Abstract
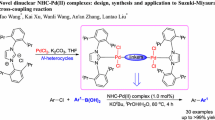
The catalytic activity of C,N-chelate diaminocarbene palladium(II) complexes containing a 3,4-diaryl-1H-pyrrol-2,5-diimine fragment in a copper-free Sonogashira reaction was studied. Reactions catalyzed by C,N-chelate diaminocarbene palladium(II) complexes do not require preliminary degassing, since such catalysts are air- and moisture-stable. In this work, comparative analysis of the catalytic activity of two types of C,N-chelate diaminocarbene complexes containing, along with the diaminocarbene ligand, isonitrile and chloride or two chloride ligands in the inner coordination sphere has been carried out. The steric and electronic effects of the substituents in the catalyst on the reaction yield has been studied.


Similar content being viewed by others
REFERENCES
Sonogashira, K., Tohda, Y., and Hagihara, N., Tetrahedron Lett., 1975, vol. 16, p. 4467. https://doi.org/10.1016/s0040-4039(00)91094-3
Biffis, A., Centomo, P., Zotto, A.D., and Zecca, M., Chem. Rev., 2018, vol. 118, p. 2249. https://doi.org/10.1021/acs.chemrev.7b00443
Bai, X., Wu, C., Ge, S., and Lu, Y., Angew. Chem., Int. Ed., 2020, vol. 59, p. 2764. https://doi.org/10.1002/anie.201913148
Chinchilla, R. and Najera, C., Chem. Rev., 2007, vol. 107, p. 874. https://doi.org/10.1021/cr050992x
Chinchilla, R. and Najera, C., Chem. Soc. Rev., 2011, vol. 40, p. 5084. https://doi.org/10.1039/C1CS15071E
Cacchi, S., Morera, E., and Ortar, G., Synthesis, 1986, vol. 4, p. 320. https://doi.org/10.1055/s-1986-31599
Schilz, M. and Plenio, H., J. Org. Chem., 2012, vol. 77, p. 2798. https://doi.org/10.1021/jo202644g
Wan, C.-W., Burghart, A., Chen, J., Bergstrom, F., Johanson, L.B.A., Wolford, M.F., Kim, T.G., Topp, M.R., Hochstrasser, R.M., and Burgess, K., Chem. Eur. J., 2003, vol. 9, p. 4430. https://doi.org/10.1002/chem.200304754
Shirakawa, E., Kitabata, T., Otsuka, H., and Tsuchimoto, T., Tetrahedron, 2005, vol. 61, p. 9878. https://doi.org/10.1016/j.tet.2005.07.099
Mikhaylov, V.N., Sorokoumov, V.N., Liakhov, D.M., Tskhovrebov, A.G., and Balova, I.A., Catalysts, 2018, vol. 8, p. 141. https://doi.org/10.3390/catal8040141
Yang, L., Guan, P., He, P., Chen, Q., Cao, C., Peng, Y., Shi, Z., Pang, G., and Shi, Y., Dalton Trans., 2012, vol. 41, p. 5020. https://doi.org/10.1039/c2dt12391f
Fortman, G.C. and Nolan, S.P., Chem. Soc. Rev., 2011, vol. 40, p. 5151. https://doi.org/10.1039/c1cs15088j
Touj, N., Yas, S.¸ Özdemir, N., Hamdi, N., and Özdemir, I., J. Organometal. Chem., 2018, vol. 860, p. 59. https://doi.org/10.1016/j.jorganchem.2018.01.017
Valishina, E.A., da Silva, M.F.C.G., Kinzhalov, M.A., Timofeeva, S.A., Buslaeva, T.M., Haukka, M., Pombeiro, A.J.L., Boyarskiy, V.P., Kukushkin, V.Yu., and Luzyanin, K.V., J. Mol. Cat. A: Chem., 2014, vol. 395, p. 162. https://doi.org/10.1016/j.molcata.2014.08.018
Sonogashira, K., Metal-Catalyzed Cross-Coupling Reactions, Diederich, F. and Stang, P.J., Eds., Weinheim: Wiley-VCH, 1998, p. 203.
Brandsma, L., Vasilevsky, S.F., and Verkruijsse, H.D., Application of Transition Metal Catalysts in Organic Synthesis, Berlin: Springer, 1998, p. 179.
Sonogashira, K., Handbook of Organopalladium Chemistry for Organic Synthesis, Negishi, E. and de Meijere, A., Eds., New York: Wiley-VCH, 2002.
Sonogashira, K., J. Organometal. Chem., 2002, vol. 653, p. 46. https://doi.org/10.1016/S0022-328X(02)01158-0
Negishi, E. and Luigi, A., Chem. Rev., 2003, vol. 103, p. 1979. https://doi.org/10.1021/cr020377i
Littke, A.F. and Fu, G.C., Angew. Chem., Int. Ed., 2002, vol. 41, p. 4176. https://doi.org/10.1002/1521-3773(20021115)41:22<
Boubakri, L., Mansour, L., Harrath, A.H., Özdemir, I., Yaşar, S., and Hamdi, N., J. Coord. Chem., 2018, vol. 71, p. 183. https://doi.org/10.1080/00958972.2018.1430791
Boyarskaya, D.V., Bulatov, E., Boyarskaya, I.A., Chulkova, T.G., Rassadin, V.A., Tolstopjatova, E.G., Kolesnikov, I.E., Avdontceva, M.S., Panikorovskii, T.L., Suslonov, V.V., and Haukka, M., Organometallics, 2019, vol. 38, p. 300. https://doi.org/10.1021/acs.organomet.8b00725
Crociani, B., Boschi, T., and Belluco, U., Inorg. Chem., 1970, vol. 9, p. 2021. https://doi.org/10.1021/ic50091a013
Boyarskaya, D., Avdontceva, M., and Chulkova, T., Acta Crystallogr., Sect. C, 2015, vol. 71, p. 155. https://doi.org/10.1107/S2053229615001588
Afanasenko, A.M., Boyarskaya, D.V., Boyarskaya, I.A., Chulkova, T.G., Grigoriev, Y.M., Kolesnikov, I.E., Avdontceva, M.S., Panikorovskii, T.L., Panin, A.I., Vereshchagin, A.N., and Elinson, M.N., J. Mol. Struct., 2017, vol. 1146, p. 554. https://doi.org/10.1016/j.molstruc.2017.06.048
Afanasenko, A.M., Novikov, A.S., Chulkova, T.G., Grigoriev, Y.M., Kolesnikov, I.E., Selivanov, S.I., Starova, G.L., Zolotarev, A.A., Vereshchagin, A.N., and Elinson, M.N., J. Mol. Struct., 2020, vol. 1199, p. 126789. https://doi.org/10.1016/j.molstruc.2019.07.036
Afanasenko, A.M., Chulkova, T.G., Boyarskaya, I.A., Islamova, R.M., Legin, A.A., Keppler, B.K., Selivanov, S.I., Vereshchagin, A.N., Elinson, M.N., and Haukka, M., J. Organometal. Chem., 2020, vol. 923, p. 121435. https://doi.org/10.1016/j.jorganchem.2020.121435
Pu, X., Li, H., and Colacot, T.J., J. Org. Chem., 2013, vol. 78, p. 568. https://doi.org/10.1021/jo302195y
ACKNOWLEDGMENTS
The work was performed using the equipment of the Magnetic Resonance Research Center, St. Petersburg State University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Boyarskaya, D.V., Chulkova, T.G. Effect of the Structure of C,N-Chelate Diaminocarbene Palladium(II) Complexes on Their Catalytic Activity in the Sonogashira Reaction. Russ J Org Chem 56, 1937–1941 (2020). https://doi.org/10.1134/S107042802011007X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042802011007X