Abstract
Larch caterpillars are widely distributed in the Great Xing’an boreal forests; however, the relationship between caterpillar defoliation dynamics and climatic factors is poorly understood. The aims of this study are to investigate the primary weather conditions that might influence forest defoliation and to identify the most important life stage of the larch caterpillar at which forest defoliation might be mitigated by incorporating more inhibitory influences from climatic factors. The life cycle of the larch caterpillar was partitioned into four stages and multiple linear regression and mixed effect models were combined with a relative weight analysis approach to evaluate the importance and influence of meteorological variables on defoliation dynamics. The results show that warmer temperatures in growing seasons and overwintering periods can increase the defoliation area, while rainy and humid growing seasons decrease the defoliation area. Total precipitation during the early instar larval period had the greatest power to explain the variance in defoliation dynamics and had a very strong inhibitory effect, followed by the accumulative temperatures of the late instar larval period which had a positive impact, and precipitation during the middle instar larval period which had a negative impact. Weather conditions during the early instar larval period had the greatest influence on the area defoliated and accounted for 40% of the explained variance. This study demonstrates that climatic warming and drying will increase the risk of larch caterpillar outbreaks in the Great Xing’an Mountains.
Similar content being viewed by others
Introduction
Forest insects are critical destructive agents that can directly exert stress effects on forest growth or even lead to substantial tree mortality, thereby decreasing timber production and ecosystem services related to water and soil retention and carbon sinks (Dale et al. 2001; Kurz et al. 2008; Medvigy et al. 2012; Zhang et al. 2014; Flower and Gonzalez-Meler 2015). As a natural disturbance, forest insects may indirectly interact with other disturbances (e.g., wildfire, drought and pathogens), and amplify their negative effects on ecosystem resilience, which may trigger irreparable ecosystem shifts (Kane et al. 2017; Seidl et al. 2017). Monitoring and predicting outbreaks of forest insects are topics that have received increasing attention due to the benefits produced by the implementation of practical governance measures in advance to improve forest health and maintain valuable ecological functions.
Climatic changes, especially warming and drought, are important driving factors that regulate insect outbreaks through direct impacts on insect physiology and behavior and indirect impacts on host plants and predators (Liebhold et al. 2000; Bale et al. 2002; Bentz et al. 2010; Kolb et al. 2016). For instance, minimum winter temperatures are one of the predominant environmental obstacles that forest insects must overcome in order to survive (Han et al. 2005; Bale and Hayward 2010). Many studies have shown that high-latitude regions will experience stronger warming trends in winter (Dale et al. 2001; Bale and Hayward 2010; Venette 2013), which may significantly shorten the duration of winter and decrease the inhibitory effects of cold temperatures on forest insects. Under such trends, it is predicted that more insects will successfully survive winter, negatively affecting forest health. Warming is expected to dramatically influence the population dynamics of forest insects; therefore, temperature is frequently regarded as an important predictor of forest insect outbreaks (Yamamura et al. 2005; Bentz et al. 2016; Zidon et al. 2016).
Similarly, changes in precipitation patterns and associated water deficits are also identified as critical factors controlling the breeding and growth of insects. Heavy rainfall or hailstorms during the breeding period can mechanically injure or kill eggs that adhere to foliage or twigs. Cold and rainy weather can inhibit the dispersal and feeding of newly hatched larvae and slow their growth (McManus et al. 1980; Rouault et al. 2006; Jaworski and Hilszczański 2013). Moist environments may induce the dispersal of diseases caused by bacteria, fungi, or viruses, which may influence the population and growth of forest insects (McManus et al. 1980; Han et al. 2004). Moreover, physiological stress caused by warming and/or water deficits that increase host plant susceptibility to forest insects are also well-documented globally (Bale et al. 2002; Rouault et al. 2006; van Mantgem et al. 2009; Allen et al. 2010; Kolb et al. 2016).
Defoliating insects such as the pine caterpillar, gypsy moth and spruce budworm are the major insects causing disturbance across Eurasian and North American boreal forests, and these groups have received increasing public attention as a result of their pervasive influence and the weak resistance of boreal ecosystems to insect disturbance (Gray 2008; Bouchard and Pothier 2010; Weed et al. 2013; Zhang et al. 2014). Previous studies have shown that the population dynamics of forest insects are largely driven by the spatial synchrony with extreme climatic events (Han et al. 2005; Wang et al. 2007; Hódar et al. 2012) and by phenological asynchrony with host plants or predators (Liebhold et al. 2000; Bjornstad et al. 2010; Pureswaran et al. 2015). These studies have provided notable conclusions that have helped to disentangle the mechanisms underlying how environmental factors regulate insect population dynamics. Most of those discussions have focused on forest insects that have relatively short life histories, while the influences of climatic factors on the population dynamics of forest insects with longer life histories, i.e., insects that require several years to complete a whole lifecycle, have received relatively little attention.
Forest insects in boreal regions usually require multiple years to develop body growth and physiological functions (Bale et al. 2002). This multiyear life history increases the complexity of research endeavors attempting to clarify the climate-driven outbreak mechanisms of these forest insects. Previous studies have emphasized the linkages between inter- or intra-annual temperature variations and population dynamics of forest insects, while much less is known on other climatic factors that may also exert similar or consistent influences on the insect population at different developmental stages. Since forest insects may develop various adaption strategies to overcome severe exogenous environments (Bale et al. 2002; Pimentel et al. 2010), clarifying the linkages between weather conditions and population dynamics may be helpful for forming a comprehensive understanding about regulatory mechanisms that occur at the regional scale. Specifically, identifying an insect’s sensitive life-history stage in terms of local weather conditions and quantifying the relative importance of those variables on population dynamics are of notable practical significance for forest insect management.
Compared with boreal ecosystems in other regions, the forests in the Great Xing’an Mountains, which serve as an important ecological barrier to extreme climate (e.g., the Siberian cold current) and natural disasters (e.g., floods) in northeastern China, may be particularly vulnerable to climate change effects because they are located within a border zone between boreal and temperate climates (Ni 2011). The primary type of forest insect disturbance that has caused serious forest losses is the larch caterpillar, Dendrolimus superans Butler, which requires two years to complete its lifecycle in this region (Yuan et al. 2008; Chen et al. 2010). An extreme outbreak of larch caterpillar occurred from 1989 to 1991, which caused over one million ha of forest damage in the Great Xing’an Mountains (Liu et al. 1994). The mean population density of insects was approximately 283 individuals per tree, and the maximum might have reached 2000 per tree (Liu et al. 2002). Thus, the objectives of this study were: (1) to identify the primary weather conditions that govern fluctuations in larch caterpillar populations and are linked to its special life-history traits; and, (2) to determine the critical life stage in which the insect population would be sensitive to the influence of climatic factors. To achieve these goals, historical larch caterpillar records and daily meteorological observations from six forestry bureaus in the Great Xing’an boreal forests were used to examine these relationships. We evaluated the relative influence of variables related to temperature, relative humidity and precipitation, which were calculated from daily observations of four life stages of the larch caterpillar, on episodic outbreaks of this defoliator based on a linear regression model and linear mixed effect model.
Materials and methods
Study site
The study area is located within six forestry bureaus (local administrative agencies) of the Great Xing’an Mountains in Northeast China (121° 10′ 41″–127° 01′ 06″ E, 50° 05′ 53″–53° 33′ 38″ N), Jiagedaqi, Songling, Amuer, Xinlin, Hanjiayuan and Shibazhan, which occupy more than half of the total area and are administered by Heilongjiang Province (Fig. 1). The topography in the Great Xing’an Mountains is gently undulating with an average slope of 10°. The elevation ranges from 130 m in the northeast to 1500 m in the southwest. The climate is mid-latitude continental cold-temperate, the mean annual temperature is − 1.8 °C, and the monthly mean January and July temperatures are − 20 °C and 30 °C, respectively. The mean annual precipitation is approximately 550 mm and presents great inter-annual variations, with more than 60% falling between May and October. The frost-free period is less than 130 days in the south and less than 100 days in the north. The forests in the study area are classified as cool-temperate coniferous forests and form the southern extension of the eastern Siberian boreal forests. Dahurian larch (Larix gmelinii Rupr.) and white birch (Betula platyphylla Sukaczev) are the dominant canopy species, mixed with Scotch pine (Pinus sylvestris var. mongolica Litv.), Korean spruce (Picea koraiensis Nakai), Mongolian oak (Quercus mongolica Fisch. ex Ledeb.), and two species of aspen (Populus davidiana Dode and Populus suaveolens Fisch.) (Fang et al. 2015). As a result of heavy forest harvesting since the middle of the last century and frequent wildfires, the forest landscape of the study area is primarily composed of middle and young age stands.
Life cycle of the larch caterpillar
The larch caterpillar, D. superans, is an endemic species in China that can seriously endanger coniferous species, such as Dahurian larch (its preferred host plant), Scotch pine and Korean spruce (Liu et al. 1994; Yu et al. 2016). Generally the insect produces one generation per year in the southern part of northeastern China, although it has one generation every two years in our study area due to the relatively short period of appropriate temperatures for development occur (Liu et al. 1994; Tian et al. 1998). Previous studies have found that the larch caterpillar has ninth instar larvae and begins to overwinter during the second to fourth instar stages at the end of September (Fig. 2). The caterpillars then leave the host tree and remain in ground litter or grasses from October to the following April. Low temperatures during winter can freeze the larvae, which then thaw when temperatures rebound. At the end of April of the following year, the caterpillars will ascend the tree and consume the foliage until the second overwintering period, which begins during the fifth to seventh instar stages (Yue et al. 1996; Chen et al. 2010). After entering its second diapause period, the surviving larvae will continue their development and pupate at the end of May. After a 20-day pupal stage, adults lay eggs on the needles during the second half of June. The second generation of larch caterpillar eggs will hatch during June and July and eventually change to the larval stage in approximately 15 days. By reviewing its life cycle, we can identify key temporal nodes at which climatic factors may regulate the population by directly impacting their physiology or by modifying the phenology of their host plants, thus indirectly impacting the larch caterpillars.
Data source
Larch caterpillars can impact substantially forest growth in a short time, although these are usually nonlethal to host tree species; however, when the degree of defoliation is over 75%, tree mortality may occur (Yu and Yin 1980). With regards to the low mortality induced by the larch caterpillar, the defoliation area is considered a more reliable indicator to reflect the adverse consequences of larch caterpillars from a forest health perspective than tree mortality. In this study, the area of defoliated forests was used as a proxy to reflect the dynamics of the larch caterpillar population density. Historical defoliation data between 2003 and 2017 were obtained from annual reports of the six local forest pest control and quarantine stations, which are completed and maintained by the these forestry bureaus (Fig. 3). Thirty-four valid records of annual defoliation areas of larch caterpillar were obtained from these six forestry bureaus. These records were double-checked by a higher administrative department as the figures of defoliation area related to financial inputs for pest control. Years without valid records were considered those without pronounced outbreak events.
The estimation of defoliation area was based on field surveys carried out along the predesigned routes that covered the stands where larch caterpillar outbreaks occurred as well as new 100 m × 100 m sampling plots. Populations of larch caterpillars were checked using the shaking-off collection method for 20 larch trees within each plot. A forest sub-compartment was classified as a defoliated area when the population density of the insect was greater than 20 caterpillars per tree according to a national guideline of defoliation severity classification. Twenty caterpillars per tree is an empirical population threshold for reporting pine caterpillar disaster, and a lower population density does not warrant a report to higher levels of the forest insect pest control department (see http://www.forestpest.org/forecasting/forecastmethods/). The defoliation area was estimated based on the proportion of defoliated sub compartments. Although this estimation was somewhat coarse, such defoliation data are the most reliable official records of larch caterpillar outbreaks in the study area.
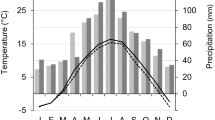
Historical weather data for the five forestry bureaus were obtained from the Climatic Data Center, National Meteorological Information Center, and the China Meteorological Administration (http://data.cma.cn/). These weather stations are part of a network of meteorological stations across China. Daily statistics of weather variables, such as average temperature, precipitation, and relative humidity were obtained from 1975 to 2017 (Fig. 4). The historical weather observations show increasing average annual temperatures and decreasing relative humidities and precipitation, which suggest a warmer and drier climate in the Great Xiang’an Mountains.
By reviewing the biological characteristics of the larch caterpillar, previous studies have revealed that larvae at the seventh to ninth instar stages consume more than 97% of the total leaf amount during the entire life cycle (Yu and Yin 1980; Liu et al. 1994; Yue et al. 1996). Because larch caterpillar field surveys were mainly carried out between May and June, middle and late instar larvae coexisted in the forests. It was hypothesized that the defoliation area during a specific year was primarily caused by late instar larvae. The life cycle of the late instar larvae was followed, and based on a review of previous studies, 15 weather variables were selected that were considered to impact survival rates during key life stages (Table 1). These included the breeding period, the early instar larval period, the middle instar larval period and the late instar larval period. The 15 weather variables were grouped into four categories based on their temporal information that closely relates each variable to the four key life stages. At the same time, these variables were also separated into three categories, as they reflect the corresponding environmental conditions of temperature, precipitation and relative humidity.
Statistical analyses
A multiple linear regression (MLR) model was used to analyze the relative importance of weather conditions on the forest area defoliated by larch caterpillars. To avoid overfitting as a result of the limited dataset, we used a maximum of three explanatory variables to establish the models. Because explanatory variables selected from the same weather category often represent high collinearity, one meteorological variable was chosen from each of the three weather categories (i.e., temperature, precipitation, relative humidity) to establish a candidate model. To reduce redundant candidate models, we first used the Pearson correlation coefficient (r) generated by the bivariate correlation test to exclude explanatory variables that have very low linear correlations (r < 0.30) with the response variable. By visualizing the relationships between variables, exponential transformation was carried out for accumulated daily temperatures below the supercooling point (SCP, − 22 °C) during the first overwintering period (EATO) and accumulated daily temperatures below the SCP during the second overwintering period (MATO) to ensure that these two variables were linearly correlated with the response variable. Cook’s distance was calculated for each correlation test to eliminate outliers that have Cook’s distance greater than three times the means. A stepwise regression approach was also used as a supplement to choose a candidate model because some predictor variables may not pass the bivariate correlation test but can still explain a high portion of the remaining variation when interacting with the most dominant explanatory variable.
Although such modeling strategy could reduce collinearity among explanatory variables, a variance inflation factor (VIF) was still used to detect the collinearity between explanatory variables following the protocol of Zuur et al. (2010). A general rule of thumb is if the VIF is greater than 10, then collinearity is considered high, and if greater than 4, then collinearity occurs. To ensure that significant collinearity problems did not occur, we simply excluded the models if their explanatory variables had VIF values higher than 4. The VIF values were calculated using the “car” package in the R statistical software program (R Core Development Team).
The coefficient of determination (R2) is the most popular quantification of model performance and thus is frequently used as a basis to evaluate the relative importance of explanatory variables. However, this measure may be problematic when independently quantifying the unique contribution of an explanatory variable to the model fitting because of the collinearity among explanatory variables. Although a stringent process was carried out to mitigate collinearity, there was still a need to discriminate the direct effects from the joint effects with other variables when partitioning the explained variance. The proportional contribution of each explanatory variable to the coefficient of determination was calculated based on the relative weight analysis approach proposed by Johnson (2000). The “relaimpo” package was used to calculate the relative weights for each variable contained in the regression model (Grömping 2006). To evaluate the individual strengths of three different categories of meteorological variables on modeling the defoliation area of larch caterpillars, the accumulated relative weights of the explanatory variables was calculated. It was assumed that meteorological variables that had higher accumulated values of relative weight would provide more explanatory power. Similarly, the accumulated relative weights for each stage of the life cycle were also compared to demonstrate the effect of weather conditions on specific life stages to identify the factor with the greatest importance for determining the defoliation area of the larch caterpillar.
The influence of the meteorological variables on the defoliation area of larch caterpillars was evaluated based on the values and directions (i.e., positive or negative) of the estimated slope coefficients. Since the defoliation area may be related to other biotic or abiotic conditions belonging to a given forestry bureau, such as the inherent nature of local forests, the topography and human activities associated with pest control, these underlying variations are not well reflected by the meteorological variables. The influence (slopes) of weather conditions on the defoliation area may be biased if the forestry bureau-related variations (intercepts) are assumed to be uniform in the models. The nested structure of our sampling data allows us to add a flexible random effect in the regression model to address individual differences among forestry bureaus. Such mixed effects modeling consists of fixed and random components. Here, the explanatory variables derived from weather observations are defined as fixed effects and specified the forestry bureaus as the random effect. We used the “lem4” package to run the linear mixed effects (LME) model with random intercepts (Bates et al. 2014).
Many studies have noted the importance of standardizing variables into comparable scales for multivariate analysis because the explanatory variables have different ranges and represent different meteorological meanings. Thus, the raw data was standardized based on a Z-score scaling method to ensure that their influence was measured at comparable scales. A negative Z-score indicates that the raw value is below the mean, and a positive Z-score indicates that the raw value is above the mean. However, current LME functions cannot quantify the statistical significance of the fixed effects from explanatory variables in terms of the p-value; therefore, we performed a likelihood ratio (LR) test to verify the significance (at alpha = 5% level) of an explanatory variable when introducing this variable into the LME model. Significant variables were not selected based solely on the quantification of the LR test outputs because the reliability of testing fixed effects is still debatable (Bolker et al. 2009). Akaike information criterion (AIC) was also used to evaluate the significance of a variable to the LME model. In general, lower AIC values represent less information loss. Similar to the LR test, we calculated the difference in the AIC when applied to the LME model with and without a specific variable. A positive AIC difference indicates that the variable can reduce information loss. The combined subset of variables selected from these two test methods were identified as key regulators to provide a more comprehensive interpretation.
Results
Evaluation of model performance
Nine of fifteen meteorological variables were well correlated (|r|> 0.30) with the defoliation area of the larch caterpillar (Appendix Fig. S1); they included four temperature variables, three precipitation variables and two relative humidity variables. Together with one model selected by the stepwise regression approach, 25 (i.e., 4 × 3 × 2 + 1) MLR models and corresponding LME models were generated (Table 2). The maximum VIF values of the MLR models were all less than 2, which indicated weak collinearity among the explanatory variables. According to the model outputs, 12 MLR models out of 25 explained 28.3–42.1% of the variance in the annual defoliation area among different forestry bureaus at the p < 0.05 level. In addition, four MLR models could explain approximately 25% variance in defoliation area at the p < 0.10 level. The MLR model, whose explanatory variables consisted of the accumulated temperature (daily temperature above 8 °C) of the growing season (LATG), total precipitation during the growing season (LTP) and mean daily relative humidity during the growing season (EMRH), explained the maximum variance in the defoliation area of the larch caterpillar, while variable combinations such as the accumulated temperature (daily temperature above 8 °C) of the growing season (MATG)-total precipitation during the growing season (ETP)-EMRH, LATG-total precipitation during the growing season (MTP)-EMRH, LATG-MTP-mean daily relative humidity during the growing season (MMRH) and MATG-MTP-EMRH explained over 38.0% of the variance. In addition, the results showed that MLR models with temperature variables associated with the growth period had more explanatory power than models with temperature variables associated with the overwintering period. By incorporating a random effect in terms of spatial location, the results showed residual standard errors (RSE) improved for seven of these 25 models, including four of the significant models.
Importance analysis
According to the relative importance of explanatory variables in terms of their proportional contribution to the coefficient of determination, the relative importance of selected variables varied considerably between different models. The results clearly show that the ETP, which represented the total precipitation of the early instar larvae period, exerted the most relative importance on improving the model explanatory power because it independently contributed approximately 15% (13.5–25.7%) to the explained variance for most of the models (Fig. 5). In addition to the ETP, the LATG (11.1–17.8%), MTP (8.3–16.3%) and EMRH (7.2–15.9%) also independently contributed over 10% explained variance for most of the models when they were included. The two winter temperature variables contributed the least explained variance (EATO: 1.3–7.5%; MATO: 0.5–5.3%) when they were combined with two other kinds of meteorological variables.
Relative importance of nine selected variables based on the proportion of explained variance. See Table 1 for definitions of the variable abbreviations
The relative importance of explanatory variables were grouped based on their temporal attributes (Fig. 6a) and physical meanings (Fig. 6b) and the relative proportion of the total explained variance for each group was calculated. The results indicate that nearly 40% of the total explained variance was contributed by variables related to the early instar larval period, especially the ETP and EMRH (Fig. 6a). Four meteorological variables belonging to the middle instar larval period together contributed over 31% of the total explained variance, while the LATG, which was the only variable of the late instar larval period, contributed over 24% of the total explained variance. In contrast, the only variable selected from the breeding period contributed less than 5% to the total explained variance, indicating the relatively weak importance of weather conditions during this period for determining the defoliation area two years later. These results emphasize the key role of weather conditions during the early and middle instar larval periods in determining the population dynamics of the late instar larvae in future springs. When the relative weights of the explanatory variables were grouped into the three categories of relative humidity, temperature and precipitation (Fig. 6b), variables related to precipitation contributed 40% of the total explained variance, while variables related to temperature and relative humidity explained approximately 33.6% and 26%, respectively. The precipitation variables were more strongly correlated with the larch caterpillar population density than the other two weather parameters.
Effects of weather conditions on defoliation area
The results of the tests of significance based on the LR test and AIC showed that 35 out of 72 slope coefficients estimated from the LME models were statistically significant (Table 2). The directions of effects from selected meteorological variables were consistent with the correlation tests. The results show that temperature-related variables had significant positive effects on the defoliated area (Fig. 7). Higher EATO and MATO indicated relatively warmer winters, and higher MATG and LATG hotter growing seasons. Although the standardized slope coefficients varied among the models, two cumulative temperature variables of growing seasons, LATG and MATG, clearly exhibited a stronger influence on the area of defoliation than the two cumulative low winter temperature variables, EATO and MATO, which can reflect the degree of coldness in two overwintering periods. Our results also clearly show that variables related to precipitation and relative humidity exhibited strong inhibitory effects on the defoliation area of the larch caterpillar, especially the total amount of precipitation during the early instar larvae period (i.e., ETP), which produced the steepest slope in the negative direction. In general, precipitation variables have a stronger negative influence than relative humidity variables at the same life stage. Overall, the results demonstrate that a growing season with frequent hot and dry periods will increase the defoliation area.
Discussion
The results indicate that weather conditions are closely associated with the area defoliated in the study area. Nine variables exerted a notable influence on caterpillar outbreak, and the variables ETP, LATG, MTP and EMRH exerted a considerable influence on improving model performance. Weather variables during the early instar larval and breeding stages played key roles in regulating the populations of larch caterpillars. Among these variables, total precipitation at the early instar larval period was the most important variable because it contributed the greatest proportion of the explained variance in the linear models. The coefficients generated by the mixed effects models indicate that the defoliation area could decrease significantly with increasing total precipitation during the early and middle instar larval periods, as well as during the breeding period. This finding suggests that increased precipitation had a very strong inhibitory effect on larch caterpillar outbreaks. In contrast, drought (low precipitation) in the growing season can exacerbate defoliation, especially under high temperatures and low relative humidity. Hot droughts during the growing season can increase the suitability of host plants for feeding and growth of insect larvae. Water deficits induced by hot, dry conditions can lead to stomatal closure of leaves, reducing transpiration and maintaining water potential; however, the trees will become warmer and nutrients will be concentrated at the same time, which is favorable to defoliating insects (Fleming and Candau 1998). Previous studies revealed that heavy rainfall or continuous drizzle may inhibit the development of forest insects. Heavy rainfall may kill the eggs or young larvae due to strong mechanical damage or water immersion (Chen et al. 2010; Khaliq et al. 2014). This hypothesis is strongly supported by the negative correlations of precipitation variables observed in this study.
The effects of temperature on forest insects have attracted considerable attention in recent decades. Insect disturbances by both defoliators and wood-boring insects are projected to intensify under warmer climates during both the growing season and overwintering period. Our results clearly show that larch caterpillar outbreaks in the study area follow such dynamics. Although a cool climate is usually observed during the growing season in boreal forests, such as in our study area, under a warmer growing season, the feeding activities of insects will increase which will cause more severe forest defoliation. Warmer temperatures will enable larch caterpillars to develop faster and attain higher survival rates than under normal conditions. The notably positive influence and explanatory power of temperature in the growing season of the late and middle instar larval periods supports this notion. In addition, the modulation effect of winter temperature is another important limitation for larch caterpillar outbreaks. The results clearly show positive but nonlinear relationships between the accumulated daily temperatures below the supercooling point of − 22 °C and the area defoliated. The duration of the supercooling period could dramatically determine the area defoliated, suggesting that warming in winter could facilitate outbreaks of larch caterpillars the following year because more insects survived overwintering. This finding is in accordance with numerous studies, highlighting the ecological importance of the supercooling period as a predominant agent regulating population dynamics of forest insects (Renault et al. 2002). Among the temperature variables, our results did not identify significant relationships between extreme low winter temperatures and area of defoliation, suggesting that the larvae of larch caterpillars during diapause are insensitive or tolerant to ephemeral coldness in winter.
Relative humidity reflects the saturation status of water vapor in the atmosphere at a given temperature, which is different from the amount of moisture available as measured by precipitation. At a lower relative humidity, moisture evaporates at a faster rate. The effects of relative humidity on forest insect outbreaks have received less attention than the effects of temperature and precipitation, primarily because of the high variability of relative humidity and the difficulty interpreting its effects (Tauber et al. 1998; Jaworski and Hilszczański 2013). Low relative humidity can lead to water deficits in host plants under the combined impact of temperature and precipitation; therefore, relative humidity can indirectly influence insect development and survivability (Rouault et al. 2006). Although relative humidity had less overall relative importance than precipitation or temperature for explaining variance in the area of defoliation, relative humidity of the early and middle instar larval periods had consistently negative effects on defoliation area relative to precipitation. The EMRH had a stronger effect and explanatory power than most of the temperature and precipitation variables. Humid environments, especially those that have almost continuous drizzle, may exacerbate spore dispersal of Beauveria bassiana, recognized as an important pathogenic bacterium that can trigger a sharp decrease in larch caterpillar populations because the early instar larvae are vulnerable to disease (Xu et al. 2008a; b). Nevertheless, exposure to high or insufficient humidity for lengthy periods can generate adverse effects on development or survivability of insects because such conditions can directly modulate the interior water balance of the insect body (Tauber et al. 1998; Han et al. 2004; Chang and Gao 2008) Persistent low humidity can lead to high mortality in insects via dehydration (Rouault et al. 2006). From this perspective, relationships between relative humidity and defoliation area were expected to be nonlinear as a result of adverse effects at both extremes. However, we did not think that such nonlinearity needed to be reflected in our data because the relative humidity of the growing season seldom reaches the critical point at which larch caterpillars are killed by persistent dehydration. Moreover, defoliators can obtain moisture by feeding to maintain water balance (Tauber et al. 1998; Rouault et al. 2006).
Our study provides information for the development of scientific pest control strategies to maintain forest health and resilience. The results highlight the important role of weather conditions in modifying the intensity of defoliation caused by larch caterpillars. Furthermore, this research indicates that weather conditions can be linked to specific life-history traits of the caterpillar, which may be useful in developing pest control plans. For instance, the results show that weather conditions at the early instar larval stage had the strongest influence on the extent of the defoliation. Extreme weather conditions may disrupt the activities of insects and the biological habits upon which insects depend to successfully survive, develop, or mate. Current pest control efforts can be improved by incorporating the inhibitory effects of such weather conditions. For example, the introduction of bacteria that function as pathogens, important biological control agents of the larch caterpillar, can be carried out on days with higher relative humidity during the early instar larval period. Additionally, our results demonstrate the ecological importance of supercooling periods in regulating defoliation. Such natural regulation mechanisms can be applied to improve the effectiveness of pest control techniques by destroying the insect’s overwintering shelters (e.g., surface litter) in geographical hotspots of high insect populations during the early instar larval period with extreme cold temperatures.
There are several limitations regarding the use of defoliation data to predict the outbreak of larch caterpillars using empirical models. The first concerns the quality of the defoliation data. Although uniform standards of field pest investigation are strictly enforced by the national forest service and local pest control stations, these datasets may be subject to bias due to changes in the forestry bureaus, different field investigators, and/or the legacy effects of past insect outbreaks and pest control intensity. The other limitation involves the temporal and spatial integrality of the defoliation datasets. Since we only collected some temporally fragmented datasets based on official forest insect reports, our results may represent only a general overview of the relationships and relative importance of meteorological factors involved in regulating larch caterpillar defoliation dynamics. Although the results are reasonable and were mostly cross-validated by previous studies, it is possible that extrapolating insect outbreaks based on the empirical models generated in our study may have missing defoliation information. However, several recent studies claim that remote sensing can be applied to retrieve continuous forest disturbance history (e.g., insect, fire and harvesting) at a decadal time scale (Neigh et al. 2014; McDowell et al. 2015; Meigs et al. 2015). Monitoring forest changes related to larch caterpillars over a greater spatial area would be beneficial for obtaining a more comprehensive understanding of the relationships between weather conditions and larch caterpillar outbreaks.
Furthermore, the impacts of forest insects can be influenced by biotic factors associated with stand structure and composition (Charbonneau et al. 2012; Robert et al. 2012). Changes in stand structure and composition as a result of forest management operations such as thinning, harvesting and planting may alter the stand’s susceptibility to insects and ultimately cause spatial variability in tree mortality and defoliation severity. Although such impacts work at finer spatial scales than the one used in this study, we believe that they may induce uncertainty that our models cannot explain.
Conclusions
Meteorological factors are closely associated with the defoliation dynamics of larch caterpillars in the Great Xing’an boreal forests. By considering the special life-history traits of the caterpillar in the study area, the analyses revealed that the meteorological variables calculated from the precipitation at the early instar larval period explained most of the variance in the defoliation area. Precipitation and relative humidity had significant negative effects on the regulation of defoliation by larch caterpillars. In contrast, temperature variables had significant positive effects, especially the accumulative temperatures during the late and middle instar larval periods. The duration of supercooling periods during the early and middle instar larval periods can substantially limit the area defoliated. Overall, the study demonstrates that the early instar larval period is the key period during which the population of larch caterpillars may be controlled by exploiting the regulatory effects of weather. In this context, the results show that larch caterpillar outbreaks may occur if the early and middle instar larvae experience severe drought during their growing seasons or warm winters during overwintering. These results may provide valuable information to help forestry agencies prevent and control forest insect pests and adapt to novel insect dynamics against the background of global climate change.
References
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259(4):660–684
Bale JS, Hayward SA (2010) Insect overwintering in a changing climate. J Exp Biol 213(6):980–994
Bale JS, Masters GJ, Hodkinson ID, Awmack C, Bezemer TM, Brown VK, Butterfield J, Buse A, Coulson JC, Farrar J, Good JEG, Harrington R, Hartley S, Jones TH, Lindroth RL, Press MC, Symrnioudis I, Watt AD, Whittaker JB (2002) Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Glob Change Biol 8(1):1–16
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv preprint arXiv 1406:5823
Bentz BJ, Régnière J, Fettig CJ, Hansen EM, Hayes JL, Hicke JA, Kelsey RG, Negrón JF, Seybold SJ (2010) Climate change and bark beetles of the western United States and Canada: direct and indirect effects. Bioscience 60(8):602–613
Bentz BJ, Duncan JP, Powell JA (2016) Elevational shifts in thermal suitability for mountain pine beetle population growth in a changing climate. Forestry 89(3):271–283
Björkman C, Bylund H, Klapwijk MJ, Kollberg I, Schroeder M (2011) Insect pests in future forests: More severe problems? Forests 2(2):474–485
Bjornstad ON, Robinet C, Liebhold AM (2010) Geographic variation in North American gypsy moth cycles: subharmonics, generalist predators, and spatial coupling. Ecology 91(1):106–118
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White JSS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24(3):127–135
Bouchard M, Pothier D (2010) Spatiotemporal variability in tree and stand mortality caused by spruce budworm outbreaks in eastern Quebec. Can J For Res 40(1):86–94
Chang XN, Gao H (2008) Effects of environmental moisture and precipitation on insects: a review. Chin J Ecol 27(4):619–625 (in Chinese)
Charbonneau D, Lorenzetti F, Doyon F, Mauffette Y (2012) The influence of stand and landscape characteristics on forest tent caterpillar (Malacosoma disstria) defoliation dynamics: the case of the 1999–2002 outbreak in northwestern Quebec. Can J For Res 42(10):1827–1836
Chen SH, Zhang XL (2011) Meteorological conditions and area forecast for occurrence of Dendrolimus superans in Inner Mongolia. J Northeast For Univ 39(11):135–136 (in Chinese)
Chen HW, Hu YM, Chang Y, Bu RC, Liu M, Liu N (2010) Advances in the researches of Dendrolimus superans of Daxingan’ Mountain of China. J Northwest For Univ 26(1):119–127 (in Chinese)
Dale VH, Joyce LA, McNulty S, Neilson RP, Ayres MP, Flannigan MD, Hanson PJ, Irland LC, Lugo AE, Peterson CJ (2001) Climate change and forest disturbances: climate change can affect forests by altering the frequency, intensity, duration, and timing of fire, drought, introduced species, insect and pathogen outbreaks, hurricanes, windstorms, ice storms, or landslides. Bioscience 51(9):723–734
Fang L, Yang J, Zu JX, Li GC, Zhang JS (2015) Quantifying influences and relative importance of fire weather, topography, and vegetation on fire size and fire severity in a Chinese boreal forest landscape. For Ecol Manag 356:2–12
Fleming RA, Candau JN (1998) Influences of climatic change on some ecological processes of an insect outbreak system in Canada’s boreal forests and the implications for biodiversity. Environ Monit Assess 49(2–3):235–249
Flower CE, Gonzalez-Meler MA (2015) Responses of temperate forest productivity to insect and pathogen disturbances. Annu Rev Plant Biol 66:547–569
Gray DR (2008) The relationship between climate and outbreak characteristics of the spruce budworm in eastern Canada. Clim Change 87(3–4):361–383
Grömping U (2006) Relative importance for linear regression in R: the package relaimpo. J Stat Softw 17(1):1–27
Han RD, He Z, Ge F (2004) Factors influencing the population dynamics of pine caterpillars. Chin Bull Entomol 41(6):504–511 (in Chinese)
Han RD, Ge F, Yardim EN, He Z (2005) The effect of low temperatures on diapause and non-diapause larvae of the pine caterpillar, Dendrolimus tabulaeformis Tsai et Liu (Lepidoptera: Lasiocampidae). Appl Entomol Zool 40(3):429–435
Hódar JA, Zamora R, Cayuela L (2012) Climate change and the incidence of a forest pest in Mediterranean ecosystems: can the North Atlantic Oscillation be used as a predictor? Clim Change 113(3–4):699–711
Hou ZG, Li MS, Wang L, Xiao JJ, Huang DP, Wang CY (2012) Impacts of precipitation variations on crop diseases and pests in China. Sci Agric Sinica 45(10):1935–1945 (in Chinese)
Jaworski T, Hilszczański J (2013) The effect of temperature and humidity changes on insects development their impact on forest ecosystems in the expected climate change. For Res Pap 74(4):345–355
Johnson JW (2000) A heuristic method for estimating the relative weight of predictor variables in multiple regression. Multivar Behav Res 35(1):1–19
Kane JM, Varner JM, Metz MR, van Mantgem PJ (2017) Characterizing interactions between fire and other disturbances and their impacts on tree mortality in western U.S. Forest. For Ecol Manag 405:188–199
Khaliq A, Javed M, Sohail M, Sagheer M (2014) Environmental effects on insects and their population dynamics. J Entomol Zool Stud 2(2):1–7
Kolb TE, Fettig CJ, Ayres MP, Bentz BJ, Hicke JA, Mathiasen R, Stewart JE, Weed AS (2016) Observed and anticipated impacts of drought on forest insects and diseases in the United States. For Ecol Manag 380:321–334
Kurz WA, Dymond CC, Stinson G, Rampley GJ, Neilson ET, Carroll AL, Ebata T, Safranyik L (2008) Mountain pine beetle and forest carbon feedback to climate change. Nature 452(7190):987–990
Liebhold A, Elkinton J, Williams D, Muzika RM (2000) What causes outbreaks of the gypsy moth in North America? Popul Ecol 42(3):257–266
Lin HF, Fan MZ, Li ZZ, Hu C (1998) Pathogenic effect of Beauveria bassiana infected on Dendrolimus punctatus under different temperature and humidity. Chin J Appl Ecol 9(2):195–200 (in Chinese)
Liu KY, Meng QF, Wang LC, Zhang GC, Pang ZH, Wang F, Liu JM, Ma DM (1994) Study on the bionomics of Dendrolimus superans in Daxingan’ Mountain. J Northeast For Univ 22(2):1–5 (in Chinese)
Liu ZM, Yan M, Zhang XD (2002) Methodical study on monitoring wide-range forest insect pest by meteorsat. J Nat Disasters 11(3):109–114 (in Chinese)
McDowell NG, Coops NC, Beck PS, Chambers JQ, Gangodagamage C, Hicke JA, Huang CY, Kennedy R, Krofcheck DJ, Litvak M, Meddens AJ, Muss J, Negron-Juarez R, Peng C, Schwantes AM, Swenson JJ, Vernon LJ, Williams AP, Xu C, Zhao M, Running SW, Allen CD (2015) Global satellite monitoring of climate-induced vegetation disturbances. Trends Plant Sci 20(2):114–123
McManus ML, Schneeberger N, Reardon R, Mason G (1980) The gypsy moth. US Department of Agriculture, Forest Service. https://www.srs.fs.usda.gov/pubs/10894 Accessed 3 Dec 2020
Medvigy D, Clark K, Skowronski N, Schäfer K (2012) Simulated impacts of insect defoliation on forest carbon dynamics. Environ Res Lett 7(4):045703
Meigs GW, Kennedy RE, Gray AN, Gregory MJ (2015) Spatiotemporal dynamics of recent mountain pine beetle and western spruce budworm outbreaks across the Pacific Northwest Region, USA. For Ecol Manag 339:71–86
Neigh C, Bolton D, Williams J, Diabate M (2014) Evaluating an automated approach for monitoring forest disturbances in the Pacific northwest from logging, fire and insect outbreaks with Landsat time series data. Forests 5(12):3169–3198
Ni J (2011) Impacts of climate change on Chinese ecosystems: key vulnerable regions and potential thresholds. Reg Environ Change 11(1):49–64
Pimentel C, Ferreira C, Nilsson JÅ (2010) Latitudinal gradients and the shaping of life-history traits in a gregarious caterpillar. Biol J Lin Soc 100(1):224–236
Pureswaran DS, De Grandpré L, Paré D, Taylor A, Barrette M, Morin H, Régnière J, Kneeshaw DD (2015) Climate-induced changes in host tree-insect phenology may drive ecological state-shift in boreal forests. Ecology 96(6):1480–1491
Renault D, Salin C, Vannier G, Vernon P (2002) Survival at low temperatures in insects: what is the ecological significance of the supercooling point? CryoLetters 23(4):217–228
Robert LE, Kneeshaw D, Sturtevant BR (2012) Effects of forest management legacies on spruce budworm (Choristoneura fumiferana) outbreaks. Can J For Res 42(3):463–475
Rouault G, Candau JN, Lieutier F, Nageleisen LM, Martin JC, Warzée N (2006) Effects of drought and heat on forest insect populations in relation to the 2003 drought in Western Europe. Ann For Sci 63(6):613–624
Seidl R, Thom D, Kautz M, Martin-Benito D, Peltoniemi M, Vacchiano G, Wild J, Ascoli D, Petr M, Honkaniemi J, Lexer MJ, Trotsiuk V, Mairota P, Svoboda M, Fabrika M, Nagel TA, Reyer CPO (2017) Forest disturbances under climate change. Nat Clim Change 7(6):395–402
Tang HY, Niu BL (2010) Influence of meteorological factors on reproduction of Dendrolimus superans and forecast of spawning quantity. J Northeast For Univ 38(1):84–87 (in Chinese)
Tauber MJ, Tauber CA, Nyrop JP, Villani MG (1998) Moisture, a vital but neglected factor in the seasonal ecology of insects: hypotheses and tests of mechanisms. Environ Entomol 27(3):523–530
Tian F, Bi XH, Shen GT, Zhou B, Li WB (1998) Study on the forecast technique and control index of Larch Caterpillar of a cycle in two years. For Res 11(4):49–56 (in Chinese)
van Mantgem PJ, Stephenson NL, Byrne JC, Daniels LD, Franklin JF, Fule PZ, Harmon ME, Larson AJ, Smith JM, Taylor AH, Veblen TT (2009) Widespread increase of tree mortality rates in the western United States. Science 323(5913):521–524
Venette R (2013) Incorporating climate change into pest risk models for forest pathogens: a role for cold stress in an era of global warming? NeoBiota 18:131–150
Wang J, Ji LZ, Khomutova M (2007) Relationships between forest insect pest occurrence area and meteorological factors in GreatXing’an Mountains of Heilongjiang Province, Northeast China. Chin J Ecol 26:673–677 (in Chinese)
Weed AS, Ayres MP, Hicke JA (2013) Consequences of climate change for biotic disturbances in North American forests. Ecol Monogr 83(4):441–470
Xia RX, Xu LH (1993) Study on the super-cooling point of the overwintering larvae of three kinds of pine caterpillar. Scientia Silver Sinicae 29(2):186–189 (in Chinese)
Xu GY, Jiang ZM, Fang SA, Chen ZL (2008a) Different larvae ages and amount of Beauveria bassiana spores in forests influence the white muscardine to pine moth. Agric Tech Serv 25(8):148–150
Xu GY, Li DX, Chen JD, Wang J, Li Q, Li TB (2008b) The effect of meteological factors and forest factors on occurence of Dendrolimus tabulaeformis (Lepidoptera: Lasiocampidae). Agric Tech Serv 25(8):161–162
Yamamura K, Yokozawa M, Nishimori M, Ueda Y, Yokosuka T (2005) How to analyze long-term insect population dynamics under climate change: 50-year data of three insect pests in paddy fields. Popul Ecol 48(1):31–48
Yu EY, Yin TL (1980) Larch caterpillar prediction research-leaf amount, mutilated, total amount of damage and amount of forest stand needle volume measurement. J Jinlin For Sci Technol 1(980):56–59 (in Chinese)
Yu WY, Ji RP, Feng R, Zhao XL, Wu JW, Zhang SJ, Zhang YS (2013) The study on meteorological forecast methods of dendrolimus occurrence area in Liaoning. Chin Agric Sci Bull 29(13):56–59 (in Chinese)
Yu Y, Fang L, Wang FX, Ma W, Tong YF, Yang J (2016) Spatial distribution of larch caterpillar and its driving factors. Chin J Ecol 35(5):1285–1293 (in Chinese)
Yuan F, Luo YQ, Shi J, Keliövaara K, Qi GX, Li XJ, Han YS, Chen C (2008) Invasive sequence and ecological niche of main insect borers of Larix gmelinii forest in Aershan. Inner Mong For Stud China 10(1):9–13
Yue SK, Wang ZY, Huang YQ, Song YH, Ping GY, Qu HX, Lei Y (1996) Bionomicas and natural enemies of Dendrolimus Superans. J Northeast For Univ 24(4):2–8 (in Chinese)
Zeng JP, Ge F, Su JW, Wang Y (2008) The effect of temperature on the diapause and cold hardiness of Dendrolimus tabulaeformis (Lepidoptera: Lasiocampidae). Eur J Entomol 105:599–606
Zhang FL, Jing TZ, Wang ZY, Xie SP, Zhang H (2011) Path analysis of meteorological factors and population Dendrolimus superans density. Hubei Agric Sci 50(19):3977–3979 (in Chinese)
Zhang XQ, Lei YC, Ma ZH, Kneeshaw D, Peng CH (2014) Insect-induced tree mortality of boreal forests in eastern Canada under a changing climate. Ecol Evol 4(12):2384–2394
Zidon R, Tsueda H, Morin E, Morin S (2016) Projecting pest population dynamics under global warming: the combined effect of inter- and intra-annual variations. Ecol Appl 26(4):1198–1210
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1(1):3–14
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: The work was supported by the National Key R&D Program of China (2017YFA0604403).
The online version is available at http://www.springerlink.com.
Corresponding editor: Tao Xu
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fang, L., Yu, Y., Fang, G. et al. Effects of meteorological factors on the defoliation dynamics of the larch caterpillar (Dendrolimus superans Butler) in the Great Xing’an boreal forests. J. For. Res. 32, 2683–2697 (2021). https://doi.org/10.1007/s11676-020-01277-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-020-01277-6