Abstract
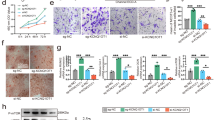
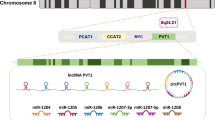
Long noncoding RNAs (lncRNAs) have been revealed to be related to multiple physiological and pathology processes such as development, carcinogenesis, and osteogenesis. It is reported that lncRNAs might exert function in osteoblast differentiation and bone formation. Here, we determined this study to clarify whether lncRNA CCAT1 could regulate osteoblast proliferation and differentiation in ovariectomized rats with osteoporosis. The osteoporosis models were established by bilateral ovariectomy and treated with CCAT1 siRNAs to discuss the effect of CCAT1 on pathological changes and osteocyte apoptosis in ovariectomized rats with osteoporosis. The osteoblasts from ovariectomized rats were cultured in vitro, which were then treated with CCAT1 siRNAs to explore the role of CCAT1 in osteoblast proliferation and differentiation. Moreover, the relationships among CCAT1, miR-34a-5p, and SMURF2 were confirmed. CCAT1 and SMURF2 were amplified while miR-34a-5p expression was inhibited in bone tissues and osteoblasts of ovariectomized rats with osteoporosis. Inhibited CCAT1 improved pathology and restricted osteocyte apoptosis of bone tissues in ovariectomized rats with osteoporosis in vivo, and also enhanced differentiation, mineralization abilities, and proliferation, and suppressed apoptosis of osteoblasts from ovariectomized rats in vitro through upregulating miR-34a-5p expression. LncRNA CCAT1 could competitively bind with miR-34a-5p to prevent the degradation of its target gene SMURF2. Results of this research suggested that the CCAT1 inhibits the proliferation and differentiation of osteoblasts in rats with osteoporosis by binding to miR-34a-5p, providing novel biomarkers for osteoporosis treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Caires ELP, Bezerra MC, Junqueira A, Fontenele SMA, Andrade SCA, d’Alva CB. Treatment of postmenopausal osteoporosis: a literature-based algorithm for use in the public health care system. Rev Bras Reumatol Engl Ed. 2017;57:254–63.
Kulenovic I, Rasic S, Kulenovic E. Osteoporosis: current trends in diagnosis and management. Bosn J Basic Med Sci. 2006;6:24–8.
Yeap SS, Hew FL, Damodaran P, Chee W, Lee JK, Goh EML, et al. A summary of the Malaysian Clinical Guidance on the management of postmenopausal and male osteoporosis, 2015. Osteoporos Sarcopenia. 2016;2:1–12.
Bultink IE, Lems WF. Osteoarthritis and osteoporosis: what is the overlap? Curr Rheumatol Rep. 2013;15:328.
Wang T, Yang L, Jiang J, Liu Y, Fan Z, Zhong C, et al. Pulsed electromagnetic fields: promising treatment for osteoporosis. Osteoporos Int. 2019;30:267–76.
Su P, Mu S, Wang Z. Long noncoding RNA SNHG16 promotes osteosarcoma cells migration and invasion via sponging miRNA-340. DNA Cell Biol. 2019;38:170–5.
Cai N, Li C, Wang F. Silencing of LncRNA-ANCR promotes the osteogenesis of osteoblast cells in postmenopausal osteoporosis via targeting EZH2 and RUNX2. Yonsei Med J. 2019;60:751–9.
Wang Q, Li Y, Zhang Y, Ma L, Lin L, Meng J, et al. LncRNA MEG3 inhibited osteogenic differentiation of bone marrow mesenchymal stem cells from postmenopausal osteoporosis by targeting miR-133a-3p. Biomed Pharmacother. 2017;89:1178–86.
Zhao J, Cheng L. Long non-coding RNA CCAT1/miR-148a axis promotes osteosarcoma proliferation and migration through regulating PIK3IP1. Acta Biochim Biophys Sin (Shanghai). 2017;49:503–12.
Yu J, Jiang L, Gao Y, Sun Q, Liu B, Hu Y, et al. LncRNA CCAT1 negatively regulates miR-181a-5p to promote endometrial carcinoma cell proliferation and migration. Exp Ther Med. 2019;17:4259–66.
Wang G, Zhao F, Yang D, Wang J, Qiu L, Pang X. Human amniotic epithelial cells regulate osteoblast differentiation through the secretion of TGFbeta1 and microRNA-34a-5p. Int J Mol Med. 2018;41:791–9.
Kang H, Chen H, Huang P, Qi J, Qian N, Deng L, et al. Glucocorticoids impair bone formation of bone marrow stromal stem cells by reciprocally regulating microRNA-34a-5p. Osteoporos Int. 2016;27:1493–505.
Krzeszinski JY, Wei W, Huynh H, Jin Z, Wang X, Chang TC, et al. miR-34a blocks osteoporosis and bone metastasis by inhibiting osteoclastogenesis and Tgif2. Nature. 2014;512:431–5.
Rokavec M, Li H, Jiang L, Hermeking H. The p53/miR-34 axis in development and disease. J Mol Cell Biol. 2014;6:214–30.
Klupp F, Giese C, Halama N, Franz C, Lasitschka F, Warth A, et al. E3 ubiquitin ligase Smurf2: a prognostic factor in microsatellite stable colorectal cancer. Cancer Manag Res. 2019;11:1795–803.
Qu Y, Zhang Q, Cai X, Li F, Ma Z, Xu M, et al. Exosomes derived from miR-181-5p-modified adipose-derived mesenchymal stem cells prevent liver fibrosis via autophagy activation. J Cell Mol Med. 2017;21:2491–502.
Huang W, Zeng C, Hu S, Wang L, Liu J. ATG3, a target of miR-431-5p, promotes proliferation and invasion of colon cancer via promoting autophagy. Cancer Manag Res. 2019;11:10275–85.
Li G, Liu H, Zhang X, Liu X, Zhang G, Liu Q. The protective effects of microRNA-26a in steroid-induced osteonecrosis of the femoral head by repressing EZH2. Cell Cycle. 2020;19:551–66.
Ayuk SM, Abrahamse H, Houreld NN. The role of photobiomodulation on gene expression of cell adhesion molecules in diabetic wounded fibroblasts in vitro. J Photochem Photobio B. 2016;161:368–74.
Cai X, Yi X, Zhang Y, Zhang D, Zhi L, Liu H. Genetic susceptibility of postmenopausal osteoporosis on sulfide quinone reductase-like gene. Osteoporos Int. 2018;29:2041–7.
Wang X, Lan Z, He J, Lai Q, Yao X, Li Q, et al. LncRNA SNHG6 promotes chemoresistance through ULK1-induced autophagy by sponging miR-26a-5p in colorectal cancer cells. Cancer Cell Int. 2019;19:234.
Han C, Li X, Fan Q, Liu G, Yin J. CCAT1 promotes triple-negative breast cancer progression by suppressing miR-218/ZFX signaling. Aging (Albany NY). 2019;11:4858–75.
Li Y, Zhu G, Ma Y and Qu H. lncRNA CCAT1 contributes to the growth and invasion of gastric cancer via targeting miR-219-1. J Cell Biochem. 2019, 120:19457–68.
Ji S, Wang S, Zhao X, Lv L. Long noncoding RNA NEAT1 regulates the development of osteosarcoma through sponging miR-34a-5p to mediate HOXA13 expression as a competitive endogenous RNA. Mol Genet Genom Med. 2019;7:e673.
Li X, Diao Z, Ding J, Liu R, Wang L, Huang W, et al. The downregulation of SnoN expression in human renal proximal tubule epithelial cells under high-glucose conditions is mediated by an increase in Smurf2 expression through TGF-beta1 signaling. Int J Mol Med. 2016;37:415–22.
Komori T. Glucocorticoid signaling and bone biology. Horm Metab Res. 2016;48:755–63.
Lin TH, Gibon E, Loi F, Pajarinen J, Cordova LA, Nabeshima A, et al. Decreased osteogenesis in mesenchymal stem cells derived from the aged mouse is associated with enhanced NF-kappaB activity. J Orthop Res. 2017;35:281–8.
Xie K, Liu L, Chen J, Liu F. Exosomes derived from human umbilical cord blood mesenchymal stem cells improve hepatic ischemia reperfusion injury via delivering miR-1246. Cell Cycle. 2019;18:3491–501.
Liu Y, Zhao C, Ma Q, Li Y. Sinomenine retards LPS-elicited inflammation via down-regulating CCAT1 in HaCaT cells. Life Sci. 2019;233:116703.
Ramkumar C, Kong Y, Trabucco SE, Gerstein RM, Zhang H. Smurf2 regulates hematopoietic stem cell self-renewal and aging. Aging Cell. 2014;13:478–86.
Kim JH, Ham S, Lee Y, Suh GY, Lee YS. TTC3 contributes to TGF-beta1-induced epithelial-mesenchymal transition and myofibroblast differentiation, potentially through SMURF2 ubiquitylation and degradation. Cell Death Dis. 2019;10:92.
Acknowledgements
We would like to acknowledge the reviewers for their helpful comments on this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics statement
Animal experiments were strictly in accordance with the Guide to the Management and Use of Laboratory Animals issued by the National Institutes of Health. The protocol of animal experiments was approved by the Institutional Animal Care and Use Committee of the First People’s Hospital of Yunnan Province.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, F., Jiang, C., Bu, G. et al. Silencing long noncoding RNA colon cancer-associated transcript-1 upregulates microRNA-34a-5p to promote proliferation and differentiation of osteoblasts in osteoporosis. Cancer Gene Ther 28, 1150–1161 (2021). https://doi.org/10.1038/s41417-020-00264-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-020-00264-7
This article is cited by
-
Long non-coding RNAs in osteoporosis: from mechanisms of action to therapeutic potential
Human Cell (2023)
-
Comprehensive analysis of lncRNA-miRNA-mRNA networks during osteogenic differentiation of bone marrow mesenchymal stem cells
BMC Genomics (2022)
-
The management of bone defect using long non-coding RNA as a potential biomarker for regulating the osteogenic differentiation process
Molecular Biology Reports (2022)