institute (to MSKCC), and funds from the University of Florida (to JPM).
Author information
Authors and Affiliations
Contributions
SM, JPM and LLW participated in research design and wrote and contributed to the writing of manuscript. TJC, SOE, CAS, HMS and LLW conducted experiments. SM, SC and RU contributed new reagents or analytic tools. JPM and LLW performed data analysis. All authors critically reviewed the content and approved the final version for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests.
Ethical Approval
All animal studies were preapproved by the University of Florida (Gainesville, FL, USA) Institutional Animal Care and Use Committee, in accordance with the 2011 National Institute of Health Guide for the Care and Use of Laboratory Animals. Animal studies are reported in compliance with the ARRIVE guidelines (Kilkenny et al. 2010). Initial sample sizes were approximated by power analysis, with animals assigned to groups randomly. Drug treatment experiments were conducted in a blinded fashion.
Informed Consent
This manuscript has been approved for publication by all authors.
Additional information
This manuscript contributes to the Special Issue to honor Professor Gavril W. Pasternak, who passed on Feb 22nd, 2019. The authors dedicate this work to his memory, teachings and friendship.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wilson, L.L., Chakraborty, S., Eans, S.O. et al. Kratom Alkaloids, Natural and Semi-Synthetic, Show Less Physical Dependence and Ameliorate Opioid Withdrawal. Cell Mol Neurobiol 41, 1131–1143 (2021). https://doi.org/10.1007/s10571-020-01034-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-01034-7