Abstract
A nano-complex containing fish scale collagen, konjac glucomannan, camellia chrysantha polyphenols and ginsenoside Rb1 (GCPR) have been prepared successfully by gelation method. Infrared spectroscopy (IR), scanning field emission scanning electron microscopy (FESEM), Energy-dispersive X-ray spectroscopy (EDX), dynamic light scattering (DLS), ultraviolet–visible spectroscopy (UV–Vis) were used to determine characteristics of obtained GCPR nano-complex. The GCPR nano-complex is in spherical shape with the average particle size is 115.4 nm (95.1%) and 593.0 nm (4.1%). Polyphenols and ginsenoside Rb1 could interact with collagen and konjac glucomannan through hydrogen bonding and dipole–dipole interactions, therefore, the drug release from the GCPR nano-complex in pH 2 buffer solution (simulated gastric fluid in the human body) and pH 7.4 buffer solution (simulated intestinal fluid in the human body) could be controlled. In particular, biological activity of the GCPR nano-complex in inhibition of cancer cells, anti-inflammatory and antioxidant also was investigated. The obtained results show that the GCPR nano-complex exhibits a positive effect on inhibition of cancer cells and anti-inflammatory in comparison with polyphenol or ginsenoside Rb1. Besides, the GCPR nano-complex and polyphenol also exhibit non-toxic to normal cells and antioxidation ability. This nano-complex is promising for application in biomedicine.
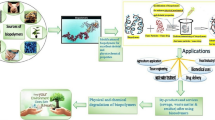
Graphic Abstract








Similar content being viewed by others
References
Arruebo M, Vilaboa N, Sáez-Gutierrez B, Lambea J, Tres A, Valladares M, González-Fernández A (2011) Assessment of the evolution of cancer treatment therapies. Cancers 3(3):3279
Baskar R, Kuo Ann L, Richard Y, Kheng-Wei Y (2012) Cancer and radiation therapy: current advances and future directions. Int J Med Sci 9(3):193
Nicholson LB (2016) The immune system. Essays Biochem 60(3):275
Wang H, Khor TO, Shu L, Su ZY, Fuentes F, Lee JH, Kong AN (2012) Plants vs. cancer: a review on natural phytochemicals in preventing and treating cancers and their druggability. Anticancer Agents Med Chem 12(10):1281
Hu XQ, Sun Y, Lau E, Zhao M, Su SB (2016) Advances in synergistic combinations of chinese herbal medicine for the treatment of cancer. Curr Cancer Drug Targets 16(4):346
Scalbert A, Manach C, Morand C, Remesy C, Jimenez L (2005) Dietary polyphenols and the prevention of diseases. Crit Rev Food Sci Nutr 45(4):287
Fu QY, Li QS, Lin XM, Qiao RY, Yang R, Li XM, Dong ZB, Xiang LP, Zheng XQ, Lu JL, Yuan CB, Ye JH, Liang YR (2017) Antidiabetic effects of tea. Molecules 22(5):1
Higdon JV, Frei B (2003) Tea catechins and polyphenols: health effects, metabolism, and antioxidant functions. Crit Rev Food Sci Nutr 43(1):89
Rafieian KM, Movahedi M (2017) Breast cancer chemopreventive and chemotherapeutic effects of Camellia sinensis (green tea): an updated review. Electron Physician 9(2):3838
Jin-Bin W, Li X, Song H, Yong-Hong L, Yu-Zheng P, Jun-Xiang R, Qin X, Yong-Xin C, Cai-Li N, Zhi-Heng S (2015) Characterization and determination of antioxidant components in the leaves of Camellia chrysantha (Hu) Tuyama based on composition activity relationship approach. J Food Drug Anal 23:40
Yi-Fang L, Shu-Hua O, Yi-Qun C, Ting-Mei W, Wei-Xi L, Hai-Yan T, Hong C, Hiroshi K, Rong-Rong H (2017) A comparative analysis of chemical compositions in Camellia sinensis var. puanensis Kurihara, a novel Chinese tea, by HPLC and UFLC-Q-TOF-MS/MS. Food Chem 216:282
Simos YV, Verginadis II, Toliopoulos IK, Velalopoulou AP, Karagounis IV, Karkabounas SC, Evangelou AM (2012) Effects of catechin and epicatechin on superoxide ismutase and glutathione peroxidase activity, in vivo. Redox Rep 17:181
Lixia S, Xiangshe W, Xueqin Z, Dejian H (2011) Polyphenolic antioxidant profiles of yellow camellia. Food Chem 129:351
Jia-Ni L, Hui-Yi L, Ning-Sun Y (2013) Chemical constituents and anticancer activity of yellow camellias against MDA-MB-231 human breast cancer cells. Food Chem 61:9638
Lai W, Debmalya R, Sen LS, Tao YS, Li S (2017) Hypoglycemic effect of Camellia chrysantha extract on type 2 diabetic mice model. Bangladesh J Pharmacol 12:359
Gopal J, Muthu M, Paul D, Doo-Hwan K, Sechul C (2016) Bactericidal activity of greentea extracts: the importance of catechin containing nano particles. Sci Rep 6:19710
Yokozawa T, Nakagawa T, Kitani K (2002) Antioxidative activity of green polyphenol in cholesterol-fed rats. J Agric Food Chem 50(12):3549
Lau AJ, Toh DF, Chua TK, Pang YK, Woo SO, Koh HL (2009) Antiplatelet and anticoagulant effects of Panax notoginseng: comparison of raw and steamed Panax notoginseng with Panax ginseng and Panax quinquefolium. J Ethnopharmacol 125(3):380
Shibata S, Ando T, Tanaka O, Meguro Y, Soma K, Lida Y (1965) Saponins and sapogenins of Panax ginseng C.A. Meyer and some other Panax spp. Yakugaku Zasshi 85:753
Shibata S, Tanaka O, Soma K, Ando T, Lida Y, Nakamura H (1965) Studies on saponins and sapogenins of ginseng. The structure of panaxatriol. Tetrahedron Lett 42:207
Attele AS, Wu JA, Yuan CS (1999) Ginseng pharmacology: multiple constituents and multiple actions. Biochem Pharmacol 58:1685
Lee DC, Lau AS (2011) Effects of Panax ginseng on tumor necrosis factor-α-mediated inflammation: a mini-review. Molecules 16:2802
Ying C (1998) Pharmacodynamical research and clinical application of Panax pseudo-ginseng saponins. Guangxi Med J 20(6):1109
Yuangui Y, Zhengcai J, Yingbo Y, Yanhai Z, Li Y, Zhengtao W (2020) Phytochemical analysis of Panax species: a review. J Ginseng Res (inpress). https://doi.org/10.1016/j.jgr.2019.12.009
FangXu Q, LingFang L, FengChen D (2003) Pharmacokinetics and bioavailability of ginsenoside Rb1 and Rg1 from Panax notoginseng in rats. J Ethnopharmacol 84(2–3):187
Yuan Q, Jiang YW, Ma TT, Fang QH, Pan L (2014) Attenuating effect of ginsenoside Rb1 on LPS-induced lung injury in rats. J Inflamm (Lond) 11(1):40
Zhou Q, Jiang L, Xu C, Luo D, Zeng C, Liu P, Hu H (2014) Ginsenoside Rg1 inhibits platelet activation and arterial thrombosis. Thromb Res 133(1):57
Guo-Xiang L, Zai-Qun L (2008) The protective effects of ginsenosides on human erythrocytes against hemin-induced hemolysis. Food Chem Toxicol 46:886
Yuan Q, Hui-Ying L, Xiao-Xi G, Yan L, Cheng-Xiao W, Jiang-Hua H, Tian-Rui X, Ye Y, Xiu-Ming C (2018) Converting ginsenosides from stems and leaves of Panax notoginseng by microwave processing and improving their anticoagulant and anticancer activities. RSC Adv 8:40471
Yiying B, Gwang-Jin A, Keunyoung K, Thien N, Sue S, Ok-Nam B, Kyung-Min L, Jin-Ho C (2019) Ginsenoside Rg3, a component of ginseng, induces pro-thrombotic activity of erythrocytes via hemolysis-associated phosphatidylserine exposure. Food Chem Toxicol 131:11055
Orive G, Gascon AR, Hernández RM, Domínguez-Gil A, Pedraz JL (2004) Techniques: new approaches to the delivery of biopharmaceuticals. Trends Pharmacol Sci 25:382
Sadukhan S, Bakshi P, Maiti S (2014) Tailored bio-polymeric nanomicellar carriers: a promising approach for the delivery of poorly water soluble drugs. J Adv Pharm Educ Res 4(1):41
Tanbour R, Martins AM, Pitt WG, Husseini GA (2016) Drug delivery systems based on polymeric micelles and ultrasound: a review. Curr Pharml Des 22(19):2796
Swamy BY, Prasad CV, Prabhakar MN, Rao KC, Subha MCS, Chung I (2013) Biodegradable chitosan-g-poly(methacrylamide) microspheres for controlled release of hypertensive drug. J Polym Environ 21:1128–1134
Singh M, O’Hagan D (1998) The preparation and characterization of polymeric antigen delivery systems for oral administration. Adv Drug Deliv Rev 34(2–3):285
Ngwuluka NC, Ochekpe N (2015) Natural polymers: drug delivery. Encyclopedia of biomedical polymers and polymeric biomaterials. Taylor and Francis, New York, pp 5603–5618. https://doi.org/10.1081/E-EBP
Holban AM, Mihai A (2016) Nanoarchitectured polysaccharide-based drug carrier for ocular therapeutics. Nanoarchitecton Smart Deliv Drug Target. https://doi.org/10.1016/C2015-0-06101-9
Yetisgin AA, Cetinel S, Zuvin M, Kosar A, Kutlu O (2020) Therapeutic nanoparticles and their targeted delivery applications. Molecules 25(9):2193
Patra JK, Das G, Fraceto LF, Campos E, Rodriguez-Torres M, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S, Habtemariam S, Shin HS (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16(1):71
Qin Y, Guo XW, Li L, Wang HW, Kim W (2013) The antioxidant property of chitosan green tea polyphenols complex induces trans-glutaminase activation in wound healing. J Med Food 16(6):487
Zhang Y, Xie B, Gan X (2005) Advance in the applications of konjac glucomannan and its derivatives. Carbohydr Polym 60:27
Doi K (1995) Effect of konjac fibre (glucomannan) on glucose and lipids. Eur J Clin Nutr 3:190
Sood N, Baker WL, Coleman CI (2008) Effect of glucomannan on plasma lipid and glucose concentrations, body weight, and blood pressure: systematic review and meta-analysis. Am J Clin Nutr 88:1167
Fatuma F, Chunlei X, Wei-Yan Q, Hanmei X (2018) Collagen from marine biological sources and medical. Chem Biodivers 15(5):e1700557
Daniela C, Maria O, Giovanni V, Chiara L, Isabella D, Salvatore I, Donatella P (2020) Marine collagen from alternative and sustainable sources: extraction. Process Appl Mar Drugs 18(4):214
Yamamoto K, Igawa K, Sugimoto K, Yoshizawa Y, Yanagiguchi K, Ikeda T, Yamada S, Hayashi Y (2014) Biological safety of fish (tilapia) collagen. BioMed Res Int 2014:630757
Aras O, Kazanci M (2015) Production of collagen micro- and nanofibers for potential drug-carrier systems. J Enzyme Inhib Med Chem 30(6):1013
Zhang R, Wang X, Wang J, Cheng M (2018) Synthesis and characterization of konjac glucomannan/carrageenan/nano-silica films for the preservation of postharvest white mushrooms. Polymers 11(1):6
Naghshineh N, Tahvildari K, Nozari M (2019) Preparation of chitosan, sodium alginate, gelatin and collagen biodegradable sponge composites and their application in wound healing and curcumin delivery. J Polym Environ 27:2819–2830
Voicu G, Geanaliu-Nicolae RE, Pîrvan AA, Andronescu E, Iordache F (2016) Synthesis, characterization and bioevaluation of drug-collagen hybrid materials for biomedical applications. Int J Pharm 510:474
Ziemys A, Yokoi K, Kojic M (2015) Capillary collagen as the physical transport barrier in drug delivery to tumor microenvironment. Tissue Barriers 3(3):e1037418
Yang C, Wu H, Wang J (2019) Formulation and evaluation of controlled-release of steroidal saponins-loaded collagen microspheres. Mater Technol 34:1
Petrisor G, Ion RM, Brachais CH, Boni G, Plasseraud L, Couvercelle JP, Chambin O (2012) In Vitro Release of local anaesthetic and anti-inflammatory drugs from crosslinked collagen based device. J Macromol Sci A 49(9):699
Chinh NT, Manh VQ, Thuy PT, Trung VQ, Quan VA, Ginag BL, Hoang T (2020) Novel pH-sensitive hydrogel beads based on carrageenan and fish scale collagen for allopurinol drug delivery. J Polym Environ 28(6):1795
Lu-Hui W, Guo-Qing H, Tong-Cheng X, Jun-Xia X (2019) Characterization of carboxymethylated konjac glucomannan for potential application in colon-targeted delivery. Food Hydrocoll 94:354
Huiqun Y, Chaoboo X (2008) Synthesis and properties of novel hydrogels from oxidized konjac glucomannan crosslinked gelatin for in vitro drug delivery. Carbohydr Polym 72(3):479
Yi Y, Lin W, Ruo-Jun M, Jingni G, Yuyan W, Yuanzhao L, Jiaqi M, Jie P, Chunhua W (2018) Effects of konjac glucomannan on the structure, properties, and drug release characteristics of agarose hydrogels. Carbohydr Polym 190:196
Chinh NT, Manh VQ, Trung VQ, Lam TD, Huynh MD, Tung NQ, Trinh ND, Hoang T (2019) Characterization of collagen derived from tropical freshwater carp fish scale wastes and its amino acid sequence. Nat Prod Commun 14(7):1–12
Tim M (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assay. J Immunol Methods 65:55
Dirsch VM, Stuppner H, Vollmar AM (1998) The Griess assay: suitable for a bio-guided fractionation of anti-inflammatory plant extracts? Planta Med 64:423
Marxen K, Vanselow KH, Lippemeier S, Hintze R, Ruser A, Ulf-Peter H (2007) Determination of DPPH radical oxidation caused by methanolic extracts of some Microalgal species by linear regression analysis of spectrophotometric measurements. Sensors 7:2080
Rezaei A, Nasirpour A (2019) Evaluation of release kinetics and mechanisms of curcumin and curcumin-β-cyclodextrin inclusion complex incorporated in electrospun almond gum/PVA nanofibers in simulated saliva and simulated gastrointestinal conditions. BioNano Sci 9:438
Acknowledgements
This research is funded by Vietnam Academy of Science and Technology under Grant Number DLTE00.04/20-21, period of 2020–2021.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The Authors declare that there are no competing interests associated with this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ngo, D.Q., Nguyen, T.C., Nguyen, T.D. et al. A Novel Biopolymer Nano-Complex Based on Fish Scale Collagen, Konjac Glucomannan, Camellia Chrysantha Polyphenols and Ginsenoside Rb1: Preparation, Characterization and Its Bioactivity. J Polym Environ 29, 2150–2163 (2021). https://doi.org/10.1007/s10924-020-02022-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-020-02022-0