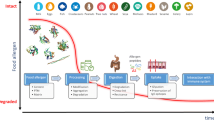
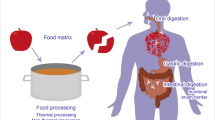
Abstract
Key determinants for the development of an allergic response to an otherwise ‘harmless’ food protein involve different factors like the predisposition of the individual, the timing, the dose, the route of exposure, the intrinsic properties of the allergen, the food matrix (e.g. lipids) and the allergen modification by food processing. Various physicochemical parameters can have an impact on the allergenicity of animal proteins. Following our previous review on how physicochemical parameters shape plant protein allergenicity, the same analysis was proceeded here for animal allergens.
We found that each parameter can have variable effects, ranging on an axis from allergenicity enhancement to resolution, depending on its nature and the allergen. While glycosylation and phosphorylation are common, both are not universal traits of animal allergens. High molecular structures can favour allergenicity, but structural loss and uncovering hidden epitopes can also have a similar impact. We discovered that there are important knowledge gaps in regard to physicochemical parameters shaping protein allergenicity both from animal and plant origin, mainly because the comparability of the data is poor. Future biomolecular studies of exhaustive, standardised design together with strong validation part in the clinical context, together with data integration model systems will be needed to unravel causal relationships between physicochemical properties and the basis of protein allergenicity.


Similar content being viewed by others
Abbreviations
- 2D:
-
Secondary structure
- 3D:
-
Tertiary structure
- 4D:
-
Quaternary structure
- BAT:
-
Basophil activation test
- DBPCFC:
-
Double-blind placebo-controlled food challenge
- EAST:
-
Enzyme allergosorbent test
- ELISA:
-
Enzyme-linked immunosorbent assay
- HPP:
-
High-pressure processing
- HHP:
-
High-hydrostatic pressure
- IgE:
-
Immunoglobulin E
- IgG:
-
Immunoglobulin G
- OFC:
-
Open food challenge
- PEF:
-
Pulsed electric fields
- PTM:
-
Post-translational modifications
- PUV:
-
Pulsed ultraviolet
- RAST:
-
Radioallergosorbent test
- RBL:
-
Rat basophilic leukaemia
- SPT:
-
Skin prick tests
- Th1, Th2:
-
T helper cell type 1 or 2
- WHO/IUIS:
-
World Health Organiztion/International Union of Immunological Societies
References
Berin MC, Sampson HA (2013) Food allergy: an enigmatic epidemic. Trends Immunol 34:390–397. https://doi.org/10.1016/j.it.2013.04.003
Sampson HA (2016) Food allergy: past, present and future. Allergol Int 65:363–369. https://doi.org/10.1016/j.alit.2016.08.006
Sicherer SH, Sampson HA (2018) Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J Allergy Clin Immunol 141:41–58. https://doi.org/10.1016/j.jaci.2017.11.003
Costa J, Bavaro SL, Benedé S, Diaz-Perales A, Bueno-Diaz C, Gelencser E, Klueber J, Larré C, Lozano-Ojalvo D, Lupi R, Mafra I, Mazzucchelli G, Molina E, Monaci L, Martín-Pedraza L, Piras C, Rodrigues PM, Roncada P, Schrama D, Cirkovic-Velickovic T, Verhoeckx K, Villa C, Kuehn A, Hoffmann-Sommergruber K, Holzhauser T (2020) Are physicochemical properties shaping the allergenic potency of plant allergens? Clin Rev Allergy Immunol. https://doi.org/10.1007/s12016-020-08810-9
Radauer C, Bublin M, Wagner S, Mari A, Breiteneder H (2008) Allergens are distributed into few protein families and possess a restricted number of biochemical functions. J Allergy Clin Immunol 121:847-852.e847. https://doi.org/10.1016/j.jaci.2008.01.025
The Database of Allergen Families, Medical University of Vienna, Vienna, Austria (2020) http://www.meduniwien.ac.at/allfam/. Accessed 6 April 2020
World Health Organization/International Union of Immunological Societies (WHO/IUIS) Allergen Nomenclature Sub-committee (2020) http://www.allergen.org/. Accessed 31 March 2020
Wang CLA, Coluccio LM (2010) New insights into the regulation of the actin cytoskeleton by tropomyosin. Int Rev Cell Mol Biol 281:91–128. https://doi.org/10.1016/S1937-6448(10)81003-2
Gimona M (2008) Dimerization of tropomyosins. In: Gunning P (ed) Tropomyosin. Springer, New York, NY, pp 73–84. https://doi.org/10.1007/978-0-387-85766-4_6
Reese G, Ayuso R, Lehrer SB (1999) Tropomyosin: an invertebrate pan-allergen. Int Arch Allergy Immunol 119:247–258. https://doi.org/10.1159/000024201
Klueber J, Costa J, Randow S, Codreanu-Morel F, Verhoeckx K, Bindslev-Jensen C, Ollert M, Hoffmann-Sommergruber K, Morisset M, Holzhauser T, Kuehn A (2020) Homologous tropomyosins from vertebrate and invertebrate: recombinant calibrator proteins in functional biological assays for tropomyosin allergenicity assessment of novel animal foods. Clin Exp Allergy 50:105–116. https://doi.org/10.1111/cea.13503
Miegel A, Kobayashi T, Maéda Y (1992) Isolation, purification and partial characterization of tropomyosin and troponin subunits from the lobster tail muscle. J Muscle Res Cell Motil 13:608–618. https://doi.org/10.1007/bf01738250
Mills ENC, Johnson PE, Alexeev Y (2012) Food Antigens. In: James JM, Burks W, Eigenmann P (eds) Food Allergy. W.B. Saunders, Edinburgh, pp 15–32. https://doi.org/10.1016/B978-1-4377-1992-5.00002-8
Liu R, Holck AL, Yang E, Liu C, Xue W (2013) Tropomyosin from tilapia (Oreochromis mossambicus) as an allergen. Clin Exp Allergy 43:365–377. https://doi.org/10.1111/cea.12056
González-Fernández J, Alguacil-Guillén M, Cuéllar C, Daschner A (2018) Possible allergenic role of tropomyosin in patients with adverse reactions after fish intake. Immunol Invest 47:416–429. https://doi.org/10.1080/08820139.2018.1451882
Ruethers T, Taki AC, Karnaneedi S, Nie S, Kalic T, Dai D, Daduang S, Leeming M, Williamson NA, Breiteneder H, Mehr SS, Kamath SD, Campbell DE, Lopata AL (2020) Expanding the allergen repertoire of salmon and catfish. Allergy (in press). https://doi.org/10.1111/all.14574
Broekman H, Verhoeckx KC, den Hartog Jager CF, Kruizinga AG, Pronk-Kleinjan M, Remington BC, Bruijnzeel-Koomen CA, Houben GF, Knulst AC (2016) Majority of shrimp-allergic patients are allergic to mealworm. J Allergy Clin Immunol 137:1261–1263. https://doi.org/10.1016/j.jaci.2016.01.005
Hauser M, Roulias A, Ferreira F, Egger M (2010) Panallergens and their impact on the allergic patient. Allergy Asthma Clin Immunol 6:1–14. https://doi.org/10.1186/1710-1492-6-1
Rahman AMA, Kamath S, Lopata AL, Helleur RJ (2010) Analysis of the allergenic proteins in black tiger prawn (Penaeus monodon) and characterization of the major allergen tropomyosin using mass spectrometry. Rapid Commun Mass Spectrom 24:2462–2470. https://doi.org/10.1002/rcm.4664
Moral L, Toral T (2016) Sensitisation to mites and other animal-derived home aeroallergens in children and its concordance as a measure of covariation of sensitisation. Allergol Immunopathol 44:427–432. https://doi.org/10.1016/j.aller.2016.02.004
Remington BC, Westerhout J, Meima MY, Blom WM, Kruizinga AG, Wheeler MW, Taylor SL, Houben GF, Baumert JL (2020) Updated population minimal eliciting dose distributions for use in risk assessment of 14 priority food allergens. Food Chem Toxicol 139:111259. https://doi.org/10.1016/j.fct.2020.111259
Westerhout J, Baumert JL, Blom WM, Allen KJ, Ballmer-Weber B, Crevel RWR, Dubois AEJ, Fernández-Rivas M, Greenhawt MJ, Hourihane JOB, Koplin JJ, Kruizinga AG, Le T-M, Sampson HA, Shreffler WG, Turner PJ, Taylor SL, Houben GF, Remington BC (2019) Deriving individual threshold doses from clinical food challenge data for population risk assessment of food allergens. J Allergy Clin Immunol 144:1290–1309. https://doi.org/10.1016/j.jaci.2019.07.046
Ballmer-Weber BK, Fernandez-Rivas M, Beyer K, Defernez M, Sperrin M, Mackie AR, Salt LJ, Hourihane JOB, Asero R, Belohlavkova S, Kowalski M, de Blay F, Papadopoulos NG, Clausen M, Knulst AC, Roberts G, Popov T, Sprikkelman AB, Dubakiene R, Vieths S, van Ree R, Crevel R, Mills ENC (2015) How much is too much? Threshold dose distributions for 5 food allergens. J Allergy Clin Immunol 135:964–971. https://doi.org/10.1016/j.jaci.2014.10.047
Matricardi PM, Kleine-Tebbe J, Hoffmann HJ, Valenta R, Hilger C, Hofmaier S, Aalberse RC, Agache I, Asero R, Ballmer-Weber B, Barber D, Beyer K, Biedermann T, Bilò MB, Blank S, Bohle B, Bosshard PP, Breiteneder H, Brough HA, Caraballo L, Caubet JC, Crameri R, Davies JM, Douladiris N, Ebisawa M, Eigenmann PA, Fernandez-Rivas M, Ferreira F, Gadermaier G, Glatz M, Hamilton RG, Hawranek T, Hellings P, Hoffmann-Sommergruber K, Jakob T, Jappe U, Jutel M, Kamath SD, Knol EF, Korosec P, Kuehn A, Lack G, Lopata AL, Mäkelä M, Morisset M, Niederberger V, Nowak-Węgrzyn AH, Papadopoulos NG, Pastorello EA, Pauli G, Platts-Mills T, Posa D, Poulsen LK, Raulf M, Sastre J, Scala E, Schmid JM, Schmid-Grendelmeier P, Hage M, Ree R, Vieths S, Weber R, Wickman M, Muraro A, Ollert M (2016) EAACI molecular allergology user’s guide. Pediatr Allergy Immunol 27:1–250. https://doi.org/10.1111/pai.12563
Stephen JN, Sharp MF, Ruethers T, Taki A, Campbell DE, Lopata AL (2017) Allergenicity of bony and cartilaginous fish – molecular and immunological properties. Clin Exp Allergy 47:300–312. https://doi.org/10.1111/cea.12892
Wopfner N, Dissertori O, Ferreira F, Lackner P (2007) Calcium-binding proteins and their role in allergic diseases. Immunol Allerg Clin North Am 27:29–44. https://doi.org/10.1016/j.iac.2006.10.003
Griesmeier U, Vázquez-Cortés S, Bublin M, Radauer C, Ma Y, Briza P, Fernández-Rivas M, Breiteneder H (2010) Expression levels of parvalbumins determine allergenicity of fish species. Allergy 65:191–198. https://doi.org/10.1111/j.1398-9995.2009.02162.x
Kuehn A, Swoboda I, Arumugam K, Hilger C, Hentges F (2014) Fish allergens at a glance: variable allergenicity of parvalbumins, the major fish allergens. Front Immunol 5:179. https://doi.org/10.3389/fimmu.2014.00179
Kuehn A, Scheuermann T, Hilger C, Hentges F (2010) Important variations in parvalbumin content in common fish species: a factor possibly contributing to variable allergenicity. Int Arch Allergy Immunol 153:359–366. https://doi.org/10.1159/000316346
Jenkins JA, Breiteneder H, Mills ENC (2007) Evolutionary distance from human homologs reflects allergenicity of animal food proteins. J Allergy Clin Immunol 120:1399–1405. https://doi.org/10.1016/j.jaci.2007.08.019
Swain AL, Kretsinger RH, Amma EL (1989) Restrained least squares refinement of native (calcium) and cadmium-substituted carp parvalbumin using X-ray crystallographic data at 1.6-A resolution. J Biol Chem 264:16620–16628. https://www.jbc.org/content/264/28/16620
Taylor SL, Hefle SL, Bindslev-Jensen C, Atkins FM, Andre C, Bruijnzeel-koomen C, Burks AW, Bush RK, Ebisawa M, Eigenmann PA, Host A, Hourihane JOB, Isolauri E, Hill DJ, Knulst A, Lack G, Sampson HA, Moneret-Vautrin DA, Rance F, Vadas PA, Yunginger JW, Zeiger RS, Salminen JW, Madsen C, Abbott P (2004) A consensus protocol for the determination of the threshold doses for allergenic foods: how much is too much? Clin Exp Allergy 34:689–695. https://doi.org/10.1111/j.1365-2222.2004.1886.x
Van Do T, Elsayed S, Florvaag E, Hordvik I, Endresen C (2005) Allergy to fish parvalbumins: studies on the cross-reactivity of allergens from 9 commonly consumed fish. J Allergy Clin Immunol 116:1314–1320. https://doi.org/10.1016/j.jaci.2005.07.033
Ruethers T, Raith M, Sharp MF, Koeberl M, Stephen JN, Nugraha R, Le TTK, Quirce S, Nguyen HXM, Kamath SD, Mehr SS, Campbell DE, Bridges CR, Taki AC, Swoboda I, Lopata AL (2018) Characterization of Ras k 1 a novel major allergen in Indian mackerel and identification of parvalbumin as the major fish allergen in 33 Asia-Pacific fish species. Clin Exp Allergy 48:452–463. https://doi.org/10.1111/cea.13069
Bugajska-Schretter A, Grote M, Vangelista L, Valent P, Sperr WR, Rumpold H, Pastore A, Reichelt R, Valenta R, Spitzauer S (2000) Purification, biochemical, and immunological characterisation of a major food allergen: different immunoglobulin E recognition of the apo- and calcium-bound forms of carp parvalbumin. Gut 46:661–669. https://doi.org/10.1136/gut.46.5.661
Kalic T, Morel-Codreanu F, Radauer C, Ruethers T, Taki AC, Swoboda I, Hilger C, Hoffmann-Sommergruber K, Ollert M, Hafner C, Lopata AL, Morisset M, Breiteneder H, Kuehn A (2019) Patients allergic to fish tolerate ray based on the low allergenicity of its parvalbumin. J Allergy Clin Immunol 7:500-508.e511. https://doi.org/10.1016/j.jaip.2018.11.011
Kuehn A, Codreanu-Morel F, Lehners-Weber C, Doyen V, Gomez-André SA, Bienvenu F, Fischer J, Ballardini N, Hage M, Perotin JM, Silcret-Grieu S, Chabane H, Hentges F, Ollert M, Hilger C, Morisset M (2016) Cross-reactivity to fish and chicken meat – a new clinical syndrome. Allergy 71:1772–1781. https://doi.org/10.1111/all.12968
Ballardini N, Nopp A, Hamsten C, Vetander M, Melén E, Nilsson C, Ollert M, Flohr C, Kuehn A, van Hage M (2017) Anaphylactic reactions to novel foods: case report of a child with severe crocodile meat allergy. Pediatrics 139:e20161404. https://doi.org/10.1542/peds.2016-1404
Hilger C, Grigioni F, Thill L, Mertens L, Hentges F (2002) Severe IgE-mediated anaphylaxis following consumption of fried frog legs: definition of α-parvalbumin as the allergen in cause. Allergy 57:1053–1058. https://doi.org/10.1034/j.1398-9995.2002.23677.x
Kuehn A, Lehners C, Hilger C, Hentges F (2009) Food allergy to chicken meat with IgE reactivity to muscle α-parvalbumin. Allergy 64:1557–1558. https://doi.org/10.1111/j.1398-9995.2009.02094.x
Aas K, Elsayed SM (1969) Characterization of a major allergen (cod). Effect of enzymic hydrolysis on the allergenic activity. J Allergy 44:333–343. https://doi.org/10.1016/0021-8707(69)90025-2
Aas K, Lundkvist U (1973) The radioallergosorbent test with a purified allergen from codfish. Clin Exp Allergy 3:255–261. https://doi.org/10.1111/j.1365-2222.1973.tb01331.x
Elsayed SM, Aas K (1970) Characterization of a major allergen (Cod). Chemical composition and immunological properties. Int Arch Allergy Immunol 38:536–548. https://doi.org/10.1159/000230307
Kuehn A, Hutt-Kempf E, Hilger C, Hentges F (2011) Clinical monosensitivity to salmonid fish linked to specific IgE-epitopes on salmon and trout beta-parvalbumins. Allergy 66:299–301. https://doi.org/10.1111/j.1398-9995.2010.02463.x
Das Dores S, Chopin C, Villaume C, Fleurence J, Guéant JL (2002) A new oligomeric parvalbumin allergen of Atlantic cod (Gad mI) encoded by a gene distinct from that of Gad cI. Allergy 57:79–83. https://doi.org/10.1034/j.1398-9995.57.s72.1.x|
Rosmilah M, Shahnaz M, Masita A, Noormalin A, Jamaludin M (2005) Identification of major allergens of two species of local snappers: Lutjanus argentimaculatus (merah/red snapper) and Lutjanus johnii (jenahak/golden snapper). Trop Biomed 22:171–177
Untersmayr E, Jensen-Jarolim E (2008) The role of protein digestibility and antacids on food allergy outcomes. J Allergy Clin Immunol 121:1301–1308. https://doi.org/10.1016/j.jaci.2008.04.025
Carral CP, Martín-Lázaro J, Ledesma A, de la Torre F (2010) Occupational asthma caused by turbot allergy in 3 fish-farm workers. J Investig Allergol Clin Immunol 20:349–351. http://www.jiaci.org/issues/vol20issue4/vol20issue04-11.htm
Jeebhay MF, Cartier A (2010) Seafood workers and respiratory disease: an update. Curr Opin Allergy Clin Immunol 10:104–113. https://doi.org/10.1097/ACI.0b013e3283373bd0
Strong SJ, Ellington WR (1995) Isolation and sequence analysis of the gene for arginine kinase from the chelicerate arthropod, Limulus polyphemus: insights into catalytically important residues. Biochim Biophys Acta - Protein Struct Molec Enzym 1246:197–200. https://doi.org/10.1016/0167-4838(94)00218-6
Azzi A, Clark SA, Ellington WR, Chapman MS (2004) The role of phosphagen specificity loops in arginine kinase. Protein Sci 13:575–585. https://doi.org/10.1110/ps.03428304
Yang Y, Liu G-Y, Yang H, Hu M-J, Cao M-J, Su W-J, Jin T, Liu G-M (2019) Crystal structure determination of Scylla paramamosain arginine kinase, an allergen that may cause cross-reactivity among invertebrates. Food Chem 271:597–605. https://doi.org/10.1016/j.foodchem.2018.08.003
Ayuso R, Sánchez-Garcia S, Lin J, Fu Z, Ibáñez MD, Carrillo T, Blanco C, Goldis M, Bardina L, Sastre J, Sampson HA (2010) Greater epitope recognition of shrimp allergens by children than by adults suggests that shrimp sensitization decreases with age. J Allergy Clin Immunol 125:1286-1293.e1283. https://doi.org/10.1016/j.jaci.2010.03.010
Bauermeister K, Wangorsch A, Garoffo LP, Reuter A, Conti A, Taylor SL, Lidholm J, DeWitt ÅM, Enrique E, Vieths S, Holzhauser T, Ballmer-Weber B, Reese G (2011) Generation of a comprehensive panel of crustacean allergens from the North Sea shrimp Crangon crangon. Mol Immunol 48:1983–1992. https://doi.org/10.1016/j.molimm.2011.06.216
Yu C-J, Lin Y-F, Chiang B-L, Chow L-P (2003) Proteomics and immunological analysis of a novel shrimp allergen, Pen m 2. J Immunol 170:445–453. https://doi.org/10.4049/jimmunol.170.1.445
Sookrung N, Chaicumpa W, Tungtrongchitr A, Vichyanond P, Bunnag C, Ramasoota P, Tongtawe P, Sakolvaree Y, Tapchaisri P (2006) Periplaneta americana arginine kinase as a major cockroach allergen among Thai patients with major cockroach allergies. Environ Health Perspect 114:875–880. https://doi.org/10.1289/ehp.8650
Hales BJ, Martin AC, Pearce LJ, Laing IA, Hayden CM, Goldblatt J, Le Souëf PN, Thomas WR (2006) IgE and IgG anti-house dust mite specificities in allergic disease. J Allergy Clin Immunol 118:361–367. https://doi.org/10.1016/j.jaci.2006.04.001
Giuffrida MG, Villalta D, Mistrello G, Amato S, Asero R (2014) Shrimp allergy beyond Tropomyosin in Italy: clinical relevance of Arginine Kinase, Sarcoplasmic calcium binding protein and Hemocyanin. Eur Ann Allergy Clin Immunol 46:172–177. https://pdfs.semanticscholar.org/8bbe/3555bcd4198868baf4552459c98593fdc368.pdf
Monaci L, Tregoat V, van Hengel AJ, Anklam E (2006) Milk allergens, their characteristics and their detection in food: a review. Eur Food Res Technol 223:149–179. https://doi.org/10.1007/s00217-005-0178-8
Restani P, Ballabio C, Di Lorenzo C, Tripodi S, Fiocchi A (2009) Molecular aspects of milk allergens and their role in clinical events. Anal Bioanal Chem 395:47–56. https://doi.org/10.1007/s00216-009-2909-3
Fox P (2001) Milk proteins as food ingredients. Int J Dairy Technol 54:41–55. https://doi.org/10.1046/j.1471-0307.2001.00014.x
Barłowska J, Szwajkowska M, Litwińczuk Z, Król J (2011) Nutritional value and technological suitability of milk from various animal species used for dairy production. Compr Rev Food Sci Food Saf 10:291–302. https://doi.org/10.1111/j.1541-4337.2011.00163.x
Sood SM, Herbert PJ, Slatter CW (1997) Structural studies on casein micelles of human milk: dissociation of β-casein of different phosphorylation levels induced by cooling and ethylenediaminetetraacetate. J Dairy Sci 80:628–633. https://doi.org/10.3168/jds.S0022-0302(97)75980-0
Zicarelli L (2004) Buffalo milk: its properties, dairy yield and mozzarella production. Vet Res Commun 28:127–135. https://doi.org/10.1023/B:VERC.0000045390.81982.4d
Hinz K, O’Connor PM, Huppertz T, Ross RP, Kelly AL (2012) Comparison of the principal proteins in bovine, caprine, buffalo, equine and camel milk. J Dairy Res 79:185–191. https://doi.org/10.1017/S0022029912000015
Ehlayel MS, Hazeima KA, Al-Mesaifri F, Bener A (2011) Camel milk: an alternative for cow’s milk allergy in children. Allergy Asthma Proc 32:255–258. https://doi.org/10.2500/aap.2011.32.3429
Restani P, Gaiaschi A, Plebani A, Beretta B, Cavagni G, Fiocchi A, Poiesi C, Velonà T, Ugazio AG, Galli CL (1999) Cross-reactivity between milk proteins from different animal species. Clin Exp Allergy 29:997–1004. https://doi.org/10.1046/j.1365-2222.1999.00563.x
Bernard H (1999) IgE cross-reactivity with caseins from different species in humans allergic to cow’s milk. Food Agric Immunol 11:101–111. https://doi.org/10.1080/09540109999960
Chruszcz M, Mikolajczak K, Mank N, Majorek KA, Porebski PJ, Minor W (2013) Serum albumins - unusual allergens. Biochim Biophys Acta 1830:5375–5381. https://doi.org/10.1016/j.bbagen.2013.06.016
Majorek KA, Porebski PJ, Dayal A, Zimmerman MD, Jablonska K, Stewart AJ, Chruszcz M, Minor W (2012) Structural and immunologic characterization of bovine, horse, and rabbit serum albumins. Mol Immunol 52:174–182. https://doi.org/10.1016/j.molimm.2012.05.011
Choi G-S, Kim J-H, Lee H-N, Sung J-M, Lee J-W, Park H-S (2009) Occupational asthma caused by inhalation of bovine serum albumin powder. Allergy Asthma Immunol Res 1:45–47. https://doi.org/10.4168/aair.2009.1.1.45
Voltolini S, Spigno F, Cioè A, Cagnati P, Bignardi D, Minale P (2013) Bovine serum albumin: A double allergy risk. Eur Ann Allergy Clin Immunol 45:144–147. http://www.eurannallergyimm.com/cont/journals-articles/84/volume-bovine-serum-albumin-double-allergy.asp
Liccardi G, Asero R, D’Amato M, D’Amato G (2011) Role of sensitization to mammalian serum albumin in allergic disease. Curr Allergy Asthma Rep 11:421–426. https://doi.org/10.1007/s11882-011-0214-7
Martelli A, De Chiara A, Corvo M, Restani P, Fiocchi A (2002) Beef allergy in children with cow’s milk allergy; cow’s milk allergy in children with beef allergy. Ann Allergy Asthma Immunol 89:38–43. https://doi.org/10.1016/S1081-1206(10)62121-7
Posthumus J, James HR, Lane CJ, Matos LA, Platts-Mills TAE, Commins SP (2013) Initial description of pork-cat syndrome in the United States. J Allergy Clin Immunol 131:923–925. https://doi.org/10.1016/j.jaci.2012.12.665
Hilger C, Kohnen M, Grigioni F, Lehners C, Hentges F (1997) Allergic cross-reactions between cat and pig serum albumin. Allergy 52:179–187. https://doi.org/10.1111/j.1398-9995.1997.tb00972.x
Quirce S, Marañón F, Umpiérrez A, De Las Heras M, Fernández-Caldas E, Sastre J (2001) Chicken serum albumin (Gal d 5*) is a partially heat-labile inhalant and food allergen implicated in the bird-egg syndrome. Allergy 56:754–762. https://doi.org/10.1034/j.1398-9995.2001.056008754.x|
Henrissat B, Callebaut I, Fabrega S, Lehn P, Mornon JP, Davies G (1995) Conserved catalytic machinery and the prediction of a common fold for several families of glycosyl hydrolases. Proc Natl Acad Sci USA 92:7090–7094. https://doi.org/10.1073/pnas.92.15.7090
Mine Y, Rupa P (2004) Immunological and biochemical properties of egg allergens. Worlds Poult Sci J 60:321–330. https://doi.org/10.1079/WPS200420
Lesnierowski G, Kijowski J (2007) Lysozyme. In: Huopalahti R, López-Fandiño R, Anton M, Schade R (eds) Bioactive Egg Compounds. Springer, Berlin, Heidelberg, pp 33–42. https://doi.org/10.1007/978-3-540-37885-3_6
Blake CCF, Koenig DF, Mair GA, North ACT, Phillips DC, Sarma VR (1965) Structure of Hen egg-white lysozyme: a three-dimensional fourier synthesis at 2 Å resolution. Nature 206:757–761. https://doi.org/10.1038/206757a0
Young ACM, Tilton RF, Dewan JC (1994) Thermal expansion of hen egg-white lysozyme: comparison of the 1.9 Å resolution structures of the tetragonal form of the enzyme at 100 K and 298 K. J Mol Biol 235:302–317. https://doi.org/10.1016/S0022-2836(05)80034-8
Geng F, Wang J, Liu D, Jin Y, Ma M (2017) Identification of N-glycosites in chicken egg white proteins using an Omics strategy. J Agric Food Chem 65:5357–5364. https://doi.org/10.1021/acs.jafc.7b01706
Asperger A, Marx K, Albers C, Molin L, Pinato O (2015) Low abundant N-linked glycosylation in hen egg white lysozyme is localized at nonconsensus sites. J Proteome Res 14:2633–2641. https://doi.org/10.1021/acs.jproteome.5b00175
Escudero C, Quirce S, Fernández-Nieto M, de Miguel J, Cuesta J, Sastre J (2003) Egg white proteins as inhalant allergens associated with baker’s asthma. Allergy 58:616–620. https://doi.org/10.1034/j.1398-9995.2003.00201.x
Pérez-Calderón R, Gonzalo-Garijo M, Lamilla-Yerga A, Mangas-Santos R, Moreno-Gastón I (2007) Recurrent angioedema due to lysozyme allergy J Investig Allergol Clin Immunol 17:264–266. http://www.jiaci.org/summary/vol17-issue4-num241
Stuart DI, Acharya KR, Walker NPC, Smith SG, Lewis M, Phillips DC (1986) α-Lactalbumin possesses a novel calcium binding loop. Nature 324:84–87. https://doi.org/10.1038/324084a0
Permyakov EA, Berliner LJ (2000) α-Lactalbumin: structure and function. FEBS Lett 473:269–274. https://doi.org/10.1016/S0014-5793(00)01546-5
Hochwallner H, Schulmeister U, Swoboda I, Spitzauer S, Valenta R (2014) Cow’s milk allergy: from allergens to new forms of diagnosis, therapy and prevention. Methods 66:22–33. https://doi.org/10.1016/j.ymeth.2013.08.005
Hochwallner H, Schulmeister U, Swoboda I, Focke-Tejkl M, Civaj V, Balic N, Nystrand M, Härlin A, Thalhamer J, Scheiblhofer S (2010) Visualization of clustered IgE epitopes on α-lactalbumin. J Allergy Clin Immunol 125:1279-1285 e1279. https://doi.org/10.1016/j.jaci.2010.03.007
Shoormasti RS, Fazlollahi M, Barzegar S, Teymourpour P, Yazdanyar Z, Lebaschi Z, Nourizadeh M, Tazesh B, Movahedi M, Kashani H, Pourpak Z, Moin M (2016) The most common cow’s milk allergenic proteins with respect to allergic symptoms in Iranian patients. Iran J Allergy Asthma Immunol 15:161–165. https://ijaai.tums.ac.ir/index.php/ijaai/article/view/686
Aisen P, Listowsky I (1980) Iron transport and storage proteins. Ann Rev Biochem 49:357–393. https://doi.org/10.1146/annurev.bi.49.070180.002041
Lambert LA, Perri H, Halbrooks PJ, Mason AB (2005) Evolution of the transferrin family: conservation of residues associated with iron and anion binding. Comp Biochem Physiol B-Biochem Mol Biol 142:129–141. https://doi.org/10.1016/j.cbpb.2005.07.007
Kurokawa H, Mikami B, Hirose M (1995) Crystal structure of diferric hen ovotransferrin at 2.4 Å resolution. J Mol Biol 254:196–207. https://doi.org/10.1006/jmbi.1995.0611
Ibrahim HR (2000) Ovotransferrin. In: Naidu AS (ed) Natural food antimicrobial systems. CRC Press, Boca Raton, FL, pp 211–226
Williams J, Elleman TC, Kingston IB, Wilkins AG, Kuhn KA (1982) The primary structure of hen ovotransferrin. Eur J Biochem 122:297–303. https://doi.org/10.1111/j.1432-1033.1982.tb05880.x
Kim J, Lee J, Park M-R, Han Y, Shin M, Ahn K (2014) Special consideration is required for the component-resolved diagnosis of egg allergy in infants. Ann Allergy Asthma Immunol 112:53–57. https://doi.org/10.1016/j.anai.2013.09.010
Baker EN, Baker HM (2005) Lactoferrin. Cell Mol Life Sci 62:2531. https://doi.org/10.1007/s00018-005-5368-9
Pakdaman R, Petitjean M, El Hage Chahine J-M (1998) Transferrins. Eur J Biochem 254:144–153. https://doi.org/10.1046/j.1432-1327.1998.2540144.x
Gaudin J-C, Rabesona H, Choiset Y, Yeretssian G, Chobert J-M, Sakanyan V, Drouet M, Haertlé T (2008) Assessment of the immunoglobulin E-mediated immune response to milk-specific proteins in allergic patients using microarrays. Clin Exp Allergy 38:686–693. https://doi.org/10.1111/j.1365-2222.2008.02952.x
Ganfornina MD, Sanchez D, Greene LH, Flower DR (2006) The lipocalin protein family: protein sequence, structure and relationship to the calycin superfamily. In: Åkerstrom B, Borregaard N, Flower D, Salier JP (eds) Lipocalins. Landes Bioscience, Georgetown, pp 17–27. https://doi.org/10.1201/9781498712736
Grzyb J, Latowski D, Strzałka K (2006) Lipocalins - a family portrait. J Plant Physiol 163:895–915. https://doi.org/10.1016/j.jplph.2005.12.007
Virtanen T, Kinnunen T, Rytkönen-Nissinen M (2012) Mammalian lipocalin allergens - insights into their enigmatic allergenicity. Clin Exp Allergy 42:494–504. https://doi.org/10.1111/j.1365-2222.2011.03903.x
Rouvinen J, Virtanen T, Mäntyjärvi R (2001) Search for the determinants of allergenicity in proteins of the lipocalin family. J Chromatogr B: Biomed Sci Appl 756:199–206. https://doi.org/10.1016/S0378-4347(01)00109-8
Hilger C, Kuehn A, Hentges F (2012) Animal lipocalin allergens. Curr Allergy Asthma Rep 12:438–447. https://doi.org/10.1007/s11882-012-0283-2
Weng Y-C, Wang G, Messing RO, Chou W-H (2015) Identification of lipocalin-2 as a PKCδ phosphorylation substrate in neutrophils. J Biomed Sci 22:21. https://doi.org/10.1186/s12929-015-0129-z
Hilger C, Swiontek K, Arumugam K, Lehners C, Hentges F (2012) Identification of a new major dog allergen highly cross-reactive with Fel d 4 in a population of cat- and dog-sensitized patients. J Allergy Clin Immunol 129:1149-1151.e1142. https://doi.org/10.1016/j.jaci.2011.10.017
Nilsson OB, Binnmyr J, Zoltowska A, Saarne T, van Hage M, Grönlund H (2012) Characterization of the dog lipocalin allergen Can f 6: the role in cross-reactivity with cat and horse. Allergy 67:751–757. https://doi.org/10.1111/j.1398-9995.2012.02826.x
Flower DR, North ACT, Sansom CE (2000) The lipocalin protein family: structural and sequence overview. Biochim Biophys Acta-Protein Struct Molec Enzym 1482:9–24. https://doi.org/10.1016/S0167-4838(00)00148-5
Lakshmi B, Mishra M, Srinivasan N, Archunan G (2015) Structure-based phylogenetic analysis of the Lipocalin superfamily. PLoS ONE 10:e0135507. https://doi.org/10.1371/journal.pone.0135507
Virtanen T (2001) Lipocalin allergens. Allergy 56:48–51. https://doi.org/10.1034/j.1398-9995.2001.00915.x
Jensen-Jarolim E, Pacios LF, Bianchini R, Hofstetter G, Roth-Walter F (2016) Structural similarities of human and mammalian lipocalins, and their function in innate immunity and allergy. Allergy 71:286–294. https://doi.org/10.1111/all.12797
Bello M, Fragoso-Vázquez MJ, Correa Basurto J (2016) Energetic and conformational features linked to the monomeric and dimeric states of bovine BLG. Int J Biol Macromol 92:625–636. https://doi.org/10.1016/j.ijbiomac.2016.07.071
Järvinen K-M, Chatchatee P, Bardina L, Beyer K, Sampson HA (2001) IgE and IgG binding epitopes on α-lactalbumin and β-lactoglobulin in cow’s milk allergy. Int Arch Allergy Immunol 126:111–118. https://doi.org/10.1159/000049501
Rawlings ND, Barrett AJ, Thomas PD, Huang X, Bateman A, Finn RD (2018) The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res 46:D624–D632. https://doi.org/10.1093/nar/gkx1134
Laskowski M, Kato I (1980) Protein inhibitors of proteinases. Ann Rev Biochem 49:593–626. https://doi.org/10.1146/annurev.bi.49.070180.003113
Rawlings ND, Tolle DP, Barrett AJ (2004) Evolutionary families of peptidase inhibitors. Biochem J 378:705–716. https://doi.org/10.1042/bj20031825
Kato I, Schrode J, Kohr WJ, Laskowski M (1987) Chicken ovomucoid: determination of its amino acid sequence, determination of the trypsin reactive site, and preparation of all three of its domains. Biochemistry 26:193–201. https://doi.org/10.1021/bi00375a027
Rupa P, Nakamura S, Mine Y (2007) Genetically glycosylated ovomucoid third domain can modulate immunoglobulin E antibody production and cytokine response in BALB/c mice. Clin Exp Allergy 37:918–928. https://doi.org/10.1111/j.1365-2222.2007.02720.x
Matsuda T, Nakamura R, Nakashima I, Hasegawa Y, Shimokata K (1985) Human IgE antibody to the carbohydrate-containing third domain of chicken ovomucoid. Biochem Biophys Res Commun 129:505–510. https://doi.org/10.1016/0006-291X(85)90180-9
Caubet J-C, Wang J (2011) Current understanding of egg allergy. Pediatr Clin North Am 58:427–443. https://doi.org/10.1016/j.pcl.2011.02.014
Tan JW-L, Campbell DE, Turner PJ, Kakakios A, Wong M, Mehr S, Joshi P (2013) Baked egg food challenges - clinical utility of skin test to baked egg and ovomucoid in children with egg allergy. Clin Exp Allergy 43:1189–1195. https://doi.org/10.1111/cea.12153
Irving JA, Pike RN, Lesk AM, Whisstock JC (2000) Phylogeny of the serpin superfamily: implications of patterns of amino acid conservation for structure and function. Genome Res 10:1845–1864. https://doi.org/10.1101/gr.147800
Law RH, Zhang Q, McGowan S, Buckle AM, Silverman GA, Wong W, Rosado CJ, Langendorf CG, Pike RN, Bird PI, Whisstock JC (2006) An overview of the serpin superfamily. Genome Biol 7:216. https://doi.org/10.1186/gb-2006-7-5-216
Nisbet AD, Saundry RH, Moir AJG, Fothergill LA, Fothergill JE (1981) The complete amino-acid sequence of hen ovalbumin. Eur J Biochem 115:335–345. https://doi.org/10.1111/j.1432-1033.1981.tb05243.x
An HJ, Peavy TR, Hedrick JL, Lebrilla CB (2003) Determination of N-glycosylation sites and site heterogeneity in glycoproteins. Anal Chem 75:5628–5637. https://doi.org/10.1021/ac034414x
Stein PE, Leslie AGW, Finch JT, Carrell RW (1991) Crystal structure of uncleaved ovalbumin at 1.95 Å resolution. J Mol Biol 221:941–959. https://doi.org/10.1016/0022-2836(91)80185-W
Lin Y-T, Wu C-T, Huang J-L, Cheng J-H, Yeh K-W (2016) Correlation of ovalbumin of egg white components with allergic diseases in children. J Microbiol Immunol Infect 49:112–118. https://doi.org/10.1016/j.jmii.2014.01.002
Pelz BJ, Bryce PJ (2015) Pathophysiology of food allergy. Pediatr Clin N Am 62:1363–1375. https://doi.org/10.1016/j.pcl.2015.07.004
Remington B, Broekman HCH, Blom WM, Capt A, Crevel RWR, Dimitrov I, Faeste CK, Fernandez-Canton R, Giavi S, Houben GF, Glenn KC, Madsen CB, Kruizinga AK, Constable A (2018) Approaches to assess IgE mediated allergy risks (sensitization and cross-reactivity) from new or modified dietary proteins. Food Chem Toxicol 112:97–107. https://doi.org/10.1016/j.fct.2017.12.025
Yuan F, Lv L, Li Z, Mi N, Chen H, Lin H (2017) Effect of transglutaminase-catalyzed glycosylation on the allergenicity and conformational structure of shrimp (Metapenaeus ensis) tropomyosin. Food Chem 219:215–222. https://doi.org/10.1016/j.foodchem.2016.09.139
Ayuso R, Lehrer SB, Reese G (2002) Identification of continuous, allergenic regions of the major shrimp allergen Pen a 1 (tropomyosin). Int Arch Allergy Immunol 127:27–37. https://doi.org/10.1159/000048166
Carnés J, Ferrer Á, Huertas ÁJ, Andreu C, Larramendi CH, Fernández-Caldas E (2007) The use of raw or boiled crustacean extracts for the diagnosis of seafood allergic individuals. Ann Allergy Asthma Immunol 98:349–354. https://doi.org/10.1016/S1081-1206(10)60881-2
Sánchez R, Martínez J, Castro A, Pedrosa M, Quirce S, Rodríguez-Pérez R, Gasset M (2016) The amyloid fold of Gad m 1 epitopes governs IgE binding. Sci Rep 6:32801. https://doi.org/10.1038/srep32801
Swoboda I, Bugajska-Schretter A, Verdino P, Keller W, Sperr WR, Valent P, Valenta R, Spitzauer S (2002) Recombinant carp parvalbumin, the major cross-reactive fish allergen: a tool for diagnosis and therapy of fish allergy. J Immunol 168:4576–4584. https://doi.org/10.4049/jimmunol.168.9.4576
Shen H-W, Cao M-J, Cai Q-F, Ruan M-M, Mao H-Y, Su W-J, Liu G-M (2012) Purification, cloning, and immunological characterization of arginine kinase, a novel allergen of Octopus fangsiao. J Agric Food Chem 60:2190–2199. https://doi.org/10.1021/jf203779w
Benedé S, López-Expósito I, Giménez G, Grishina G, Bardina L, Sampson HA, Molina E, López-Fandiño R (2014) In vitro digestibility of bovine β-casein with simulated and human oral and gastrointestinal fluids. Identification and IgE-reactivity of the resultant peptides. Food Chem 143:514–521. https://doi.org/10.1016/j.foodchem.2013.07.110
Stanic D, Monogioudi E, Dilek E, Radosavljevic J, Atanaskovic-Markovic M, Vuckovic O, Raija L, Mattinen M, Buchert J, Cirkovic Velickovic T (2010) Digestibility and allergenicity assessment of enzymatically crosslinked beta-casein. Mol Nutr Food Res 54:1273–1284. https://doi.org/10.1002/mnfr.200900184
Ouahidi I, El Hamsas AEY, Aarab L (2011) Modulation of egg white protein allergenicity under physical and chemical treatments. Food Agric Immunol 22:57–68. https://doi.org/10.1080/09540105.2010.526202
Martos G, López-Fandiño R, Molina E (2013) Immunoreactivity of hen egg allergens: Influence on in vitro gastrointestinal digestion of the presence of other egg white proteins and of egg yolk. Food Chem 136:775–781. https://doi.org/10.1016/j.foodchem.2012.07.106
Shin M, Han Y, Ahn K (2013) The influence of the time and temperature of heat treatment on the allergenicity of egg white proteins. Allergy Asthma Immunol Res 5:96–101. https://doi.org/10.4168/aair.2013.5.2.96
Mine Y, Zhang JW (2002) Comparative studies on antigenicity and allergenicity of native and denatured egg white proteins. J Agric Food Chem 50:2679–2683. https://doi.org/10.1021/jf0112264
Chen Y, Tu Z, Wang H, Zhang L, Sha X, Pang J, Yang P, Liu G, Yang W (2016) Glycation of β-lactoglobulin under dynamic high pressure microfluidization treatment: Effects on IgE-binding capacity and conformation. Food Res Int 89:882–888. https://doi.org/10.1016/j.foodres.2016.10.020
Meng X, Bai Y, Gao J, Li X, Chen H (2017) Effects of high hydrostatic pressure on the structure and potential allergenicity of the major allergen bovine β-lactoglobulin. Food Chem 219:290–296. https://doi.org/10.1016/j.foodchem.2016.09.153
Shin M, Lee J, Ahn K, Lee SI, Han Y (2013) The influence of the presence of wheat flour on the antigenic activities of egg white proteins. Allergy Asthma Immunol Res 5:42–47. https://doi.org/10.4168/aair.2013.5.1.42
Lee J-O, Sung D, Park SH, Lee J, Kim J, Shon D-H, Ahn K, Han Y (2017) Effect of acid treatment on allergenicity of peanut and egg. J Sci Food Agric 97:2116–2121. https://doi.org/10.1002/jsfa.8017
Lee J-W, Seo J-H, Kim J-H, Lee S-Y, Byun M-W (2007) Comparison of the changes of the antigenicities of a hen’s egg albumin by a gamma and an electron beam irradiation. Rad Phys Chem 76:879–885. https://doi.org/10.1016/j.radphyschem.2006.06.010
Martos G, Contreras P, Molina E, López-Fandiño R (2010) Egg white ovalbumin digestion mimicking physiological conditions. J Agric Food Chem 58:5640–5648. https://doi.org/10.1021/jf904538w
Benedé S, López-Expósito I, López-Fandiño R, Molina E (2014) Identification of IgE-binding peptides in hen egg ovalbumin digested in vitro with human and simulated gastroduodenal fluids. J Agric Food Chem 62:152–158. https://doi.org/10.1021/jf404226w
Jiménez-Saiz R, López-Expósito I, Molina E, López-Fandiño R (2013) IgE-binding and in vitro gastrointestinal digestibility of egg allergens in the presence of polysaccharides. Food Hydrocolloids 30:597–605. https://doi.org/10.1016/j.foodhyd.2012.07.014
Wróblewska B, Kaliszewska A (2012) Cow’s milk proteins immunoreactivity and allergenicity in processed food. Czech J Food Sci 30:211–219. https://doi.org/10.17221/525/2010-CJFS
Mazzucchelli G, Holzhauser T, Velickovic TC, Diaz-Perales A, Molina E, Roncada P, Rodrigues P, Verhoeckx K, Hoffmann-Sommergruber K (2018) Current (food) allergenic risk assessment: Is it fit for novel foods? Status quo and identification of gaps. Mol Nutri Food Res 62:1700278. https://doi.org/10.1002/mnfr.201700278
Broekman HCH, Eiwegger T, Upton J, Bøgh KL (2015) IgE - the main player of food allergy. Drug Discov Today Dis Models 17–18:37–44. https://doi.org/10.1016/j.ddmod.2016.07.001
Hemmings O, Kwok M, McKendry R, Santos AF (2018) Basophil activation test: old and new applications in allergy. CurrAllergy Asthma Rep 18:77. https://doi.org/10.1007/s11882-018-0831-5
Santos AF, Brough HA (2017) Making the most of in vitro tests to diagnose food allergy. J Allergy Clin Immunol Pract 5:237–248. https://doi.org/10.1016/j.jaip.2016.12.003
Falcone FH, Alcocer MJC, Okamoto-Uchida Y, Nakamura R (2015) Use of humanized rat basophilic leukemia reporter cell lines as a diagnostic tool for detection of allergen-specific IgE in allergic patients: time for a reappraisal? Curr Allergy Asthma Rep 15:67. https://doi.org/10.1007/s11882-015-0568-3
Zhang Z, Xiao H, Zhang X, Zhou P (2019) Conformation, allergenicity and human cell allergy sensitization of tropomyosin from Exopalaemon modestus: effects of deglycosylation and Maillard reaction. Food Chem 276:520–527. https://doi.org/10.1016/j.foodchem.2018.10.032
Song Y, Li Z, Gao Q, Pavase TR, Lin H (2016) Effect of malonaldehyde cross-linking on the ability of shrimp tropomyosin to elicit the release of inflammatory mediators and cytokines from activated RBL-2H3 cells. J Sci Food Agric 96:4263–4267. https://doi.org/10.1002/jsfa.7637
Gámez C, Zafra MP, Sanz V, Mazzeo C, Ibáñez MD, Sastre J, del Pozo V (2015) Simulated gastrointestinal digestion reduces the allergic reactivity of shrimp extract proteins and tropomyosin. Food Chem 173:475–481. https://doi.org/10.1016/j.foodchem.2014.10.063
Kamath SD, Rahman AMA, Voskamp A, Komoda T, Rolland JM, O’Hehir RE, Lopata AL (2014) Effect of heat processing on antibody reactivity to allergen variants and fragments of black tiger prawn: a comprehensive allergenomic approach. Mol Nutr Food Res 58:1144–1155. https://doi.org/10.1002/mnfr.201300584
Jiménez-Saiz R, Benedé S, Miralles B, López-Expósito I, Molina E, López-Fandiño R (2014) Immunological behavior of in vitro digested egg-white lysozyme. Mol Nutr Food Res 58:614–624. https://doi.org/10.1002/mnfr.201300442
Morisawa Y, Kitamura A, Ujihara T, Zushi N, Kuzume K, Shimanouchi Y, Tamura S, Wakiguchi H, Saito H, Matsumoto K (2009) Effect of heat treatment and enzymatic digestion on the B cell epitopes of cow’s milk proteins. Clin Exp Allergy 39:918–925. https://doi.org/10.1111/j.1365-2222.2009.03203.x
Roth-Walter F, Pacios LF, Gomez-Casado C, Hofstetter G, Roth GA, Singer J, Diaz-Perales A, Jensen-Jarolim E (2014) The major cow milk allergen Bos d 5 manipulates T-helper cells depending on its load with siderophore-bound iron. PLoS ONE 9:e104803. https://doi.org/10.1371/journal.pone.0104803
Stanic-Vucinic D, Stojadinovic M, Atanaskovic-Markovic M, Ognjenovic J, Grönlund H, van Hage M, Lantto R, Sancho AI, Velickovic TC (2012) Structural changes and allergenic properties of β-lactoglobulin upon exposure to high-intensity ultrasound. Mol Nutr Food Res 56:1894–1905. https://doi.org/10.1002/mnfr.201200179
Benedé S, López-Fandiño R, Reche M, Molina E, López-Expósito I (2013) Influence of the carbohydrate moieties on the immunoreactivity and digestibility of the egg allergen ovomucoid. PLoS ONE 8:e80810. https://doi.org/10.1371/journal.pone.0080810
Martos G, Lopez-Exposito I, Bencharitiwong R, Berin MC, Nowak-Węgrzyn A (2011) Mechanisms underlying differential food allergy response to heated egg. J Allergy Clin Immunol 127:990-997.e992. https://doi.org/10.1016/j.jaci.2011.01.057
El Mecherfi KE, Curet S, Lupi R, Larré C, Rouaud O, Choiset Y, Rabesona H, Haertlé T (2019) Combined microwave processing and enzymatic proteolysis of bovine whey proteins: the impact on bovine β-lactoglobulin allergenicity. J Food Sci Technol 56:177–186. https://doi.org/10.1007/s13197-018-3471-9
Huang J, Liu C, Wang Y, Wang C, Xie M, Qian Y, Fu L (2018) Application of in vitro and in vivo models in the study of food allergy. Food Sci Human Wellness 7:235–243. https://doi.org/10.1016/j.fshw.2018.10.002
Cases B, García-Ara C, Boyano M, Pérez-Gordo M, Pedrosa M, Vivanco F, Quirce S, Pastor-Vargas C (2011) Phosphorylation reduces the allergenicity of cow casein in children with selective allergy to goat and sheep milk. J Invest Allergol Clin Immunol 21:398–400. http://www.jiaci.org/issues/vol21issue5/9.pdf
Fiocchi A, Restani P, Riva E (2000) Beef allergy in children. Nutrition 16:454–457. https://doi.org/10.1016/S0899-9007(00)00285-9
Bøgh KL, van Bilsen J, Głogowski R, López-Expósito I, Bouchaud G, Blanchard C, Bodinier M, Smit J, Pieters R, Bastiaan-Net S, de Wit N, Untersmayr E, Adel-Patient K, Knippels L, Epstein MM, Noti M, Nygaard UC, Kimber I, Verhoeckx K, O’Mahony L (2016) Current challenges facing the assessment of the allergenic capacity of food allergens in animal models. Clin Transl Allergy 6:21. https://doi.org/10.1186/s13601-016-0110-2
Gonipeta B, Kim E, Gangur V (2015) Mouse models of food allergy: How well do they simulate the human disorder? Crit Rev Food Sci Nutr 55:437–452. https://doi.org/10.1080/10408398.2012.657807
Han X-Y, Yang H, Rao S-T, Liu G-Y, Hu M-J, Zeng B-C, Cao M-J, Liu G-M (2018) The Maillard reaction reduced the sensitization of tropomyosin and arginine kinase from Scylla paramamosain, simultaneously. J Agric Food Chem 66:2934–2943. https://doi.org/10.1021/acs.jafc.7b05195
Long F, Yang X, Wang R, Hu X, Chen F (2015) Effects of combined high pressure and thermal treatments on the allergenic potential of shrimp (Litopenaeus vannamei) tropomyosin in a mouse model of allergy. Innov Food Sci Emerg Technol 29:119–124. https://doi.org/10.1016/j.ifset.2015.03.002
Pablos-Tanarro A, Lozano-Ojalvo D, Martínez-Blanco M, López-Fandiño R, Molina E (2017) Sensitizing and eliciting capacity of egg white proteins in BALB/c mice as affected by processing. J Agric Food Chem 65:4500–4508. https://doi.org/10.1021/acs.jafc.7b00953
Roth-Walter F, Berin MC, Arnaboldi P, Escalante CR, Dahan S, Rauch J, Jensen-Jarolim E, Mayer L (2008) Pasteurization of milk proteins promotes allergic sensitization by enhancing uptake through Peyer’s patches. Allergy 63:882–890. https://doi.org/10.1111/j.1398-9995.2008.01673.x
Tong P, Gao L, Gao J, Li X, Wu Z, Yang A, Chen H (2017) Iron-induced chelation alleviates the potential allergenicity of ovotransferrin in a BALB/c mouse model. Nutr Res 47:81–89. https://doi.org/10.1016/j.nutres.2017.09.009
López-Expósito I, Chicón R, Belloque J, López-Fandiño R, Berin M (2012) In vivo methods for testing allergenicity show that high hydrostatic pressure hydrolysates of β-lactoglobulin are immunologically inert. J Dairy Sci 95:541–548. https://doi.org/10.3168/jds.2011-4646
Claude M, Bouchaud G, Lupi R, Castan L, Tranquet O, Denery-Papini S, Bodinier M, Brossard C (2017) How proteins aggregate can reduce allergenicity: comparison of ovalbumins heated under opposite electrostatic conditions. J Agric Food Chem 65:3693–3701. https://doi.org/10.1021/acs.jafc.7b00676
Claude M, Lupi R, Bouchaud G, Bodinier M, Brossard C, Denery-Papini S (2016) The thermal aggregation of ovalbumin as large particles decreases its allergenicity for egg allergic patients and in a murine model. Food Chem 203:136–144. https://doi.org/10.1016/j.foodchem.2016.02.054
Hacini-Rachinel F, Vissers YM, Doucet-Ladevéze R, Blanchard C, Demont A, Perrot M, Panchaud A, Prioult G, Mercenier A, Nutten S (2014) Low-allergenic hydrolyzed egg induces oral tolerance in mice. Int Arch Allergy Immunol 164:64–73. https://doi.org/10.1159/000363110
Tong P, Chen S, Gao J, Li X, Wu Z, Yang A, Yuan J, Chen H (2018) Caffeic acid-assisted cross-linking catalyzed by polyphenol oxidase decreases the allergenicity of ovalbumin in a Balb/c mouse model. Food Chem Toxicol 111:275–283. https://doi.org/10.1016/j.fct.2017.11.026
Golias J, Schwarzer M, Wallner M, Kverka M, Kozakova H, Srutkova D, Klimesova K, Sotkovsky P, Palova-Jelinkova L, Ferreira F, Tuckova L (2012) Heat-induced structural changes affect OVA-antigen processing and reduce allergic response in mouse model of food allergy. PLoS ONE 7:e37156. https://doi.org/10.1371/journal.pone.0037156
Meng X, Li X, Gao J, Chen H (2016) Characterization of the potential allergenicity of irradiated bovine α-lactalbumin in a BALB/c mouse model. Food Chem Toxicol 97:402–410. https://doi.org/10.1016/j.fct.2016.10.010
Heilmann M, Wellner A, Gadermaier G, Ilchmann A, Briza P, Krause M, Nagai R, Burgdorf S, Scheurer S, Vieths S, Henle T, Toda M (2014) Ovalbumin modified with pyrraline, a Maillard reaction product, shows enhanced T-cell Immunogenicity. J Biol Chem 289:7919–7928. https://doi.org/10.1074/jbc.M113.523621
Seo J-H, Kim J-H, Lee J-W, Yoo Y-C, Kim MR, Park K-S, Byun M-W (2007) Ovalbumin modified by gamma irradiation alters its immunological functions and allergic responses. Int Immunopharmacol 7:464–472. https://doi.org/10.1016/j.intimp.2006.11.012
Seo J-H, Lee J-W, Kim J-H, Byun E-B, Lee S-Y, Kang I-J, Byun M-W (2007) Reduction of allergenicity of irradiated ovalbumin in ovalbumin-allergic mice. Rad Phys Chem 76:1855–1857. https://doi.org/10.1016/j.radphyschem.2007.02.094
Ahmed I, Lin H, Xu L, Li S, Costa J, Mafra I, Chen G, Gao X, Li Z (2020) Immunomodulatory effect of laccase/caffeic acid and transglutaminase in alleviating shrimp tropomyosin (Met e 1) allergenicity. J Agric Food Chem 68:7765–7778. https://doi.org/10.1021/acs.jafc.0c02366
Fei DX, Liu QM, Chen F, Yang Y, Chen ZW, Cao MJ, Liu GM (2016) Assessment of the sensitizing capacity and allergenicity of enzymatic cross-linked arginine kinase, the crab allergen. Mol Nutr Food Res 60:1707–1718. https://doi.org/10.1002/mnfr.201500936
Yang H, Min J, Han X-Y, Li X-Y, Hu J-W, Liu H, Cao M-J, Liu G-M (2018) Reduction of the histamine content and immunoreactivity of parvalbumin in Decapterus maruadsi by a Maillard reaction combined with pressure treatment. Food Funct 9:4897–4905. https://doi.org/10.1039/C8FO01167B
El Mecherfi K-E, Rouaud O, Curet S, Negaoui H, Chobert J-M, Kheroua O, Saidi D, Haertlé T (2015) Peptic hydrolysis of bovine beta-lactoglobulin under microwave treatment reduces its allergenicity in an ex vivo murine allergy model. Int J Food Sci Technol 50:356–364. https://doi.org/10.1111/ijfs.12653
Stojadinovic M, Pieters R, Smit J, Velickovic TC (2014) Cross-linking of β-lactoglobulin enhances allergic sensitization through changes in cellular uptake and processing. Toxicol Sci 140:224–235. https://doi.org/10.1093/toxsci/kfu062
Fuc E, Złotkowska D, Wróblewska B (2019) Milk and meat allergens from Bos taurus β-lactoglobulin, α-casein, and bovine serum albumin: An in-vivo study of the immune response in mice. Nutrients 11:2095. https://doi.org/10.3390/nu11092095
Benhatchi S, Addou S, Grar H, Benaissa Y, Kheroua O, Saidi D (2019) Induction of sublingual immunotherapy to cow’s milk (raw, pasteurized and sterilized) in Balb/c mice sensitized to beta-lactoglobulin. Revue Française d’Allergologie 59:9–14. https://doi.org/10.1016/j.reval.2018.09.008
Verhoeckx KCM, Vissers YM, Baumert JL, Faludi R, Feys M, Flanagan S, Herouet-Guicheney C, Holzhauser T, Shimojo R, van der Bolt N, Wichers H, Kimber I (2015) Food processing and allergenicity. Food Chem Toxicol 80:223–240. https://doi.org/10.1016/j.fct.2015.03.005
Lee P-W, Nordlee JA, Koppelman SJ, Baumert JL, Taylor SL (2012) Measuring parvalbumin levels in fish muscle tissue: Relevance of muscle locations and storage conditions. Food Chem 135:502–507. https://doi.org/10.1016/j.foodchem.2012.05.030
Somkuti J, Bublin M, Breiteneder H, Smeller L (2012) Pressure-temperature stability, Ca2+ binding, and pressure-temperature phase diagram of cod parvalbumin: Gad m 1. Biochemistry 51:5903–5911. https://doi.org/10.1021/bi300403h
Kobayashi Y, Yang T, Yu C-T, Ume C, Kubota H, Shimakura K, Shiomi K, Hamada-Sato N (2016) Quantification of major allergen parvalbumin in 22 species of fish by SDS-PAGE. Food Chem 194:345–353. https://doi.org/10.1016/j.foodchem.2015.08.037
Vicente-Serrano J, Caballero M, Rodríguez-Pérez R, Carretero P, Perez R, Blanco J, Juste S, Moneo I (2007) Sensitization to serum albumins in children allergic to cow’s milk and epithelia. Pediatr Allergy Immunol 18:503–507. https://doi.org/10.1111/j.1399-3038.2007.00548.x
Pablos-Tanarro A, Lozano-Ojalvo D, Molina E, López-Fandiño R (2018) Assessment of the allergenic potential of the main egg white proteins in BALB/c mice. J Agric Food Chem 66:2970–2976. https://doi.org/10.1021/acs.jafc.8b00402
Bogahawaththa D, Ashraf R, Chandrapala J, Donkor O, Vasiljevic T (2018) In vitro immunogenicity of various native and thermally processed bovine milk proteins and their mixtures. J Dairy Sci 101:8726–8736. https://doi.org/10.3168/jds.2018-14488
Järvinen KM, Chatchatee P (2009) Mammalian milk allergy: clinical suspicion, cross-reactivities and diagnosis. Curr Opin Allergy Clin Immunol 9:251–258. https://doi.org/10.1097/ACI.0b013e32832b3f33
Bernhisel-Broadbent J, Dintzis HM, Dintzis RZ, Sampson HA (1994) Allergenicity and antigenicity of chicken egg ovomucoid (Gal d III) compared with ovalbumin (Gal d I) in children with egg allergy and in mice. J Allergy Clin Immunol 93:1047–1059. https://doi.org/10.1016/S0091-6749(94)70054-0
Heine RG, Laske N, Hill DJ (2006) The diagnosis and management of egg allergy. Curr Allergy Asthma Rep 6:145–152. https://doi.org/10.1007/s11882-006-0053-0
Farrell HM, Qi PX, Uversky VN (2006) New views of protein structure: Applications to the caseins: Protein structure and functionality. In: Fishman ML, Qi PX, Wicker L (eds) Advances in Biopolymers, vol 935. ACS Symposium Series, vol 935. American Chemical Society, Washington DC, pp 52–70. https://doi.org/10.1021/bk-2006-0935.ch004
McMahon DJ, Oommen BS (2013) Casein micelle structure, functions, and interactions. In: McSweeney PLH, Fox PF (eds) Advanced dairy chemistry. Proteins: basic aspects, 4th Edition, vol 1A. Springer US, Boston, MA, pp 185–209. https://doi.org/10.1007/978-1-4614-4714-6_6
Farrell HM, Brown EM, L. ME (2013) Higher order structures of the caseins: a paradox? . In: McSweeney PLH, Fox PF (eds) Advanced dairy chemistry. Proteins: basic aspects, 4th Edition, vol 1A. Springer US, Boston, MA, pp 161–184. https://doi.org/10.1007/978-1-4614-4714-6_5
Tomura S, Ishizaki S, Nagashima Y, Shiomi K (2008) Reduction in the IgE reactivity of Pacific mackerel parvalbumin by mutations at Ca2+-binding sites. Fish Sci 74:411–417. https://doi.org/10.1111/j.1444-2906.2008.01538.x
Mao HY, Cao MJ, Maleki SJ, Cai QF, Su WJ, Yang Y, Liu GM (2013) Structural characterization and IgE epitope analysis of arginine kinase from Scylla paramamosain. Mol Immunol 56:463–470. https://doi.org/10.1016/j.molimm.2013.04.016
Yang Y, Cao M-J, Alcocer M, Liu Q-M, Fei D-X, Mao H-Y, Liu G-M (2015) Mapping and characterization of antigenic epitopes of arginine kinase of Scylla paramamosain. Mol Immunol 65:310–320. https://doi.org/10.1016/j.molimm.2015.02.010
Stănciuc N, Banu I, Turturică M, Aprodu I (2016) pH and heat induced structural changes of chicken ovalbumin in relation with antigenic properties. Int J Biol Macromol 93:572–581. https://doi.org/10.1016/j.ijbiomac.2016.09.025
Reese G, Ayuso R, Carle T, Lehrer SB (1999) IgE–binding epitopes of shrimp tropomyosin, the major allergen Pen a 1. Int Arch Allergy Immunol 118:300–301. https://doi.org/10.1159/000024108
Mine Y, Wei Zhang J (2002) Identification and fine mapping of IgG and IgE epitopes in ovomucoid. Biochem Biophys Res Comm 292:1070–1074. https://doi.org/10.1006/bbrc.2002.6725
Restani P, Fiocchi A, Beretta B, Velonà T, Giovannini M, Galli CL (1998) Effects of structure modifications on IgE binding properties of serum albumins. Int Arch Allergy Immunol 117:113–119. https://doi.org/10.1159/000023997
Benedé S, López-Expósito I, Molina E, López-Fandiño R (2015) Egg proteins as allergens and the effects of the food matrix and processing. Food Funct 6:694–713. https://doi.org/10.1039/C4FO01104J
Tong P, Gao J, Chen H, Li X, Zhang Y, Jian S, Wichers H, Wu Z, Yang A, Liu F (2012) Effect of heat treatment on the potential allergenicity and conformational structure of egg allergen ovotransferrin. Food Chem 131:603–610. https://doi.org/10.1016/j.foodchem.2011.08.084
Schwarcz WD, Carnelocce L, Silva JL, Oliveira AC, Gonçalves RB (2008) Conformational changes in bovine lactoferrin induced by slow or fast temperature increases. Biol Chem 389:1137–1142. https://doi.org/10.1515/BC.2008.116
Audagnotto M, Dal Peraro M (2017) Protein post-translational modifications: In silico prediction tools and molecular modeling. Comp Struct Biotechnol J 15:307–319. https://doi.org/10.1016/j.csbj.2017.03.004
Knorre DG, Kudryashova NV, Godovikova TS (2009) Chemical and functional aspects of posttranslational modification of proteins. Acta Naturae 1:29–51. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3347534/
Ruan WW, Cao MJ, Chen F, Cai QF, Su WJ, Wang YZ, Liu GM (2013) Tropomyosin contains IgE-binding epitopes sensitive to periodate but not to enzymatic deglycosylation. J Food Sci 78:C1116–C1121. https://doi.org/10.1111/1750-3841.12169
Besler M, Steinhart H, Paschke A (1997) Allergenicity of hen’s egg-white proteins: IgE binding of native and deglycosylated ovomucoid. Food Agric Immunol 9:277–288. https://doi.org/10.1080/09540109709354958
Boutrou R, Jardin J, Blais A, Tomé D, Léonil J (2008) Glycosylations of κ-casein-derived caseinomacropeptide reduce its accessibility to endo- but not exointestinal brush border membrane peptidases. J Agric Food Chem 56:8166–8173. https://doi.org/10.1021/jf801140d
Chen H-L, Mao H-Y, Cao M-J, Cai Q-F, Su W-J, Zhang Y-X, Liu G-M (2013) Purification, physicochemical and immunological characterization of arginine kinase, an allergen of crayfish (Procambarus clarkii). Food ChemToxicol 62:475–484. https://doi.org/10.1016/j.fct.2013.09.014
Bernard H, Meisel H, Creminon C, Wal JM (2000) Post-translational phosphorylation affects the IgE binding capacity of caseins. FEBS Lett 467:239–244. https://doi.org/10.1016/S0014-5793(00)01164-9
Bernard H, Negroni L, Chatel JM, Clement G, Adel-Patient K, Peltre G, Creminon C, Wal JM (2000) Molecular basis of IgE cross-reactivity between human β-casein and bovine β-casein, a major allergen of milk. Mol Immunol 37:161–167. https://doi.org/10.1016/S0161-5890(00)00029-8
Permyakov SE, Vologzhannikova AA, Emelyanenko VI, Knyazeva EL, Kazakov AS, Lapteva YS, Permyakova ME, Zhadan AP, Permyakov EA (2012) The impact of alpha-N-acetylation on structural and functional status of parvalbumin. Cell Calcium 52:366–376. https://doi.org/10.1016/j.ceca.2012.06.002
Bugajska-Schretter A, Elfman L, Fuchs T, Kaplotis S, Rumpold H, Valenta R, Spitzauer S (1998) Parvalbumin, a cross-reactive fish allergen, contains IgE-binding epitopes sensitive to periodate treatment and Ca2+ depletion. J Allergy Clin Immunol 101:67–74. https://doi.org/10.1016/S0091-6749(98)70195-2
Holt C, Carver JA, Ecroyd H, Thorn DC (2013) Invited review: Caseins and the casein micelle: Their biological functions, structures, and behavior in foods1. J Dairy Sci 96:6127–6146. https://doi.org/10.3168/jds.2013-6831
Zhu Y, Vanga SK, Wang J, Raghavan V (2018) Impact of food processing on the structural and allergenic properties of egg white. Trends Food Sci Technol 78:188–196. https://doi.org/10.1016/j.tifs.2018.06.005
Hufnagl K, Ghosh D, Wagner S, Fiocchi A, Dahdah L, Bianchini R, Braun N, Steinborn R, Hofer M, Blaschitz M, Roth GA, Hofstetter G, Roth-Walter F, Pacios LF, Jensen-Jarolim E (2018) Retinoic acid prevents immunogenicity of milk lipocalin Bos d 5 through binding to its immunodominant T-cell epitope. Sci Rep 8:1598. https://doi.org/10.1038/s41598-018-19883-0
Liu J, Ru Q, Ding Y (2012) Glycation a promising method for food protein modification: physicochemical properties and structure, a review. Food Res Int 49:170–183. https://doi.org/10.1016/j.foodres.2012.07.034
Rao Q, Jiang X, Li Y, Samiwala M, Labuza TP (2018) Can glycation reduce food allergenicity? J Agric Food Chem 66:4295–4299. https://doi.org/10.1021/acs.jafc.8b00660
Teodorowicz M, van Neerven J, Savelkoul H (2017) Food processing: The influence of the maillard reaction on immunogenicity and allergenicity of food proteins. Nutrients 9:835. https://doi.org/10.3390/nu9080835
Nakamura A, Sasaki F, Watanabe K, Ojima T, Ahn D-H, Saeki H (2006) Changes in allergenicity and digestibility of squid tropomyosin during the Maillard reaction with ribose. J Agric Food Chem 54:9529–9534. https://doi.org/10.1021/jf061070d
Nakamura A, Watanabe K, Ojima T, Ahn D-H, Saeki H (2005) Effect of Maillard reaction on allergenicity of scallop tropomyosin. J Agric Food Chem 53:7559–7564. https://doi.org/10.1021/jf0502045
Fu L, Wang C, Wang J, Ni S, Wang Y (2019) Maillard reaction with ribose, galacto-oligosaccharide or chitosan-oligosaccharide reduced the allergenicity of shrimp tropomyosin by inducing conformational changes. Food Chem 274:789–795. https://doi.org/10.1016/j.foodchem.2018.09.068
Fang L, Li G, Gu R, Cai M, Lu J (2018) Influence of thermal treatment on the characteristics of major oyster allergen Cra g 1 (tropomyosin). J Sci Food Agric 98:5322–5328. https://doi.org/10.1002/jsfa.9071
de Jongh HHJ, Robles CL, Timmerman E, Nordlee JA, Lee P-W, Baumert JL, Hamilton RG, Taylor SL, Koppelman SJ (2013) Digestibility and IgE-binding of glycosylated codfish parvalbumin. BioMed Res Int 2013:756789. https://doi.org/10.1155/2013/756789
Li Z, Jiang M, You J, Luo Y, Feng L (2014) Impact of Maillard reaction conditions on the antigenicity of parvalbumin, the major allergen in grass carp. Food Agric Immunol 25:486–497. https://doi.org/10.1080/09540105.2013.838943
Zhao Y-J, Cai Q-F, Jin T-c, Zhang L-J, Fei D-X, Liu G-M, Cao M-J (2017) Effect of Maillard reaction on the structural and immunological properties of recombinant silver carp parvalbumin. LWT-Food Sci Technol 75:25–33. https://doi.org/10.1016/j.lwt.2016.08.049
Pinto MS, Léonil J, Henry G, Cauty C, Carvalho AF, Bouhallab S (2014) Heating and glycation of β-lactoglobulin and β-casein: aggregation and in vitro digestion. Food Res Int 55:70–76. https://doi.org/10.1016/j.foodres.2013.10.030
Zhao D, Li L, Le TT, Larsen LB, Su G, Liang Y, Li B (2017) Digestibility of glyoxal-glycated β-casein and β-lactoglobulin and distribution of peptide-bound advanced glycation end products in gastrointestinal digests. J Agric Food Chem 65:5778–5788. https://doi.org/10.1021/acs.jafc.7b01951
Enomoto H, Li C-P, Morizane K, Ibrahim HR, Sugimoto Y, Ohki S, Ohtomo H, Aoki T (2007) Glycation and phosphorylation of β-lactoglobulin by dry-heating: effect on protein structure and some properties. J Agric Food Chem 55:2392–2398. https://doi.org/10.1021/jf062830n
Liu F, Teodorowicz M, van Boekel MAJS, Wichers HJ, Hettinga KA (2016) The decrease in the IgG-binding capacity of intensively dry heated whey proteins is associated with intense Maillard reaction, structural changes of the proteins and formation of RAGE-ligands. Food Funct 7:239–249. https://doi.org/10.1039/C5FO00718F
Taheri-Kafrani A, Gaudin J-C, Rabesona H, Nioi C, Agarwal D, Drouet M, Chobert J-M, Bordbar A-K, Haertle T (2009) Effects of heating and glycation of β-lactoglobulin on its recognition by IgE of sera from cow milk allergy patients. J Agric Food Chem 57:4974–4982. https://doi.org/10.1021/jf804038t
Perusko M, van Roest M, Stanic-Vucinic D, Simons PJ, Pieters RHH, Cirkovic Velickovic T, Smit JJ (2018) Glycation of the major milk allergen β-lactoglobulin changes its allergenicity by alterations in cellular uptake and degradation. Mol Nutr Food Res 62:1800341. https://doi.org/10.1002/mnfr.201800341
Yang W, Tu Z, Wang H, Zhang L, Kaltashov IA, Zhao Y, Niu C, Yao H, Ye W (2018) The mechanism of reduced IgG/IgE-binding of β-lactoglobulin by pulsed electric field pretreatment combined with glycation revealed by ECD/FTICR-MS. Food Funct 9:417–425. https://doi.org/10.1039/C7FO01082F
Yang W, Tu Z, Wang H, Zhang L, Xu S, Niu C, Yao H, Kaltashov IA (2017) Mechanism of reduction in IgG and IgE binding of β-lactoglobulin induced by ultrasound pretreatment combined with dry-state glycation: a study using conventional spectrometry and high-resolution mass spectrometry. J Agric Food Chem 65:8018–8027. https://doi.org/10.1021/acs.jafc.7b02842
Corzo-Martínez M, Soria AC, Belloque J, Villamiel M, Moreno FJ (2010) Effect of glycation on the gastrointestinal digestibility and immunoreactivity of bovine β-lactoglobulin. Int Dairy J 20:742–752. https://doi.org/10.1016/j.idairyj.2010.04.002
Bu G, Luo Y, Zheng Z, Zheng H (2009) Effect of heat treatment on the antigenicity of bovine α-lactalbumin and β-lactoglobulin in whey protein isolate. Food Agric Immunol 20:195–206. https://doi.org/10.1080/09540100903026116
Ma X, Gao J, Tong P, Yang H, Zu Q, Meng X, Lu J, Chen H (2015) Effects of Maillard reaction conditions on in vitro immunoglobulin G binding capacity of ovalbumin using response surface methodology. Food Agric Immunol 26:835–847. https://doi.org/10.1080/09540105.2015.1039496
Jiménez-Saiz R, Belloque J, Molina E, López-Fandiño R (2011) Human immunoglobulin E (IgE) binding to heated and glycated ovalbumin and ovomucoid before and after in vitro digestion. J Agric Food Chem 59:10044–10051. https://doi.org/10.1021/jf2014638
Ma XJ, Chen HB, Gao JY, Hu CQ, Li X (2013) Conformation affects the potential allergenicity of ovalbumin after heating and glycation. Food Addit Cont Part A 30:1684–1692. https://doi.org/10.1080/19440049.2013.822105
Ma X-j, Gao J-y, Chen H-b (2013) Combined effect of glycation and sodium carbonate-bicarbonate buffer concentration on IgG binding, IgE binding and conformation of ovalbumin. J Sci Food Agric 93:3209–3215. https://doi.org/10.1002/jsfa.6157
Yang W, Tu Z, Wang H, Zhang L, Song Q (2018) Glycation of ovalbumin after high-intensity ultrasound pretreatment: effects on conformation, immunoglobulin (Ig)G/IgE binding ability and antioxidant activity. J Sci Food Agric 98:3767–3773. https://doi.org/10.1002/jsfa.8890
Hilmenyuk T, Bellinghausen I, Heydenreich B, Ilchmann A, Toda M, Grabbe S, Saloga J (2010) Effects of glycation of the model food allergen ovalbumin on antigen uptake and presentation by human dendritic cells. Immunology 129:437–445. https://doi.org/10.1111/j.1365-2567.2009.03199.x
Ilchmann A, Burgdorf S, Scheurer S, Waibler Z, Nagai R, Wellner A, Yamamoto Y, Yamamoto H, Henle T, Kurts C, Kalinke U, Vieths S, Toda M (2010) Glycation of a food allergen by the Maillard reaction enhances its T-cell immunogenicity: role of macrophage scavenger receptor class A type I and II. J Allergy Clin Immunol 125:175-183.e111. https://doi.org/10.1016/j.jaci.2009.08.013
Enomoto H, Hayashi Y, Li CP, Ohki S, Ohtomo H, Shiokawa M, Aoki T (2009) Glycation and phosphorylation of α-lactalbumin by dry heating: Effect on protein structure and physiological functions. J Dairy Sci 92:3057–3068. https://doi.org/10.3168/jds.2009-2014
Kleber N, Krause I, Illgner S, Hinrichs J (2004) The antigenic response of β-lactoglobulin is modulated by thermally induced aggregation. Eur Food Res Technol 219:105–110. https://doi.org/10.1007/s00217-004-0924-3
Docena GH, Fernandez R, Chirdo FG, Fossati CA (1996) Identification of casein as the major allergenic and antigenic protein of cow’s milk. Allergy 51:412–416. https://doi.org/10.1111/j.1398-9995.1996.tb04639.x
Bloom KA, Huang FR, Bencharitiwong R, Bardina L, Ross A, Sampson HA, Nowak-Węgrzyn A (2014) Effect of heat treatment on milk and egg proteins allergenicity. Pediatr Allergy Immunol 25:740–746. https://doi.org/10.1111/pai.12283
Dupont D, Boutrou R, Menard O, Jardin J, Tanguy G, Schuck P, Haab BB, Leonil J (2010) Heat treatment of milk during powder manufacture increases casein resistance to simulated infant digestion. Food Dig 1:28–39. https://doi.org/10.1007/s13228-010-0003-0
Dupont D, Mandalari G, Mollé D, Jardin J, Rolet-Répécaud O, Duboz G, Léonil J, Mills CEN, Mackie AR (2010) Food processing increases casein resistance to simulated infant digestion. Mol Nutr Food Res 54:1677–1689. https://doi.org/10.1002/mnfr.200900582
Kato Y, Oozawa E, Matsuda T (2001) Decrease in antigenic and allergenic potentials of ovomucoid by heating in the presence of wheat flour: dependence on wheat variety and intermolecular disulfide bridges. J Agric Food Chem 49:3661–3665. https://doi.org/10.1021/jf0102766
Kim K-B-W-R, Lee SY, Song EJ, Park JG, Lee JW, Byun MW, Kim KE, Ahn DH (2010) Changes in allergenicity of porcine serum albumin by gamma irradiation. Korean J Food Sci Anim Resour 30:397–402. https://doi.org/10.5851/kosfa.2010.30.3.397
Usui M, Harada A, Ishimaru T, Sakumichi E, Saratani F, Sato-Minami C, Azakami H, Miyasaki T, Ki H (2013) Contribution of structural reversibility to the heat stability of the tropomyosin shrimp allergen. Biosci Biotechnol Biochem 77:948–953. https://doi.org/10.1271/bbb.120887
Faisal M, Vasiljevic T, Donkor ON (2019) Effects of selected processing treatments on antigenicity of banana prawn (Fenneropenaeus merguiensis) tropomyosin. Int J Food Sci Technol 54:183–193. https://doi.org/10.1111/ijfs.13922
Rolland JM, Varese NP, Abramovitch JB, Anania J, Nugraha R, Kamath S, Hazard A, Lopata AL, O’Hehir RE (2018) Effect of heat processing on IgE reactivity and cross-reactivity of tropomyosin and other allergens of Asia-Pacific mollusc species: identification of novel sydney rock oyster tropomyosin Sac g 1. Mol Nutr Food Res 62:1800148. https://doi.org/10.1002/mnfr.201800148
Bernhisel-Broadbent J, Scanlon SM, Sampson HA (1992) Fish hypersensitivity. I. In vitro and oral challenge results in fish- allergic patients. J Allergy Clin Immunol 89:730–737. https://doi.org/10.1016/0091-6749(92)90381-B
Lamberti C, Baro C, Giribaldi M, Napolitano L, Cavallarin L, Giuffrida MG (2018) Effects of two different domestic boiling practices on the allergenicity of cow’s milk proteins. J Sci Food Agric 98:2370–2377. https://doi.org/10.1002/jsfa.8728
Xu Q, Shi J, Yao M, Jiang M, Luo Y (2016) Effects of heat treatment on the antigenicity of four milk proteins in milk protein concentrates. Food Agric Immunol 27:401–413. https://doi.org/10.1080/09540105.2015.1117059
Lee J-W, Lee K-Y, Yook H-S, Lee S-Y, Kim H-Y, Jo C, Byun M-W (2002) Allergenicity of hen’s egg ovomucoid gamma irradiated and heated under different pH conditions. J Food Prot 65:1196–1199. https://doi.org/10.4315/0362-028X-65.7.1196
Carrasco PR, Klug C, Swoboda I, Augustin G, Quirce S, Hemmer W (2016) Serum albumin, an important allergen also in processed pork meat products. Allergy 71:627–627. https://doi.org/10.1111/all.12979
Restani P, Ballabio C, Cattaneo A, Isoardi P, Terracciano L, Fiocchi A (2004) Characterization of bovine serum albumin epitopes and their role in allergic reactions. Allergy 59:21–24. https://doi.org/10.1111/j.1398-9995.2004.00568.x
Quirce S, Marañón F, Umpiérrez A, de laas Heras M, Jiménez A, Fernández-Caldas E, Sastre J (2000) Identification of chicken serum albumin as a thermolabile egg allergen (Gal d 5) responsible for the bird-egg syndrome. J Allergy Clin Immunol 105:S136–S137. https://doi.org/10.1016/S0091-6749(00)90841-8
Kim M-J, Lee J-W, Yook H-S, Lee S-Y, Kim M-C, Byun M-W (2002) Changes in the antigenic and immunoglobulin E–binding properties of hen’s egg albumin with the combination of heat and gamma irradiation treatment. J Food Prot 65:1192–1195. https://doi.org/10.4315/0362-028X-65.7.1192
Azdad O, Mejrhit N, Aarab L (2018) Reduction of the allergenicity of cow’s milk alpha-lactalbumin under heat-treatment and enzymatic hydrolysis in Moroccan population. Eur Ann Allergy Clin Immunol 50:177–183. https://doi.org/10.23822/EurAnnACI.1764-1489.60
Liu M, Liu G-Y, Yang Y, Mei X-J, Yang H, Li Y, Cao M-J, Liu G-M (2018) Thermal processing influences the digestibility and immunoreactivity of muscle proteins of Scylla paramamosain. LWT-Food Sci Technol 98:559–567. https://doi.org/10.1016/j.lwt.2018.09.027
Hu G, Zheng Y, Liu Z, Deng Y, Zhao Y (2016) Structure and IgE-binding properties of α-casein treated by high hydrostatic pressure, UV-C, and far-IR radiations. Food Chem 204:46–55. https://doi.org/10.1016/j.foodchem.2016.02.113
Bogahawaththa D, Buckow R, Chandrapala J, Vasiljevic T (2018) Comparison between thermal pasteurization and high pressure processing of bovine skim milk in relation to denaturation and immunogenicity of native milk proteins. Innov Food Sci Emerg Technol 47:301–308. https://doi.org/10.1016/j.ifset.2018.03.016
Boughellout H, Choiset Y, Rabesona H, Chobert JM, Haertle T, Mounir S, Allaf K, Zidoune MN (2015) Effect of instant controlled pressure drop (DIC) treatment on milk protein’s immunoreactivity. Food Agric Immunol 26:71–81. https://doi.org/10.1080/09540105.2013.864607
Kim K, Kim SJ, Lee SY, Song EJ, Ahn DH (2008) Changes in allergenicity of porcine serum albumin by microwave, sonication, and high hydrostatic pressure. Korean J Food Sci Anim Resour 28:499–504. https://doi.org/10.5851/kosfa.2008.28.4.499
Kurpiewska K, Biela A, Loch JI, Lipowska J, Siuda M, Lewiński K (2019) Towards understanding the effect of high pressure on food protein allergenicity: β-lactoglobulin structural studies. Food Chem 270:315–321. https://doi.org/10.1016/j.foodchem.2018.07.104
Kleber N, Maier S, Hinrichs J (2007) Antigenic response of bovine β-lactoglobulin influenced by ultra-high pressure treatment and temperature. Innov Food Sci Emerg Technol 8:39–45. https://doi.org/10.1016/j.ifset.2006.05.001
Vanga SK, Singh A, Raghavan V (2017) Review of conventional and novel food processing methods on food allergens. Crit Rev Food Sci Nutri 57:2077–2094. https://doi.org/10.1080/10408398.2015.1045965
Barbosa-Cánovas GV, Altunakar B (2006) Pulsed electric fields processing of foods: An overview. In: Raso J, Heinz V (eds) Pulsed Electric Fields Technology for the Food Industry: Fundamentals and Applications. Springer, US, Boston, MA, pp 3–26. https://doi.org/10.1007/978-0-387-31122-7_1
Ekezie F-GC, Cheng J-H, Sun D-W (2018) Effects of nonthermal food processing technologies on food allergens: a review of recent research advances. Trend Food Sci Technol 74:12–25. https://doi.org/10.1016/j.tifs.2018.01.007
Shriver S, Yang W, Chung S-Y, Percival S (2011) Pulsed ultraviolet light reduces immunoglobulin E binding to Atlantic white shrimp (Litopenaeus setiferus) extract. Int J Environ Res Public Health 8:2569–2583. https://doi.org/10.3390/ijerph8072569
Yang WW, Shriver SK, Chung S-y, Percival S, Correll MJ, Rababah TM (2012) In vitro gastric and intestinal digestions of pulsed light-treated shrimp extracts. Appl Biochem Biotechnol 166:1409–1422. https://doi.org/10.1007/s12010-011-9534-2
Tammineedi CVRK, Choudhary R, Perez-Alvarado GC, Watson DG (2013) Determining the effect of UV-C, high intensity ultrasound and nonthermal atmospheric plasma treatments on reducing the allergenicity of α-casein and whey proteins. LWT - Food Sci Technol 54:35–41. https://doi.org/10.1016/j.lwt.2013.05.020
Ham J, Jeong S, Lee S, Han G, Chae H, Yoo Y, Kim D, Lee W, Jo C (2009) Irradiation effect on α-and β-caseins of milk and Queso Blanco cheese determined by capillary electrophoresis. Rad Phys Chem 78:158–163. https://doi.org/10.1016/j.radphyschem.2008.09.008
Meng X, Li X, Wang X, Gao J, Yang H, Chen H (2016) Potential allergenicity response to structural modification of irradiated bovine α-lactalbumin. Food Funct 7:3102–3110. https://doi.org/10.1039/C6FO00400H
Byun M-W, Lee J-W, Yook H-S, Jo C, Kim H-Y (2002) Application of gamma irradiation for inhibition of food allergy. Rad Phys Chem 63:369–370. https://doi.org/10.1016/S0969-806X(01)00528-X
Lee J-W, Kim J-H, Yook H-S, Kang K-O, Lee S-Y, Hwang H-J, Byun M-W (2001) Effects of gamma radiation on the allergenic and antigenic properties of milk proteins. J Food Prot 64:272–276. https://doi.org/10.4315/0362-028X-64.2.272
Zhu X, Wang W, Shen J, Xu X, Zhou G (2019) Influence of gamma irradiation on porcine serum albumin structural properties and allergenicity. J AOAC Int 101:529–535. https://doi.org/10.5740/jaoacint.17-0160
Liu Y, Li Z, Pavase T, Li Z, Liu Y, Wang N (2017) Evaluation of electron beam irradiation to reduce the IgE binding capacity of frozen shrimp tropomyosin. Food Agric Immunol 28:189–201. https://doi.org/10.1080/09540105.2016.1251394
Lee JW, Seo JH, Kim JH, Lee SY, Kim KS, Byun MW (2005) Changes of the antigenic and allergenic properties of a hen’s egg albumin in a cake with gamma-irradiated egg white. Rad Phys Chem 72:645–650. https://doi.org/10.1016/j.radphyschem.2004.03.088
Yang W, Tu Z, Wang H, Zhang L, Gao Y, Li X, Tian M (2017) Immunogenic and structural properties of ovalbumin treated by pulsed electric fields. Int J Food Prop 20:S3164–S3176. https://doi.org/10.1080/10942912.2017.1396479
Pereira RN, Costa J, Rodrigues RM, Villa C, Machado L, Mafra I, Vicente AA (2020) Effects of ohmic heating on the immunoreactivity of β-lactoglobulin – a relationship towards structural aspects. Food Funct 11:4002–4013. https://doi.org/10.1039/C9FO02834J
Onwude DI, Hashim N, Janius R, Abdan K, Chen G, Oladejo AO (2017) Non-thermal hybrid drying of fruits and vegetables: a review of current technologies. Innov Food Sci Emerging Technol 43:223–238. https://doi.org/10.1016/j.ifset.2017.08.010
Mañas P, Muñoz B, Sanz D, Condón S (2006) Inactivation of lysozyme by ultrasonic waves under pressure at different temperatures. Enzyme Microb Technol 39:1177–1182. https://doi.org/10.1016/j.enzmictec.2005.11.053
Kim SJ, Kim K, Song EJ, Lee SY, Yoon SY, Lee SJ, Lee CJ, Park JG, Lee JW, Byun MW, Ahn DH (2009) Changes of pork antigenicity by heat, pressure, sonication, microwave, and gamma irradiation. Korean J Food Sci Anim Resour 29:709–718. https://doi.org/10.5851/kosfa.2009.29.6.709
Park JG, Saeki H, Nakamura A, Kim K, Lee JW, Byun MW, Kim SM, Lim SM, Ahn DH (2007) Allergenicity changes in raw shrimp (Acetes japonicus) and Saeujeot (salted and fermented shrimp) in cabbage Kimchi due to fermentation conditions. Food Sci Biotechnol 16:1011–1017. http://www.koreascience.or.kr/article/JAKO200709905797926.view
Pessato TB, Carvalho NC, Tavano OL, Fernandes LGR, Zollner RL, Netto FM (2016) Whey protein isolate hydrolysates obtained with free and immobilized alcalase: characterization and detection of residual allergens. Food Res Int 83:112–120. https://doi.org/10.1016/j.foodres.2016.02.015
Zheng H, Shen X, Bu G, Luo Y (2008) Effects of pH, temperature and enzyme-to-substrate ratio on the antigenicity of whey protein hydrolysates prepared by Alcalase. Int Dairy J 18:1028–1033. https://doi.org/10.1016/j.idairyj.2008.05.002
Wróblewska B, Markiewicz LH, Szyc AM, Dietrich MA, Szymkiewicz A, Fotschki J (2016) Lactobacillus casei LcY decreases milk protein immunoreactivity of fermented buttermilk but also contains IgE-reactive proteins. Food Res Int 83:95–101. https://doi.org/10.1016/j.foodres.2016.02.016
Sabadin IS, Villas-Boas MB, Zollner RD, Netto FM (2012) Effect of combined treatment of hydrolysis and polymerization with transglutaminase on beta-lactoglobulin antigenicity. Eur Food Res Technol 235:801–809. https://doi.org/10.1007/s00217-012-1802-z
Ahmadova A, El-Ghaish S, Choiset Y, Rabesona H, Drouet M, Chobert J, Kuliev AA, Haertle T (2013) Modification of IgE binding to β-and αS1-caseins by proteolytic activity of Lactobacillus helveticus A75. J Food Biochem 37:491–500. https://doi.org/10.1111/j.1745-4514.2012.00664.x
Yao M, Luo Y, Shi J, Zhou Y, Xu Q, Li Z (2014) Effects of fermentation by Lactobacillus rhamnosus GG on the antigenicity and allergenicity of four cows’ milk proteins. Food Agric Immunol 25:545–555. https://doi.org/10.1080/09540105.2013.852163
Shi J, Luo Y, Xiao Y, Li Z, Xu Q, Yao M (2014) Effects of fermentation by Lactobacillus casei on the antigenicity and allergenicity of four bovine milk proteins. Int Dairy J 35:75–80. https://doi.org/10.1016/j.idairyj.2013.10.010
Golkar A, Milani JM, Vasiljevic T (2019) Altering allergenicity of cow’s milk by food processing for applications in infant formula. Crit Rev Food Sci Nutr 59:159–172. https://doi.org/10.1080/10408398.2017.1363156
Høst A, Halken S (2004) Hypoallergenic formulas – when, to whom and how long: after more than 15 years we know the right indication! Allergy 59:45–52. https://doi.org/10.1111/j.1398-9995.2004.00574.x
Ballmer-Weber BK, Brockow K, Fiocchi A, Theler B, Vogel L, Ring J, Szépfalusi Z, Mazzina O, Schaller R, Fritsché R, Vissers YM, Nutten S (2016) Hydrolysed egg displays strong decrease in allergenicity and is well tolerated by egg-allergic patients. Allergy 71:728–732. https://doi.org/10.1111/all.12852
Lin H, Li Z, Lin H, Song Y, Lv L, Hao Z (2015) Effect of pH shifts on IgE-binding capacity and conformational structure of tropomyosin from short-neck clam (Ruditapes philippinarum). Food Chem 188:248–255. https://doi.org/10.1016/j.foodchem.2015.05.007
Jiménez-Saiz R, Pineda-Vadillo C, López-Fandiño R, Molina E (2012) Human IgE binding and in vitro digestion of S-OVA. Food Chem 135:1842–1847. https://doi.org/10.1016/j.foodchem.2012.06.044
Akkerdaas J, Totis M, Barnett B, Bell E, Davis T, Edrington T, Glenn K, Graser G, Herman R, Knulst A, Ladics G, McClain S, Poulsen LK, Ranjan R, Rascle J-B, Serrano H, Speijer D, Wang R, Pereira Mouriès L, Capt A, van Ree R (2018) Protease resistance of food proteins: a mixed picture for predicting allergenicity but a useful tool for assessing exposure. Clin Transl Allergy 8:30. https://doi.org/10.1186/s13601-018-0216-9
Foster ES, Kimber I, Dearman RJ (2013) Relationship between protein digestibility and allergenicity: comparisons of pepsin and cathepsin. Toxicology 309:30–38. https://doi.org/10.1016/j.tox.2013.04.011
Vickery BP, Chin S, Burks AW (2011) Pathophysiology of food allergy. Pediatr Clin N Am 58:363–376. https://doi.org/10.1016/j.pcl.2011.02.012
Perrier C, Corthésy B (2011) Gut permeability and food allergies. Clin Exp Allergy 41:20–28. https://doi.org/10.1111/j.1365-2222.2010.03639.x
Steele L, Mayer L, Cecilia Berin M (2012) Mucosal immunology of tolerance and allergy in the gastrointestinal tract. Immunol Res 54:75–82. https://doi.org/10.1007/s12026-012-8308-4
Yu HL, Ruan WW, Cao MJ, Cai QF, Shen HW, Liu GM (2013) Identification of physicochemical properties of Scylla paramamosain allergen, arginin kinase. J Sci Food Agric 93:245–253. https://doi.org/10.1002/jsfa.5748
Astwood JD, Leach JN, Fuchs RL (1996) Stability of food allergens to digestion in vitro. Nat Biotechnol 14:1269–1273. https://doi.org/10.1038/nbt1096-1269
Martinez J, Sanchez R, Castellanos M, Fernandez-Escamilla AM, Vazquez-Cortes S, Fernandez-Rivas M, Gasset M (2015) Fish beta-parvalbumin acquires allergenic properties by amyloid assembly. Swiss Med Wkly 145:w14128. https://doi.org/10.4414/smw.2015.14128
Liu GM, Huang YY, Cai QF, Weng WY, Su WJ, Cao MJ (2011) Comparative study of in vitro digestibility of major allergen, tropomyosin and other proteins between Grass prawn (Penaeus monodon) and Pacific white shrimp (Litopenaeus vannamei). J Sci Food Agric 91:163–170. https://doi.org/10.1002/jsfa.4167
Lv L, Lin H, Li Z, Ahmed I, Chen G (2017) Determining the effect of malondialdehyde on the IgE-binding capacity of shrimp tropomyosin upon in vitro digestion. J Sci Food Agric 97:4588–4594. https://doi.org/10.1002/jsfa.8328
Jiménez-Saiz R, Martos G, Carrillo W, López-Fandiño R, Molina E (2011) Susceptibility of lysozyme to in-vitro digestion and immunoreactivity of its digests. Food Chem 127:1719–1726. https://doi.org/10.1016/j.foodchem.2011.02.047
Yao M, Xu Q, Luo Y, Shi J, Li Z (2015) Study on reducing antigenic response and IgE-binding inhibitions of four milk proteins of Lactobacillus casei 1134. J Sci Food Agric 95:1303–1312. https://doi.org/10.1002/jsfa.6823
Chicon R, Belloque J, Alonso E, Lopez-Fandino R (2009) Antibody binding and functional properties of whey protein hydrolysates obtained under high pressure. Food Hydrocolloids 23:593–599. https://doi.org/10.1016/j.foodhyd.2008.04.001
Villas-Boas MB, Benedé S, de Lima Zollner R, Netto FM, Molina E (2015) Epitopes resistance to the simulated gastrointestinal digestion of β-lactoglobulin submitted to two-step enzymatic modification. Food Res Int 72:191–197. https://doi.org/10.1016/j.foodres.2015.03.044
Yoshino K, Sakai K, Mizuha Y, Shimizuike A, Yamamoto S (2004) Peptic digestibility of raw and heat-coagulated hen’s egg white proteins at acidic pH range. Int J Food Sci Nutri 55:635–640. https://doi.org/10.1080/09637480412331350173
Benedé S, López-Expósito I, Giménez G, Grishina G, Bardina L, Sampson HA, López-Fandiño R, Molina E (2014) Mapping of IgE epitopes in in vitro gastroduodenal digests of β-lactoglobulin produced with human and simulated fluids. Food Res Int 62:1127–1133. https://doi.org/10.1016/j.foodres.2014.05.069
Takagi K, Teshima R, Okunuki H, Itoh S, Kawasaki N, Kawanishi T, Hayakawa T, Kohno Y, Urisu A, Sawada Ji (2005) Kinetic analysis of pepsin digestion of chicken egg white ovomucoid and allergenic potential of pepsin fragments. Int Arch Allergy Immunol 136:23–32. https://doi.org/10.1159/000082581
Claude M, Lupi R, Picariello G, Drouet M, Larré C, Denery-Papini S, Brossard C (2019) Digestion differently affects the ability of native and thermally aggregated ovalbumin to trigger basophil activation. Food Res Int 118:108–114. https://doi.org/10.1016/j.foodres.2017.11.040
Bublin M, Eiwegger T, Breiteneder H (2014) Do lipids influence the allergic sensitization process? J Allergy Clin Immunol 134:521–529. https://doi.org/10.1016/j.jaci.2014.04.015
Pekar J, Ret D, Untersmayr E (2018) Stability of allergens. Mol Immunol 100:14–20. https://doi.org/10.1016/j.molimm.2018.03.017
Luo C, Guo Y, Li Z, Ahmed I, Pramod SN, Gao X, Lv L, Lin H (2020) Lipid emulsion enhances fish allergen parvalbumin’s resistance to in vitro digestion and IgG/IgE binding capacity. Food Chem 302:125333. https://doi.org/10.1016/j.foodchem.2019.125333
Moreno FJ, Mackie AR, Mills ENC (2005) Phospholipid interactions protect the milk allergen α-lactalbumin from proteolysis during in vitro digestion. J Agric Food Chem 53:9810–9816. https://doi.org/10.1021/jf0515227
Mandalari G, Adel-Patient K, Barkholt V, Baro C, Bennett L, Bublin M, Gaier S, Graser G, Ladics GS, Mierzejewska D, Vassilopoulou E, Vissers YM, Zuidmeer L, Rigby NM, Salt LJ, Defernez M, Mulholland F, Mackie AR, Wickham MSJ, Mills ENC (2009) In vitro digestibility of β-casein and β-lactoglobulin under simulated human gastric and duodenal conditions: A multi-laboratory evaluation. Regul Toxicol Pharmacol 55:372–381. https://doi.org/10.1016/j.yrtph.2009.08.010
Lv L, Lin H, Li Z, Wang J, Ahmed I, Chen H (2017) Changes of structure and IgE binding capacity of shrimp (Metapenaeus ensis) tropomyosin followed by acrolein treatment. Food Funct 8:1028–1036. https://doi.org/10.1039/C6FO01479H
Acknowledgements
The authors are all part of the COST Action FA1402 entitled ImpARAS—Improving Allergy Risk Assessment Strategy for New Food Proteins. The authors thank all ImpARAS members for their active participation in the ImpARAS meetings and lively discussions.
Funding
The authors highly appreciate the support from the COST Office. This article is based upon work from COST Action FA1402, supported by COST (European Cooperation in Science and Technology, www.cost.eu). This work was also supported by Fundação para a Ciência e Tecnologia under the Partnership Agreement UIDB 50006/2020 and by the projects AlleRiskAssess—PTDC/BAA-AGR/31720/2017. C.V. is grateful to FCT grant (PD/BD/114576/2016) financed by POPH-QREN (subsidised by FSE and MCTES). J.C. acknowledges FCT for the research contract (SFRH/BPD/102404/2014). T.C.V. is grateful to the Ministry of Education, Science and Technological Development of the Republic of Serbia through grant number OI172024. P.M.R. and D.S. are grateful to FCT through project UIDB/04326/2020 and Mar2020 16-02-01-FMP-0014—“ALLYFISH”. J.K. and A.K. acknowledge PRIDE program grants (PRIDE/11012546/NEXTIMMUNE). J.K. also acknowledges FNR (Fonds National de la Recherche) and the PMC (Personalised Medicine Consortium).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, data collection, and analysis. All authors have taken part in the discussions and writing of the article. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Costa, J., Villa, C., Verhoeckx, K. et al. Are Physicochemical Properties Shaping the Allergenic Potency of Animal Allergens?. Clinic Rev Allerg Immunol 62, 1–36 (2022). https://doi.org/10.1007/s12016-020-08826-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-020-08826-1