Abstract
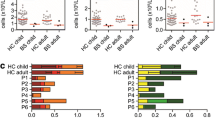
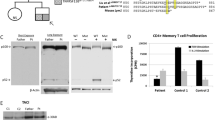
Single allele mutations in the Cell Division Control protein 42 homolog (CDC42) gene were recently shown to cause Takenouchi-Kosaki syndrome with diverse manifestations. These include persistent mild thrombocytopenia with large platelet size, severe developmental delay, growth retardation, facial dysmorphism, and other neurodevelopmental and hematological anomalies. CDC42 deficiency might also cause myelofibrosis, myeloproliferation, and severe autoinflammation. CDC42 closely interacts with the Wiskott-Aldrich Syndrome Protein, but little is still known about the immune abnormalities associated with CDC42 deficiency. Detailed immune evaluations were performed in a patient diagnosed with a CDC42 Tyr64Cys mutation. The 19-year-old female suffered from recurrent pneumonia, otitis media, and bacteremia, which resolved at 10 years of age, concordant with the initiation of amoxicillin prophylaxis. In addition, the patient had frequent viral upper respiratory tract infections, which resolved without need for medical interventions. Immune evaluations demonstrated decreased immunoglobulin levels, inability to maintain antibody responses, progressive decline in the number of CD19+ B cells, and decreased switched memory B cells. There was also a decrease in CD4+ and CD8+ T cells, markedly reduced naïve T cells, and intermittent depressed proliferation of T cells to stimulation. Natural killer cells’ number and functions were normal. However, no opportunistic infections were observed, nor was there evidence for autoinflammation. CDC42 deficiency might also be associated with decline in T and B cell function. Therefore, immunity in patients with CDC42 defects should be closely monitored, particularly among those with frequent infections or systemic autoinflammation.


Similar content being viewed by others
Abbreviations
- CDC42:
-
Cell Division Control protein 42 homolog
- HLH:
-
Hemophagocytic lymphohistiocytosis
- MAS:
-
Macrophage activation syndrome
- VB:
-
Variable beta
- WASP:
-
Wiskott-Aldrich Syndrome Protein
References
Heasman SJ, Ridley AJ. Mammalian rho GTPases: new insights into their functions from in vivo studies. Nat Rev Mol Cell Biol. 2008;9(9):690–701. https://doi.org/10.1038/nrm2476Review.
Dvorsky R, Ahmadian MR. Always look on the bright site of rho: structural implications for a conserved intermolecular interface. EMBO Rep. 2004;5(12):1130–6 Review.
Ferru-Clément R, Fresquet F, Norez C, Métayé T, Becq F, et al. Involvement of the Cdc42 pathway in CFTR post-translational turnover and in its plasma membrane stability in airway epithelial cells. PLoS One. 2015;10(3):e0118943. https://doi.org/10.1371/journal.pone.0118943.
Takenouchi T, Kosaki R, Niizuma T, Hata K, Kosaki K. Macrothrombocytopenia and developmental delay with a de novo CDC42 mutation: yet another locus for thrombocytopenia and developmental delay. Am J Med Genet A. 2015;167A(11):2822–5. https://doi.org/10.1002/ajmg.a.37275.
Takenouchi T, Okamoto N, Ida S, Uehara T, Kosaki K. Further evidence of a mutation in CDC42 as a cause of a recognizable syndromic form of thrombocytopenia. Am J Med Genet A. 2016;170A(4):852–5. https://doi.org/10.1002/ajmg.a.37526.
Martinelli S, Krumbach OHF, Pantaleoni F, Coppola S, Amin E, et al. Functional dysregulation of CDC42 causes diverse developmental phenotypes. Am J Hum Genet. 2018. https://doi.org/10.1016/j.ajhg.2017.12.015.
Bucciol G, Pillay B, Casas-Martin J, Delafontaine S, Proesmans M, et al. Systemic inflammation and myelofibrosis in a patient with Takenouchi-Kosaki syndrome due to CDC42 Tyr64Cys mutation. J Clin Immunol. 2020;40(4):567–70. https://doi.org/10.1007/s10875-020-00742-5.
Lam MT, Coppola S, Krumbach OHF, Prencipe G, Insalaco A, et al. A novel disorder involving dyshematopoiesis, inflammation, and HLH due to aberrant CDC42 function. J Exp Med. 2019;216(12):2778–99. https://doi.org/10.1084/jem.20190147.
Gernez Y, de Jesus AA, Alsaleem H, Macaubas C, Roy A, et al. Severe autoinflammation in 4 patients with C-terminal variants in cell division control protein 42 homolog (CDC42) successfully treated with IL-1β inhibition. J Allergy Clin Immunol. 2019;144(4):1122–1125.e6. https://doi.org/10.1016/j.jaci.2019.06.017.
Szczawinska-Poplonyk A, Ploski R, Bernatowska E, Pac M. A novel CDC42 mutation in an 11-year old child manifesting as syndromic immunodeficiency, autoinflammation, hemophagocytic lymphohistiocytosis, and malignancy: a case report. Front Immunol. 2020;11:318. https://doi.org/10.3389/fimmu.2020.00318.
Verboon JM, Mahmut D, Kim AR, Nakamura M, Abdulhay NJ, Nandakumar SK, et al. Infantile myelofibrosis and myeloproliferation with CDC42 dysfunction. J Clin Immunol. 2020;40(4):554–66. https://doi.org/10.1007/s10875-020-00778-7.
He T, Huang Y, Ling J, Yang J. A new patient with NOCARH syndrome due to CDC42 defect. J Clin Immunol. 2020;40(4):571–5. https://doi.org/10.1007/s10875-020-00786-7.
Bekhouche B, Tourville A, Ravichandran Y, Tacine R, Abrami L, et al. A toxic palmitoylation of Cdc42 enhances NFkappaB signaling and drives a severe autoinflammatory syndrome. J Allergy Clin Immunol. 2020;146(5):1201–4. e8. https://doi.org/10.1016/j.jaci.2020.03.020.
Motokawa M, Watanabe S, Nakatomi A, Kondoh T, Matsumoto T, Morifuji K, et al. A hot-spot mutation in CDC42 (p.Tyr64Cys) and novel phenotypes in the third patient with Takenouchi-Kosaki syndrome. J Hum Genet. 2018;63(3):387–90. https://doi.org/10.1038/s10038-017-0396-5.
Shearer WT, Rosenblatt HM, Gelman RS, Oyomopito R, Plaeger S, Stiehm ER, et al. Lymphocyte subsets in healthy children from birth through 18 years of age: the Pediatric AIDS Clinical Trials Group P1009 study. J Allergy Clin Immunol. 2003;112(5):973–80.
Bryceson YT, Pende D, Maul-Pavicic A, Gilmour KC, Ufheil H, Vraetz T, et al. A prospective evaluation of degranulation assays in the rapid diagnosis of familial hemophagocytic syndromes. Blood. 2012;119(12):2754–63. https://doi.org/10.1182/blood-2011-08-374199.
Wonderlich J, Shearer G, Livingstone A, Brooks A. Induction and measurement of cytotoxic T lymphocyte activity. Curr Protoc Immunol. 2006;Chapter 3:Unit 3.11. https://doi.org/10.1002/0471142735.im0311s72.
Chemin K, Bohineust A, Dogniaux S, Tourret M, Guégan S, Miro F, et al. Cytokine secretion by CD4+ T cells at the immunological synapse requires Cdc42-dependent local actin remodeling but not microtubule organizing center polarity. J Immunol. 2012;189(5):2159–68. https://doi.org/10.4049/jimmunol.1200156.
Guo F, Zhang S, Tripathi P, Mattner J, Phelan J, Sproles A, et al. Distinct roles of Cdc42 in thymopoiesis and effector and memory T cell differentiation. PLoS One. 2011;6(3):e18002. https://doi.org/10.1371/journal.pone.0018002.
Burbage M, Keppler SJ, Gasparrini F, Martínez-Martín N, Gaya M, Feest C, et al. Cdc42 is a key regulator of B cell differentiation and is required for antiviral humoral immunity. J Exp Med. 2015;212(1):53–72. https://doi.org/10.1084/jem.20141143.
Carlin LM, Evans R, Milewicz H, Fernandes L, Matthews DR, Perani M, et al. A targeted siRNA screen identifies regulators of Cdc42 activity at the natural killer cell immunological synapse. Sci Signal. 2011;4(201):ra81. https://doi.org/10.1126/scisignal.2001729.
Su HC, Orange JS. The growing spectrum of human diseases caused by inherited CDC42 mutations. J Clin Immunol. 2020;40(4):551–3. https://doi.org/10.1007/s10875-020-00785-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kashani, P., Marwaha, A., Feanny, S. et al. Progressive decline of T and B cell numbers and function in a patient with CDC42 deficiency. Immunol Res 69, 53–58 (2021). https://doi.org/10.1007/s12026-020-09168-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-020-09168-y