Abstract
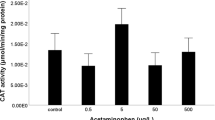
Biological organisms are constantly challenged by xenobiotics and have evolved mechanisms to reduce, neutralize, or repair toxic outcomes. The various chemical defenses all utilize energy, but their specific costs and impacts on energy budgets are currently unknown. In this study, the energetic costs associated with the induction and substrate transport of the efflux transporter P-glycoprotein (P-gp [ABCB1, MDR1]) were examined in rainbow trout. An intraperitoneal injection of the P-gp inducer clotrimazole (0, 0.1, 1.0, and 10 mg/kg) increased P-gp activity (as measured by a competitive rhodamine 123 transport assay in hepatocytes) in a dose-dependent manner reaching a maximum induction of 2.8-fold. Maximum P-gp induction occurred at 50 h post-administration with the highest dose; significant induction of P-gp activity remained elevated over constitutive values until the last sampling time point (168 h). In vitro measurements of hepatocyte respiration indicated that basal P-gp activity transporting R123 as a substrate did not significantly increase respiration rates (range 18.0 to 23.2 ng O2/min/106 cells); however, following the induction of P-gp by clotrimazole and exposure to the P-gp substrate R123, respiration rates increased significantly (3.52-fold) over baseline values. Using whole animal respirometry, it was shown that respiration rates in fish exposed to R123 only or induced with clotrimazole were not different from controls (range 1.2 to 2.1 mg O2/kg/min); however, respiration rates were significantly increased in fish with induced P-gp levels and also exposed to R123. This work indicates that basal and induced levels of P-gp activity do not incur significant energetic costs to fish; however, upon induction of P-gp and concomitant substrate exposures, energetic costs can increase and could pose challenges to organisms facing limited energy resources.





Similar content being viewed by others
Data availability
Data is available upon request to the corresponding author.
References
Albertus JA, Laine RO (2001) Enhanced xenobiotic transporter expression in normal teleost hepatocytes: response to environmental and chemotherapeutic toxins. J Exp Biol 204:217–227
Altenberg GA, Vanoye CG, Horton JK, Reuss L (1994) Unidirectional fluxes of rhodamine 123 in multidrug-resistant cells: evidence against direct drug extrusion from the plasma membrane. Proc Natl Acad Sci U S A 91:4654–4657. https://doi.org/10.1073/pnas.91.11.4654
Ambudkar SV, Cardarelli CO, Pashinsky I, Stein WD (1997) Relation between the turnover number for vinblastine transport and for vinblastine-stimulated ATP hydrolysis by human P-glycoprotein. J Biol Chem 272:21160–21166. https://doi.org/10.1074/jbc.272.34.21160
Annilo T, Chen ZQ, Shulenin S, Costantino J, Thomas L, Lou H, Stefanov S, Dean M (2006) Evolution of the vertebrate ABC gene family: analysis of gene birth and death. Genomics 88:1–11. https://doi.org/10.1016/j.ygeno.2006.03.001
Bains OS, Kennedy CJ (2004) Energetic costs of pyrene metabolism in isolated hepatocytes of rainbow trout, Oncorhynchus mykiss. Aquat Toxicol 67:217–226. https://doi.org/10.1016/j.aquatox.2004.01.008
Bains OS, Kennedy CJ (2005) Alterations in respiration rate of isolated rainbow trout hepatocytes exposed to the P-glycoprotein substrate rhodamine 123. Toxicology 214:87–98. https://doi.org/10.1016/j.tox.2005.06.002
Bard SM (1998) A biological index to predict pulp mill pollution levels. Water Environ Res 70:108–122. https://doi.org/10.2175/106143098x126955
Bard SM, Bello SM, Hahn ME, Stegeman JJ (2002a) Expression of P-glycoprotein in killifish (Fundulus heteroclitus) exposed to environmental xenobiotics. Aquat Toxicol 59:237–251. https://doi.org/10.1016/S0166-445X(01)00256-9
Bard SM, Woodin BR, Stegeman JJ (2002b) Expression of P-glycoprotein and cytochrome P450 1A in intertidal fish (Anoplarchus purpurescens) exposed to environmental contaminants. Aquat Toxicol 60:17–32. https://doi.org/10.1016/S0166-445X(01)00272-7
Bauer B, Hartz AMS, Fricker G, Miller DS (2004) Pregnane X receptor up-regulation of P-glycoprotein expression and transport function at the blood-brain barrier. Mol Pharmacol 66:413–419. https://doi.org/10.1124/mol.66.3
Bauer B, Yang X, Hartz AMS, Olson ER, Zhao R, Kalvass JC, Pollack GM, Miller DS (2006) In vivo activation of human pregnane X receptor tightens the blood-brain barrier to methadone through p-glycoprotein up-regulation. Mol Pharmacol 70:1212–1219. https://doi.org/10.1124/mol.106.023796
Bauer B, Hartz AMS, Pekcec A, Toellner K, Miller DS, Potschka H (2008) Seizure-induced up-regulation of P-glycoprotein at the blood-brain barrier through glutamate and cyclooxygenase-2 signaling. Mol Pharmacol 73:1444–1453. https://doi.org/10.1124/mol.107.041210
Bayne BL, Moore MN, Widdows J, Livingstone DR, Salkeld P (1979) Measurement of the responses of individuals to environmental stress and pollution: studies with bivalve molluscs. Philos Trans R Soc Lond Ser B Biol Sci 286:563–581. https://doi.org/10.1098/rstb.1979.0046
Beamish RJ, Lockhart WL, Van Loon JC, Harvey HH (1975) Long term acidification of a lake and resulting effects on fishes. Ambio 4:98–102
Beyers DW, Sikoski PJ (1994) Acetylcholinesterase inhibition in federally endangered Colorado squawfish exposed to carbaryl and malathion. Environ Toxicol Chem 13:935–939. https://doi.org/10.1002/etc.5620130612
Beyers DW, Rice JA, Clements WH (1999a) Evaluating biological significance of chemical exposure to fish using a bioenergetics-based stressor-response model. Can J Fish Aquat Sci 56:823–829. https://doi.org/10.1139/f99-005
Beyers DW, Rice JA, Clements WH, Henry CJ (1999b) Estimating physiological cost of chemical exposure: integrating energetics and stress to quantify toxic effects in fish. Can J Fish Aquat Sci 56:814–822. https://doi.org/10.1139/f99-006
Brattsen LB (1988) Enzymic adaptations in leaf-feeding insects to host–plant allelochemicals. J Chem Ecol 14:1919–1940
Brett JR, Zala CA (1975) Daily pattern of nitrogen excretion and oxygen consumption of sockeye salmon (Oncorhynchus nerka) under controlled conditions. J Fish Res Board Can 32:2479–2486. https://doi.org/10.1139/f75-285
Burt RK, Thorgeirsson SS (1988) Coinduction of MDR-1 multidrug-resistance and cytochrome P-450 genes in rat liver by xenobiotics. J Natl Cancer Inst 80:1383–1386. https://doi.org/10.1093/jnci/80.17.1383
Calow P (1989) Proximate and ultimate responses to stress in biological systems. Biol J Linn Soc 37:173–181. https://doi.org/10.1111/j.1095-8312.1989.tb02101.x
Calow P (1991) Physiological costs of combating chemical toxicants: ecological implications. Comp Biochem Physiol C Comp. https://doi.org/10.1016/0742-8413(91)90110-F
Calow P, Sibly RM (1990) A physiological basis of population processes: ecotoxicological implications. Funct Ecol 4:283. https://doi.org/10.2307/2389587
Chan KM, Davies PL, Childs S, Veinot L, Ling V (1992) P-glycoprotein genes in the winter flounder, Pleuronectes americanus: isolation of two types of genomic clones carrying 3′ terminal exons. BBA - Gene Struct Expr 1171:65–72. https://doi.org/10.1016/0167-4781(92)90140-U
Congdon JD, Vitt LJ, van Loben Sels RC, Ohmart RD (1982) The ecological significance of water flux rates in arboreal desert lizards of the genus Urosaurus. Physiol Zool 55:317–322. https://doi.org/10.1086/physzool.55.3.30157895
Cooper PS, Vogelbein WK, Van Veld PA (1996) Immunohistochemical and immunoblot detection of P-glycoprotein in normal and neoplastic fish liver. In: Ostrander GK (ed) Techniques in Aquatic Toxicology. CRC Press, Boca Raton
Cooper PS, Vogelbein WK, Van Veld PA (1999) Altered expression of the xenobiotic transporter P-glycoprotein in liver and liver tumours of mummichog (Fundulus heteroclitus) from a creosote-contaminated environment. Biomarkers 4:48–58. https://doi.org/10.1080/135475099230994
de Lange ECM (2004) Potential role of ABC transporters as a detoxification system at the blood-CSF barrier. Adv Drug Deliv Rev 56:1793–1809. https://doi.org/10.1016/j.addr.2004.07.009
Deeley RG, Westlake C, Cole SP (2006) Transmembrane transport of endo- and xenobiotics by mammalian ATP-binding cassette multidrug resistance proteins. Physiol Rev 86:849–899. https://doi.org/10.1152/physrev.00035.2005
Diaz de Cerio O, Bilbao E, Cajaraville MP, Cancio I (2012) Regulation of xenobiotic transporter genes in liver and brain of juvenile thicklip grey mullets (Chelon labrosus) after exposure to prestige-like fuel oil and to perfluorooctane sulfonate. Gene 498:50–58. https://doi.org/10.1016/j.gene.2012.01.067
Doi AM, Holmes E, Kleinow KM (2001) P-glycoprotein in the catfish intestine: inducibility by xenobiotics and functional properties. Aquat Toxicol 55:157–170. https://doi.org/10.1016/S0166-445X(01)00180-1
Duval WS, Fink RP, Geen GH (1981) An aquatic respirometer for use in sublethal studies. Water Res 15:817–822. https://doi.org/10.1016/0043-1354(81)90135-4
El Hafny B, Chappey O, Piciotti M, Debray M, Boval B, Roux F (1997) Modulation of P-glycoprotein activity by glial factors and retinoic acid in an immortalized rat brain microvessel endothelial cell line. Neurosci Lett 236:107–111. https://doi.org/10.1016/S0304-3940(97)00679-4
Endicott JA, Ling V (1989) The biochemistry of P-glycoprotein-mediated multidrug resistance. Annu Rev Biochem 58:137–171. https://doi.org/10.1146/annurev.biochem.58.1.137
Epel D (1998) Use of multidrug transporters as first lines of defense against toxins in aquatic organisms. In: Comparative Biochemistry and Physiology - A Molecular and Integrative Physiology, pp 23–28. https://doi.org/10.1016/S1095-6433(98)10005-3
Epel D (2003) Protection of DNA during early development: adaptations and evolutionary consequences. In: Evolution and Development, pp 83–88. https://doi.org/10.1046/j.1525-142X.2003.03013.x
Eytan GD, Regev R, Oren G, Assaraf YG (1996) The role of passive transbilayer drug movement in multidrug resistance and its modulation. J Biol Chem 271:12897–12902. https://doi.org/10.1074/jbc.271.22.12897
Fardel O, Lecureur V, Corlu A, Guillouzo A (1996) P-glycoprotein induction in rat liver epithelial cells in response to acute 3-methylcholanthrene treatment. Biochem Pharmacol 51:1427–1436. https://doi.org/10.1016/0006-2952(96)00081-0
Ferté J (2000) Analysis of the tangled relationships between P-glycoprotein-mediated multidrug resistance and the lipid phase of the cell membrane. Eur J Biochem 267:277–294. https://doi.org/10.1046/j.1432-1327.2000.01046.x
Fischer S, Loncar J, Zaja R, Schnell S, Schirmer K, Smital T, Luckenbach T (2011) Constitutive mRNA expression and protein activity levels of nine ABC efflux transporters in seven permanent cell lines derived from different tissues of rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 101:438–446. https://doi.org/10.1016/j.aquatox.2010.11.010
Fischer S, Klüver N, Burkhardt-Medicke K, Pietsch M, Schmidt AM, Wellner P, Schirmer K, Luckenbach T (2013) Abcb4 acts as multixenobiotic transporter and active barrier against chemical uptake in zebrafish (Danio rerio) embryos. BMC Biol 11:69. https://doi.org/10.1186/1741-7007-11-69
Foley WJ (1992) Nitrogen and energy retention and acid–base status in the common ringtail possum, Pseudocheirus peregrinus, evidence of the effects of absorbed allelochemicals. Physiol Zool 65:403–421
Fry F (1947) Effects of the environment on animal activity. Publ Out Fish Res Lab 55:1–62
Gant TW, O’Connor CK, Corbitt R, Thorgeirsson U, Thorgeirsson SS (1995) In vivo induction of liver P-glycoprotein expression by xenobiotics in monkeys. Toxicol Appl Pharmacol 133:269–276. https://doi.org/10.1006/taap.1995.1151
Gill KA, Walsh PJ (1990) Effects of temperature on metabolism of benzo[a]pyrene by toadfish (Opsanus beta) hepatocytes. Can J Fish Aquat Sci 47:831–837. https://doi.org/10.1139/f90-096
Gottesman MM, Pastan I (1993) Biochemistry of multidrug resistance mediated by the multidrug transporter. Annu Rev Biochem 62:385–427. https://doi.org/10.1146/annurev.bi.62.070193.002125
Guarino AM, Pritchard JB, Anderson JB, Rall DP (1974) Tissue distribution of [14C]DDT in the lobster after administration via intravascular or oral routes or after exposure from ambient sea water. Toxicol Appl Pharmacol 29:277–288. https://doi.org/10.1016/0041-008X(74)90065-9
Helder T (1981) Effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on early life stages of rainbow trout (Salmo Gairdneri, Richardson). Toxicology 19:101–112. https://doi.org/10.1016/0300-483X(81)90092-5
Hildebrand JL, Bains OS, Lee DSH, Kennedy CJ (2009) Functional and energetic characterization of P-gp-mediated doxorubicin transport in rainbow trout (Oncorhynchus mykiss) hepatocytes. Comp Biochem Physiol C Toxicol Pharmacol 149:65–72. https://doi.org/10.1016/j.cbpc.2008.07.003
Hopkins WA, Rowe CL, Congdon JD (1999) Elevated trace element concentrations and standard metabolic rate in banded water snakes (Nerodia fasciata) exposed to coal combustion wastes. Environ Toxicol Chem 18:1258–1263. https://doi.org/10.1897/1551-5028(1999)018<1258:ETECAS>2.3.CO;2
Ihunnah CA, Jiang M, Xie W (2011) Nuclear receptor PXR, transcriptional circuits and metabolic relevance. Biochim Biophys Acta (BBA) - Mol Basis Dis 1812:956–963. https://doi.org/10.1016/j.bbadis.2011.01.014
Ito K, Suzuki H, Hirohashi T, Kume K, Shimizu T, Sugiyama Y (1997) Molecular cloning of canalicular multispecific organic anion transporter defective in EHBR. Am J Physiol Gastrointest Liver Physiol 272:G16–G22. https://doi.org/10.1152/ajpgi.1997.272.1.g16
Jackson JS, Kennedy CJ (2017) Regulation of hepatic abcb4 and cyp3a65 gene expression and multixenobiotic/multidrug functional activity in the model teleost, Danio rerio (zebrafish). Comp Biochem Physiol C 200:34–41
Johansen JA, Geen GH (1990) Sublethal and acute toxicity of the ethylene glycol butyl ether ester formulation of triclopyr to juvenile coho salmon (Oncorhynchus kisutch). Arch Environ Contam Toxicol 19:610–616. https://doi.org/10.1007/BF01059083
Jones PM, George AM (2004) The ABC transporter structure and mechanism: perspectives on recent research. Cell Mol Life Sci 61:682–699. https://doi.org/10.1007/s00018-003-3336-9
Kim RB (2002) Drugs as P-glycoprotein substrates, inhibitors, and inducers. Drug Metab Rev 34:47–54. https://doi.org/10.1081/DMR-120001389
Kitchell JF (1983) Energetics. In: Fish Biomechanics. Praeger, New York, pp 312–338
Kliewer SA, Goodwin B, Willson TM (2002) The nuclear pregnane X receptor: a key regulator of xenobiotic metabolism. Endocr Rev 23:687–702. https://doi.org/10.1210/er.2001-0038
Krisfalusi M, Eroschenko VP, Cloud JG (1998) Exposure of juvenile rainbow trout (Oncorhynchus mykiss) to methoxychlor results in a dose-dependent decrease in growth and survival but does not alter male sexual differentiation. Bull Environ Contam Toxicol 60:659–666. https://doi.org/10.1007/s001289900677
Krumschnabel G, Gstir R, Manzl C, Prem C, Pafundo D, Schwarzbaum PJ (2003) Metabolic and ionic responses of trout hepatocytes to anisosmotic exposure. J Exp Biol 206:1799–1808. https://doi.org/10.1242/jeb.00370
Kurelec B (1992) The multixenobiotic resistance mechanism in aquatic organisms. Crit Rev Toxicol 22:23–43. https://doi.org/10.3109/10408449209145320
Kurelec B, Krča S, Pivčevic B, Ugarković D, Bachmann M, Imsiecke G, Müller WEG (1992) Expression of P-glycoprotein gene in marine sponges. Identification and characterization of the 125 kda drug-binding glycoprotein. Carcinogenesis 13:69–76. https://doi.org/10.1093/carcin/13.1.69
Lage H (2003) ABC-transporters: implications on drug resistance from microorganisms to human cancers. Int J Antimicrob Agents 22:188–199. https://doi.org/10.1016/S0924-8579(03)00203-6
Lin JH, Chiba M, Chen IW, Nishime JA, Deluna FA, Yamazaki M, Lin YJ (1999) Effect of dexamethasone on the intestinal first-pass metabolism of indinavir in rats: evidence of cytochrome P-450 a and P-glycoprotein induction. Drug Metab Dispos 27:1187–1193
Lončar J, Popović M, Zaja R, Smital T (2010) Gene expression analysis of the ABC efflux transporters in rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol C Toxicol Pharmacol 151:209–215. https://doi.org/10.1016/j.cbpc.2009.10.009
Mathieu MC, Lapierre I, Brault K, Raymond M (2001) Aromatic hydrocarbon receptor (AhR)·AhR nuclear translocator- and p53-mediated induction of the murine multidrug resistance mdr1 gene by 3-methylcholanthrene and benzo(a)pyrene in hepatoma cells. J Biol Chem 276:4819–4827. https://doi.org/10.1074/jbc.M008495200
Moore MN, Livingstone DR, Widdows J, Lowe DM, Pipe RK (1987) Molecular, cellular and physiological effects of oil-derived hydrocarbons on molluscs and their use in impact assessment. Philos Trans R Soc Lond B 316:603–623. https://doi.org/10.1098/rstb.1987.0041
Moore LB, Maglich JM, McKee DD, Wisely B, Willson TM, Kliewer SA, Lambert MH, Moore JT (2002) Pregnane X receptor (PXR), constitutive androstane receptor (CAR), and benzoate X receptor (BXR) define three pharmacologically distinct classes of nuclear receptors. Mol Endocrinol 16:977–986. https://doi.org/10.1210/mend.16.5.0828
Moseley RH, Jarose SM, Permoad P (1992) Organic cation transport by rat liver plasma membrane vesicles: studies with tetraethylammonium. Am J Physiol Gastrointest Liver Physiol 263:G775–G785. https://doi.org/10.1152/ajpgi.1992.263.5.g775
Murphy CA, Nisbet RM, Antczak P, Garcia-Reyero N, Gergs A, Lika K, Mathews T, Muller EB, Nacci D, Peace A, Remien CH, Schultz IR, Stevenson LM, Watanabe KH (2018) Incorporating suborganismal processes into dynamic energy budget models for ecological risk assessment. Integr Environ Assess Manag 14:615–624. https://doi.org/10.1002/ieam.4063
Okey AB, Riddick DS, Harper PA (1994) Molecular biology of the aromatic hydrocarbon (dioxin) receptor. Trends Pharmacol Sci 15:226–232. https://doi.org/10.1016/0165-6147(94)90316-6
Ortiz-Delgado JB, Segner H, Sarasquete C (2005) Cellular distribution and induction of CYP1A following exposure of gilthead seabream, Sparus aurata, to waterborne and dietary benzo(a)pyrene and 2,3,7,8-tetrachlorodibenzo-p-dioxin: an immunohistochemical approach. Aquat Toxicol 75:144–161. https://doi.org/10.1016/j.aquatox.2005.07.010
Ortiz-Delgado JB, Behrens A, Segner H, Sarasquete C (2008) Tissue-specific induction of EROD activity and CYP1A protein in Sparus aurata exposed to B(a)P and TCDD. Ecotoxicol Environ Saf 69:80–88. https://doi.org/10.1016/j.ecoenv.2006.12.021
Paetzold CS, Ross NW, Richards RC, Jones M, Hellou J, Bard SM (2009) Up-regulation of hepatic ABCC2, ABCG2, CYP1A1 and GST in multixenobiotic-resistant killifish (Fundulus heteroclitus) from the Sydney Tar Ponds, Nova Scotia, Canada. Mar Environ Res 68:37–47. https://doi.org/10.1016/j.marenvres.2009.04.002
Pesonen M, Andersson T (1992) Toxic effects of bleached and unbleached paper mill effluents in primary cultures of rainbow trout hepatocytes. Ecotoxicol Environ Saf 24:63–71. https://doi.org/10.1016/0147-6513(92)90036-3
Renfro JL, Brown MA, Parker SL, Hightower LE (1993) Relationship of thermal and chemical tolerance to transepithelial transport by cultured flounder renal epithelium. J Pharmacol Exp Ther 265:992–1000
Rice RC (1990) Environmental degradation, pollution, and the exploitation of Indonesia’s fishery resources. Indones Resour Ecol Environ:154–176
Rowe CL (1998) Elevated standard metabolic rate in a freshwater shrimp (Palaemonetes paludosus) exposed to trace element-rich coal combustion waste. Comp Biochem Physiol - A Mol Integr Physiol 121:299–304. https://doi.org/10.1016/S1095-6433(98)10141-1
Rowe CL, Kinney OM, Nagle RD, Congdon JD (1998) Elevated maintenance costs in an anuran (Rana catesbeiana) exposed to a mixture of trace elements during the embryonic and early larval periods. Physiol Zool 71:27–35. https://doi.org/10.1086/515885
Santoni-Rugiu E, Silverman JA (1997) Functional characterization of the rat mdr1b encoded P-glycoprotein: not all inducing agents are substrates. Carcinogenesis 18:2255–2263. https://doi.org/10.1093/carcin/18.11.2255
Schuetz EG, Schuetz JD, Thompson MT, Fisher RA, Madariage JR, Strom SC (1995) Phenotypic variability in induction of p-glycoprotein mrna by aromatic hydrocarbons in primary human hepatocytes. Mol Carcinog 12:61–65. https://doi.org/10.1002/mc.2940120202
Shapiro AB, Ling V (1998) The mechanism of ATP-dependent multidrug transport by P-glycoprotein, in: Acta Physiologica Scandinavica, Supplement. pp. 227–234
Sharom FJ, Yu X, Doige CA (1993) Functional reconstitution of drug transport and ATPase activity in proteoliposomes containing partially purified P-glycoprotein. J Biol Chem 268:24197–24202
Sibly RM, Calow P (1989) A life-cycle theory of responses to stress. Biol J Linn Soc 37:101–116. https://doi.org/10.1111/j.1095-8312.1989.tb02007.x
Smital T, Luckenbach T, Sauerborn R, Hamdoun AM, Vega RL, Epel D (2004) Emerging contaminants-pesticides, PPCPs, microbial degradation products and natural substances as inhibitors of multixenobiotic defense in aquatic organisms. Mutat Res Fundam Mol Mech Mutagen 552:101–117. https://doi.org/10.1016/j.mrfmmm.2004.06.006
Stegeman JJ, Hahn ME (1994) Biochemistry and molecular biology of monooxygenases: current perspectives on forms, functions, and regulation of cytochrome P450 in aquatic species. In: Aquatic Toxicology: Molecular, Biochemical, and Cellular Perspectives, pp 87–206. https://doi.org/10.1201/9781351069878
Sturm A, Segner H (2005) P-glycoproteins and xenobiotic efflux transport in fish. Biochem Mol Biol Fishes 6:495–533. https://doi.org/10.1016/S1873-0140(05)80021-9
Sturm A, Cravedi JP, Perdu E, Baradat M, Segner H (2001) Effects of prochloraz and nonylphenol diethoxylate on hepatic biotransformation enzymes in trout: a comparative in vitro/in vivo-assessment using cultured hepatocytes. Aquat Toxicol 53:229–245. https://doi.org/10.1016/S0166-445X(01)00168-0
Taniguchi K, Wada M, Kohno K, Nakamura T, Kawabe T, Kawakami M, Kagotani K, Okumura K, Akiyama SI, Kuwano M (1996) A human canalicular multispecific organic anion transporter (cMOAT) gene is overexpressed in cisplatin-resistant human cancer cell lines with decreased drug accumulation. Cancer Res 56:4124–4129
Warren CE (1971) Biology and water pollution control
Weber DN, Spieler RE (2018) Behavioral mechanisms of metal toxicity in fishes. In: Aquatic Toxicology: Molecular, Biochemical, and Cellular Perspectives, pp 421–467. https://doi.org/10.1201/9781351069878
Whitehead A, Clark BW, Reid NM, Hahn ME, Nacci D (2017) When evolution is the solution to pollution: key principles, and lessons from rapid repeated adaptation of killifish (Fundulus heteroclitus) populations. Evol Appl 10:762–783. https://doi.org/10.1111/eva.12470
Whitlock JP, Okino ST, Dong L, Ko HP, Clarke-Katzenberg R, Ma Q, Li H (1996) Induction of cytochrome P4501A1: a model for analyzing mammalian gene transcription. FASEB J 10:809–818. https://doi.org/10.1096/fasebj.10.8.8666157
Widdows J, Donkin P (1991) Role of physiological energetics in ecotoxicology. Comp Biochem Physiol C Comp. https://doi.org/10.1016/0742-8413(91)90125-D
Wieser W, Medgyesy N (1990) Aerobic maximum for growth in the larvae and juveniles of a cyprinid fish, Rutilus rutilus (L.): implications for energy budgeting in small poikilotherms. Funct Ecol 4:233. https://doi.org/10.2307/2389342
Zaja R, Munić V, Klobučar RS, Ambriović-Ristov A, Smital T (2008) Cloning and molecular characterization of apical efflux transporters (ABCB1, ABCB11 and ABCC2) in rainbow trout (Oncorhynchus mykiss) hepatocytes. Aquat Toxicol 90:322–332. https://doi.org/10.1016/j.aquatox.2008.09.012
Zucchi S, Corsi I, Luckenbach T, Bard SM, Regoli F, Focardi S (2010) Identification of five partial ABC genes in the liver of the Antarctic fish Trematomus bernacchii and sensitivity of ABCB1 and ABCC2 to Cd exposure. Environ Pollut 158:2746–2756. https://doi.org/10.1016/j.envpol.2010.04.012
Acknowledgments
We would like to thank Bruce Leighton for his assistance throughout the study.
Funding
This research was supported by a National Science and Engineering Research Council Discovery grant to CJK (#R611305).
Author information
Authors and Affiliations
Contributions
This is a solely authored manuscript. All aspects of this work including experiments, analysis and data interpretation, and manuscript writing were done by C. Kennedy.
Corresponding author
Ethics declarations
This research on animals was approved by the Simon Fraser University Animal Care Committee.
Competing interests
The author declares that he has no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kennedy, C.J. P-glycoprotein induction and its energetic costs in rainbow trout (Oncorhynchus mykiss). Fish Physiol Biochem 47, 265–279 (2021). https://doi.org/10.1007/s10695-020-00911-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-020-00911-8