Abstract
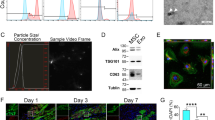
Bone marrow-derived mesenchymal stem cells (BMSCs) have been considered a promising therapeutic approach to cardiovascular disease. This study intends to compare the effect of BMSCs through a standard active cardiac support device (ASD) and intravenous injection on global myocardial injury induced by isoproterenol. BMSCs were cultured in vitro, and the transplanted cells were labeled with a fluorescent dye CM-Dil. Isoproterenol (ISO) was injected into the rats; 2 weeks later, the labeled cells were transplanted into ISO-induced heart-jury rats through the tail vein or ASD device for 5 days. The rats were sacrificed on the first day, the third day, and the fifth day after transplantation to observe the distribution of cells in the myocardium by fluorescence microscopy. The hemodynamic indexes of the left ventricle were measured before sacrificing. H&E staining and Masson’s trichrome staining were used to evaluate the cardiac histopathology. In the ASD groups, after 3 days of transplantation, there were a large number of BMSCs on the epicardial surface, and after 5 days of transplantation, BMSCs were widely distributed in the ventricular muscle. But in the intravenous injection group, there were no labeled-BMSCs distributed. In the ASD + BMSCs-three days treated group and ASD + BMSCs -five days-treated group, left ventricular systolic pressure (LVSP), the maximum rate of left ventricular pressure rise (+dP/dt), the maximum rate of left ventricular pressure decline (-dP/dt) increased compared with model group and intravenous injection group (P < 0.05). By giving BMSCs through ASD device, cells can rapidly and widely distribute in the myocardium and significantly improve heart function.






Similar content being viewed by others
References
I.M. Barbash et al., Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: Feasibility, cell migration, and body distribution. Circulation 108, 863–868 (2003). https://doi.org/10.1161/01.CIR.0000084828.50310.6A
H.S. Bernstein, D. Srivastava, Stem cell therapy for cardiac disease. Pediatr. Res. 71, 491–499 (2012). https://doi.org/10.1038/pr.2011.61
Cai B et al. (2015) Mesenchymal stem cells and Cardiomyocytes interplay to prevent myocardial hypertrophy stem cells. Transl Med 4:1425-1435. https://doi.org/10.5966/sctm.2015-0032
N.G. Campbell, K. Suzuki, Cell delivery routes for stem cell therapy to the heart: current and future approaches. J. Cardiovasc. Transl. Res. 5, 713–726 (2012). https://doi.org/10.1007/s12265-012-9378-3
C. Carr, Bone marrow-derived stromal cells home to and remain in the infarcted rat heart but fail to improve function: an in vivo cine-MRI study. Am. J. Physiol. Heart Circ. Physiol. (2008). https://doi.org/10.1152/ajpheart.00094.2008.-Basic
N. Dib, H. Khawaja, S. Varner, M. McCarthy, A. Campbell, Cell therapy for cardiovascular disease: a comparison of methods of delivery. J. Cardiovasc. Transl. Res. 4, 177–181 (2011). https://doi.org/10.1007/s12265-010-9253-z
S.A. Doppler, M.A. Deutsch, R. Lange, M. Krane, Cardiac regeneration: current therapies-future concepts. J Thorac Dis 5, 683–697 (2013). https://doi.org/10.3978/j.issn.2072-1439.2013.08.71
E. Forte et al., Cardiac cell therapy: The next (re)generation. Stem Cell Rev. 7, 1018–1030 (2011). https://doi.org/10.1007/s12015-011-9252-8
S. Fukushima et al., Choice of cell-delivery route for skeletal myoblast transplantation for treating post-infarction chronic heart failure in rat. PLoS One 3, e3071 (2008). https://doi.org/10.1371/journal.pone.0003071
C.L. Hastings, E.T. Roche, E. Ruiz-Hernandez, K. Schenke-Layland, C.J. Walsh, G.P. Duffy, Drug and cell delivery for cardiac regeneration. Adv. Drug Deliv. Rev. 84, 85–106 (2015). https://doi.org/10.1016/j.addr.2014.08.006
Q. He, Z. Ye, Y. Zhou, W.-S. Tan, Comparative study of mesenchymal stem cells from rat bone marrow and adipose tissue vol 42 (2018). https://doi.org/10.3906/biy-1802-52
A.W. Heldman et al., Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: The TAC-HFT randomized trial. JAMA 311, 62–73 (2014). https://doi.org/10.1001/jama.2013.282909
M. Hristov, Intracoronary infusion of autologous bone marrow cells and left ventricular function after acute myocardial infarction: a meta-analysis. J. Cell. Mol. Med. 10 (2006). https://doi.org/10.2755/jcmm010.003.05
R. John. J.M.P. Teerlink, M.A. Pfeffer, Progressive Ventricular Remodeling in Response to Diffuse Isoproterenol-Induced Myocardial Necrosis in Rats (1994)
S.K. Lee et al., Differential expression of cell surface proteins in human bone marrow mesenchymal stem cells cultured with or without basic fibroblast growth factor containing medium. PROTEOMICS 9, 4389–4405 (2009). https://doi.org/10.1002/pmic.200900165
W. Li, R. Gan, G. Sun, Chronic treatment of enbrel in rats with isoproterenol-induced congestive heart failure limits left ventricular dysfunction and remodeling. Chin. Med. J. 115, 1166–1169 (2002)
L. Li, Y. Zhang, Y. Li, B. Yu, Y. Xu, S. Zhao, Z. Guan, Mesenchymal stem cell transplantation attenuates cardiac fibrosis associated with isoproterenol-induced global heart failure. Transpl. Int. 21, 1181–1189 (2008). https://doi.org/10.1111/j.1432-2277.2008.00742.x
C. Li et al., Therapeutic advances in cardiac targeted drug delivery: From theory to practice. J. Drug Target. 1-14 (2020a). https://doi.org/10.1080/1061186X.2020.1818761
X. Li et al., An epicardial delivery of nitroglycerine by active hydraulic ventricular support drug delivery system improves cardiac function in a rat model. Drug Deliv Transl Res 10, 23–33 (2020b). https://doi.org/10.1007/s13346-019-00656-9
Z. Liu et al., Systemic and local delivery of mesenchymal stem cells for heart renovation: Challenges and innovations. Eur. J. Pharmacol. 876, 173049 (2020). https://doi.org/10.1016/j.ejphar.2020.173049
R. Mikrani et al., Pharmacokinetic advantage of ASD device promote drug absorption through the epicardium. Pharm. Res. 37, 173 (2020). https://doi.org/10.1007/s11095-020-02898-6
M. Naveed et al., A novel ventricular restraint device (ASD) repetitively deliver Salvia miltiorrhiza to epicardium have good curative effects in heart failure management biomedicine & pharmacotherapy. Biomed. Pharmacother. 95, 701–710 (2017). https://doi.org/10.1016/j.biopha.2017.07.126
M. Naveed et al., Suppression of TGP on myocardial remodeling by regulating the NF-κB pathway. Biomed. Pharmacother. 108, 1460–1468 (2018a). https://doi.org/10.1016/j.biopha.2018.09.168
M. Naveed et al., Cardio-supportive devices (VRD & DCC device) and patches for advanced heart failure: A review, summary of state of the art and future directions. Biomed. Pharmacother. 102, 41–54 (2018b). https://doi.org/10.1016/j.biopha.2018.03.049
M. Naveed et al., The promising future of ventricular restraint therapy for the management of end-stage heart failure. Biomed. Pharmacother. 99, 25–32 (2018c). https://doi.org/10.1016/j.biopha.2018.01.003
M. Naveed et al., A review of medicinal plants in cardiovascular disorders: benefits and risks. Am. J. Chin. Med. 48, 259–286 (2020). https://doi.org/10.1142/S0192415X20500147
L.H. Opie, P.J. Commerford, B.J. Gersh, M.A. Pfeffer, Controversies in ventricular remodelling. Lancet 367, 356–367 (2006). https://doi.org/10.1016/s0140-6736(06)68074-4
D. Orlic, J. Kajstura, S. Chimenti, D.M. Bodine, A. Leri, P. Anversa, Bone marrow stem cells regenerate infarcted myocardium. Pediatr. Transplant. 7, 86–88 (2003). https://doi.org/10.1034/j.1399-3046.7.s3.13.x
A.M. Orogo, Å.B. Gustafsson, Cell death in the myocardium: My heart won't go on IUBMB. Life 65, 651–656 (2013). https://doi.org/10.1002/iub.1180
J. Ren, C. Li, S. Ma, J. Wu, Y. Yang, Impact of dexmedetomidine on hemodynamics in rabbits. Acta Cir. Bras. 33, 314–323 (2018). https://doi.org/10.1590/s0102-865020180040000003
Y. Sawa, Current status of myocardial regeneration therapy. Gen. Thorac. Cardiovasc. Surg. 61, 17–23 (2013). https://doi.org/10.1007/s11748-012-0153-9
S. Seth et al., The ABCD (autologous bone marrow cells in dilated cardiomyopathy) trial a long-term follow-up study. J. Am. Coll. Cardiol. 55, 1643–1644 (2010). https://doi.org/10.1016/j.jacc.2009.11.070
N. Smart et al., Myocardial regeneration: Expanding the repertoire of thymosin beta4 in the ischemic heart annals of the New York. Acad. Sci. 1269, 92–101 (2012). https://doi.org/10.1111/j.1749-6632.2012.06708.x
N.A. Staffend, R.L. Meisel, DiOlistic labeling of neurons in tissue slices: a qualitative and quantitative analysis of methodological variations. Front. Neuroanat. 5, 14 (2011). https://doi.org/10.3389/fnana.2011.00014
B.-E. Strauer, The acute and long-term effects of intracoronary stem cell transplantation in 191 patients with chronic heARt failure: The STAR-heart study. Eur. J. Heart Fail. (2010). https://doi.org/10.1093/eurheartj/hfq105
B.E. Strauer, G. Steinhoff, 10 years of intracoronary and intramyocardial bone marrow stem cell therapy of the heart: From the methodological origin to clinical practice. J. Am. Coll. Cardiol. 58, 1095–1104 (2011). https://doi.org/10.1016/j.jacc.2011.06.016
J.R. Teerlink, Progressive ventricular remodeling in response to diffuse isoproterenol-induced myocardial necrosis in rats. Circ. Res. 1 (1994)
P. Tropel, D. Noel, N. Platet, P. Legrand, A.L. Benabid, F. Berger, Isolation and characterisation of mesenchymal stem cells from adult mouse bone marrow. Exp. Cell Res. 295, 395–406 (2004). https://doi.org/10.1016/j.yexcr.2003.12.030
K. Wei et al., Epicardial FSTL1 reconstitution regenerates the adult mammalian heart. Nature 525, 479–485 (2015). https://doi.org/10.1038/nature15372
R.S. Whelan, V. Kaplinskiy, R.N. Kitsis, Cell death in the pathogenesis of heart disease: mechanisms and significance. Annu. Rev. Physiol. 72, 19–44 (2010). https://doi.org/10.1146/annurev.physiol.010908.163111
S. Yue, Cardiac support device (ASD) delivers bone marrow stem cellsrepetitively to epicardium has promising curative effects in advanced heart failure. Biomed. Microdevices. (2018). https://doi.org/10.1007/s10544-018-0282-8
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant nos.30973003 and 30901993), Administration of TCM Jiangsu Province China (No. LZ11093) Qing Lan Project, National Found for Fostering Talents of Basic Science of PR China (No. J1030830). Furthermore, all the authors thank and acknowledge their respective Universities and Institutes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Z., Naveed, M., Baig, M.M.F.A. et al. Therapeutic approach for global myocardial injury using bone marrow-derived mesenchymal stem cells by cardiac support device in rats. Biomed Microdevices 23, 5 (2021). https://doi.org/10.1007/s10544-020-00538-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-020-00538-9