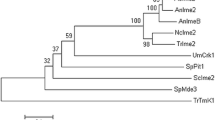
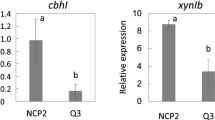
Abstract
Mitogen-activated protein kinase (MAPK) cascades are broadly conserved and play essential roles in multiple cellular processes, including fungal development, pathogenicity, and secondary metabolism. Their function, however, also exhibits species and strain specificity. Penicillium oxalicum secretes plant-biomass-degrading enzymes (PBDEs) that contribute to the carbon cycle in the natural environment and to utilization of lignocellulose in industrial processes. However, knowledge of the MAPK pathway in P. oxalicum has been relatively limited. In this study, comparative transcriptomic analysis of P. oxalicum, cultured on different carbon sources, found ten putative kinase genes with significantly modified transcriptional levels. Six of these putative kinase genes were knocked out in the parental strain ∆PoxKu70, and deletion of the gene, Fus3/Kss1-like PoxMK1 (POX00158), resulted in the largest reduction (91.1%) in filter paper cellulase production. Further tests revealed that the mutant ∆PoxMK1 lost 37.1 to 92.2% of PBDE production, under both submerged- and solid-state fermentation conditions, compared with ∆PoxKu70. In addition, the mutant ∆PoxMK1 had reduced vegetative growth and increased pigment biosynthesis. Comparative transcriptomic analysis showed that PoxMK1 deletion from P. oxalicum downregulated the expression of major PBDE genes and known regulatory genes such as PoxClrB and PoxCxrB, whereas the transcription of pigment biosynthesis-related genes was upregulated. Comparative phosphoproteomic analysis revealed that PoxMK1 deletion considerably modified phosphorylation of key transcription- and signal transduction-associated proteins, including transcription factors Mcm1 and Atf1, RNA polymerase II subunits Rpb1 and Rpb9, MAPK-associated Hog1 and Ste7, and cyclin-dependent kinase Kin28. These findings provide novel insights into understanding signal transduction and regulation of PBDE gene expression in fungi.
Key points
• PoxMK1 is involved in expression of PBDE- and pigment synthesis-related genes.
• PoxMK1 is required for vegetative growth of P. oxalicum.
• PoxMK1 is involved in phosphorylation of key TFs, kinases, and RNA polymerase II.






Similar content being viewed by others
References
Bentil JA, Thygesen A, Mensah M, Lange L, Meyer AS (2018) Cellulase production by white-rot basidiomycetous fungi: solid-state versus submerged cultivation. Appl Microbiol Biotechnol 102(14):5827–5839
de Paula RG, Antoniêto ACC, Carraro CB, Lopes DCB, Persinoti GF, Peres NTA, Martinez-Rossi NM, Silva-Rocha R, Silva RN (2018) The duality of the MAPK signaling pathway in the control of metabolic processes and cellulase production in Trichoderma reesei. Sci Rep 8:14931
Diaz AB, Blandino A, Webb C, Caro I (2016) Modelling of different enzyme productions by solid-state fermentation on several agro-industrial residues. Appl Microbiol Biotechnol 100(22):9555–9566
Espinoza FH, Farrell A, Nourse JL, Chamberlin HM, Gileadi O, Morgan DO (1998) Cak1 is required for Kin28 phosphorylation and activation in vivo. Mol Cell Biol 18:6365–6373
Georges A, Gopaul D, Wilkes CD, Aiach NG, Novikova E, Barrault MB, Alibert O, Soutourina J (2019) Functional interplay between Mediator and RNA polymerase II in Rad2/XPG loading to the chromatin. Nucleic Acids Res 47:8988–9004
Hagiwara D, Sakamoto K, Abe K, Gomi K (2016) Signaling pathways for stress responses and adaptation in Aspergillus species: stress biology in the post-genomic era. Biosci Biotech Bioch 80:1667–1680
Huberman LB, Coradetti ST, Glass NL (2017) Network of nutrient-sensing pathways and a conserved kinase cascade integrate osmolarity and carbon sensing in Neurospora crassa. P Natl Acad Sci USA 114:E8665–E8674
Knippa K, Peterson DO (2013) Fidelity of RNA polymerase II transcription: role of Rpb9 in error detection and proofreading. Biochemistry 52:7807–7817
Knoll ER, Zhu ZL, Sarkar D, Landsman D, Morse RH (2020) Kin28 depletion increases association of TFIID subunits Taf1 and Taf4 with promoters in Saccharomyces cerevisiae. Nucleic Acids Res 48:4244–4255
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 7:1870
Kuo MH, Nadeau ET, Grayhack EJ (1997) Multiple phosphorylated forms of the Saccharomyces cerevisiae Mcm1 protein include an isoform induced in response to high salt concentrations. Mol Cell Biol. 17:819–832
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12:323
Li WT, Li SS (2017) Facilitators and repressors of transcription-coupled DNA repair in Saccharomyces cerevisiae. Photochem Phoyobiol 93:259–267
Li J, Liu GD, Chen M, Li ZH, Qin YQ, Qu YB (2013) Cellodextrin transporters play important roles in cellulase induction in the cellulolytic fungus Penicillium oxalicum. Appl Microbiol Biotechnol 97:10479–10488
Li N, Kunitake E, Endo Y, Aoyama M, Kanamaru K, Kimura M, Kato M, Kobayashi T (2016a) Involvement of an SRF-MADS protein McmA in regulation of extracellular enzyme production and asexual/sexual development in Aspergillus nidulans. Biosci Biotech Bioch 80:1820–1828
Li N, Kunitake E, Endo Y, Aoyama M, Kimura M, Koyama Y, Kobayashi T (2016b) McmA-dependent and -independent regulatory systems governing expression of ClrB-regulated cellulase and hemicellulase genes in Aspergillus nidulans. Mol Microbiol 102:810–826
Li ZH, Liu GD, Qu YB (2017) Improvement of cellulolytic enzyme production and performance by rational designing expression regulatory network and enzyme system composition. Bioresource Technol 245:1718–1726
Li CX, Zhao S, Luo XM, Feng JX (2020) Weighted gene co-expression network analysis identifies critical genes for the production of cellulase and xylanase in Penicillium oxalicum. Front Microbiol 11:520–520
Liao GY, Zhao S, Zhang T, Li CX, Liao LS, Zhang FF, Luo XM, Feng JX (2018) The transcription factor TpRfx1 is an essential regulator of amylase and cellulase gene expression in Talaromyces pinophilus. Biotechnol Biofuels 11:276
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods 25:402–408
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Madi L, McBride SA, Bailey LA, Ebbole DJ (1997) rco-3, a gene involved in glucose transport and conidiation in Neurospora crassa. Genetics 146:499
Marone A, Trably E, Carrère H, Prompsy P, Guillon F, Joseph-Aimé M, Barakat A, Fayoud N, Bernet N, Escudié R (2019) Enhancement of corn stover conversion to carboxylates by extrusion and biotic triggers in solid-state fermentation. Appl Microbiol Biotechnol 103(1):489–503
Martínez-Soto D, Ruiz-Herrera J (2017) Functional analysis of the MAPK pathways in fungi. Revista Iberoamericana de Micología 34:192–202
Mok J, Kim PM, Lam HY, Piccirillo S, Zhou X, Jeschke GR, Sheridan DL, Parker SA, Desai V, Jwa M, Cameroni E, Niu H, Good M, Remenyi A, Ma JL, Sheu YJ, Sassi HE, Sopko R, Chan CS, De Virgilio C, Hollingsworth NM, Lim WA, Stern DF, Stillman B, Andrews BJ, Gerstein MB, Snyder M, Turk BE (2010) Deciphering protein kinase specificity through large-scale analysis of yeast phosphorylation site motifs. Sci Signal 3:ra12
Nguyen EV, Imanishi SY, Haapaniemi P, Yadav A, Saloheimo M, Corthals GL, Pakula TM (2016) Quantitative site-specific phosphoproteomics of Trichoderma reesei signaling pathways upon induction of hydrolytic enzyme production. J Proteome Res 15:457–467
Park HS, Yu JH (2012) Genetic control of asexual sporulation in filamentous fungi. Curr Opin Microbiol 15:669–677
Perez-Cuesta U, Aparicio-Fernandez L, Guruceaga X, Martin-Souto L, Abad-Diaz-de-Cerio A, Antoran A, Buldain I, Hernando FL, Ramirez-Garcia A, Rementeria A (2020) Melanin and pyomelanin in Aspergillus fumigatus: from its genetics to host interaction. Int Microbiol 23:55–63
Priegnitz BE, Brandt U, Pahirulzaman KA, Dickschat JS, Fleißner A (2015) The AngFus3 mitogen-activated protein kinase controls hyphal differentiation and secondary metabolism in Aspergillus niger. Eukaryot Cell 14:602–615
Salat-Canela C, Paulo E, Sánchez-Mir L, Carmona M, Ayté J, Oliva B, Hidalgo E (2017) Deciphering the role of the signal- and Sty1 kinase-dependent phosphorylation of the stress-responsive transcription factor Atf1 on gene activation. J Biol Chem 292:13635–13644
Segorbe D, Pietro AD, Pérez-Nadales E, Turrà D (2017) Three Fusarium oxysporum mitogen-activated protein kinases (MAPKs) have distinct and complementary roles in stress adaptation and cross-kingdom pathogenicity. Mol Plant Pathol 18:912–924
Su LH, Zhao S, Jiang SX, Liao XZ, Duan CJ, Feng JX (2017) Cellulase with high β-glucosidase activity by Penicillium oxalicum under solid state fermentation and its use in hydrolysis of cassava residue. World J Microbiol Biotechnol 33:37
Suh H, Hazelbaker DZ, Soares LM, Buratowski S (2013) The C-terminal domain of RPB1 functions on other RNA polymerase II subunits. Mol Cell 51:850–858
Taus T, Köcher T, Pichler P, Paschke C, Schmidt A, Henrich C, Mechtler K (2011) Universal and confident phosphorylation site localization using phosphoRS. J Proteome Res 10:5354–5362
Tong SM, Feng MG (2019) Insights into regulatory roles of MAPK-cascaded pathways in multiple stress responses and life cycles of insect and nematode mycopathogens. Appl Microbiol Biotechnol 103:577–587
Wang MY, Zhang ML, Li L, Dong Y, Jiang YM, Liu KM, Zhang RQ, Jiang BJ, Niu KL, Fang X (2017) Role of Trichoderma reesei mitogen-activated protein kinases (MAPKs) in cellulase formation. Biotechnol Biofuels 10:99
Wang L, Zhao S, Chen XX, Deng QP, Li CX, Feng JX (2018) Secretory overproduction of a raw starch-degrading glucoamylase in Penicillium oxalicum using strong promoter and signal peptide. Appl Microbiol Biotechnol 102:9291–9301
Wong SHJ, Dumas B (2010) Ste12 and ste12-like proteins, fungal transcription factors regulating development and pathogenicity. Eukaryot Cell 9:480–485
Xiong Y, Coradetti ST, Li X, Gritsenko MA, Clauss T, Petyuk V, Camp D, Smith R, Cate JHD, Yang F, Glass NL (2014) The proteome and phosphoproteome of Neurospora crassa in response to cellulose, sucrose and carbon starvation. Fungal Genet Biol 72:21–33
Xu QS, Yan YS, Feng JX (2016) Efficient hydrolysis of raw starch and ethanol fermentation: a novel raw starch-digesting glucoamylase from Penicillium oxalicum. Biotechnol Biofuels 9:216
Yan YS, Zhao S, Liao LS, He QP, Xiong YR, Wang L, Li CX, Feng JX (2017) Transcriptomic profiling and genetic analyses reveal novel key regulators of cellulase and xylanase gene expression in Penicillium oxalicum. Biotechnol Biofuels 10:279
Zhao S, Yan YS, He QP, Yang L, Yin X, Li CX, Mao LC, Liao LS, Huang JQ, Xie SB, Nong QD, Zhang Z, Jing L, Xiong YR, Duan CJ, Liu JL, Feng JX (2016) Comparative genomic, transcriptomic and secretomic profiling of Penicillium oxalicum HP7-1 and its cellulase and xylanase hyper-producing mutant EU2106, and identification of two novel regulatory genes of cellulase and xylanase gene expression. Biotechnol Biofuels 9:203
Zhao S, Liao XZ, Wang JX, Ning YN, Li CX, Liao LS, Liu Q, Jiang Q, Gu LS, Fu LH, Yan YS, Xiong YR, He QP, Su LH, Duan CJ, Luo XM, Feng JX (2019a) Transcription factor Atf1 regulates expression of cellulase and xylanase genes during solid-sate fermentation of ascomycetes. Appl Environ Microbiol 85:e01226–e01219
Zhao S, Liu Q, Wang JX, Liao XZ, Guo H, Li CX, Zhang FF, Liao LS, Luo XM, Feng JX (2019b) Differential transcriptomic profiling of filamentous fungus during solid-state and submerged fermentation and identification of an essential regulatory gene PoxMBF1 that directly regulated cellulase and xylanase gene expression. Biotechnol Biofuels 12:103
Funding
This work was financially supported by the National Natural Science Foundation of China (grants 31760023 and 31660305), the Autonomous Research Project of State Key Laboratory for Conservation and Utilization of Subtropical Agro-bioresources (SKLCUSA-a201902 and SKLCUSA-a201923), the Training Program for 1000 Young and Middle-aged Key Teachers in Guangxi at 2019, and the One Hundred Person Project of Guangxi.
Author information
Authors and Affiliations
Contributions
JXF supervised this study and revised the manuscript. SZ co-supervised and directed all experiments and revised the manuscript. BM conducted construction of deletion mutants, measurement of enzymatic activities, transcriptomes, and phosphoproteomes, and wrote the manuscript. YNN performed construction of complementary strain and enzymatic activity assay. CXL conducted bioinformatic analysis. DT and HG conducted RT-qPCR analysis. XMP performed phenotypic analyses. XML was involved in preparation of experimental materials.
Corresponding authors
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 5280 kb)
Rights and permissions
About this article
Cite this article
Ma, B., Ning, YN., Li, CX. et al. A mitogen-activated protein kinase PoxMK1 mediates regulation of the production of plant-biomass-degrading enzymes, vegetative growth, and pigment biosynthesis in Penicillium oxalicum. Appl Microbiol Biotechnol 105, 661–678 (2021). https://doi.org/10.1007/s00253-020-11020-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-11020-0