Abstract
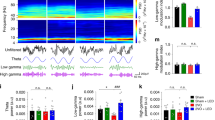
Theta and gamma rhythms in hippocampus are important to cognitive performance. The cognitive impairments following cerebral ischemia is linked with the dysfunction of theta and gamma oscillations. As the primary mechanism for learning and memory, synaptic plasticity is in connection with these neural oscillations. Although vascular endothelial growth factor (VEGF) is thought to protect synaptic function in the ischemia rats to relieve cognitive impairment, little has been done on its effect of neural dynamics with this process. The present study investigated whether the alternation of neural oscillations in the hippocampus of ischemia rats is one of the potential neuroprotective mechanisms of VEGF. Rats were treated with the intranasal administration of VEGF at 72 h following chronic global cerebral ischemia procedure. Then local field potentials (LFPs) in hippocampal CA1 and CA3 regions were recorded and analyzed. Our results showed that VEGF can improve the power of theta and gamma rhythms in CA1 region after ischemia. Chronic global cerebral ischemia reduced the theta–gamma phase-amplitude coupling (PAC) not only within CA1 area but also in the pathway from CA3 to CA1, while VEGF alleviated the decreased coupling strength. Despite these notable differences, there were no obvious changes in the PAC within CA3 region. Surprisingly, the ischemia state did not affect the phase–phase interaction of hippocampus. In conclusion, our findings demonstrated that VEGF enhanced the theta–gamma PAC strength of CA3–CA1 pathway in ischemia rats, which may futher improve the information transmission within the hippocampus. These results illustrated the potential electrophysiologic mechanism of VEGF on cognitive improvement.





Similar content being viewed by others
References
Amemiya, S., & Redish, A. D. (2018). Hippocampal theta-gamma coupling reflects state-dependent information processing in decision making. Cell Reports, 22(12), 3328–3338. https://doi.org/10.1016/j.celrep.2018.02.091.
Bandarabadi, M., Boyce, R., Gutierrez Herrera, C., Bassetti, C. L., Williams, S., Schindler, K., et al. (2019). Dynamic modulation of theta-gamma coupling during rapid eye movement sleep. Sleep. https://doi.org/10.1093/sleep/zsz182.
Barry, J. M., Sakkaki, S., Barriere, S. J., Patterson, K. P., Lenck-Santini, P. P., Scott, R. C., et al. (2016). Temporal coordination of hippocampal neurons reflects cognitive outcome post-febrile status epilepticus. EBioMedicine, 7, 175–190. https://doi.org/10.1016/j.ebiom.2016.03.039.
Baskys, A., & Hou, A. C. (2007). Vascular dementia: Pharmacological treatment approaches and perspectives. Clinical Interventions in Aging, 2(3), 327–335.
Battaglia, F. P., Benchenane, K., Sirota, A., Pennartz, C. M., & Wiener, S. I. (2011). The hippocampus: Hub of brain network communication for memory. Trends in Cognitive Sciences, 15(7), 310–318. https://doi.org/10.1016/j.tics.2011.05.008.
Belluscio, M. A., Mizuseki, K., Schmidt, R., Kempter, R., & Buzsaki, G. (2012). Cross-frequency phase-phase coupling between theta and gamma oscillations in the hippocampus. Journal of Neuroscience, 32(2), 423–435. https://doi.org/10.1523/jneurosci.4122-11.2012.
Buzsaki, G. (2002). Theta oscillations in the hippocampus. Neuron, 33(3), 325–340. https://doi.org/10.1016/s0896-6273(02)00586-x.
Canolty, R. T., Edwards, E., Dalal, S. S., Soltani, M., Nagarajan, S. S., Kirsch, H. E., et al. (2006). High gamma power is phase-locked to theta oscillations in human neocortex. Science, 313(5793), 1626–1628. https://doi.org/10.1126/science.1128115.
Colgin, L. L. (2016). Rhythms of the hippocampal network. Nature Reviews Neuroscience, 17(4), 239–249. https://doi.org/10.1038/nrn.2016.21.
Crepel, V., Epsztein, J., & Ben-Ari, Y. (2003). Ischemia induces short- and long-term remodeling of synaptic activity in the hippocampus. Journal of Cellular and Molecular Medicine, 7(4), 401–407. https://doi.org/10.1111/j.1582-4934.2003.tb00242.x.
Echeverria, V., Barreto, G. E., Avila-Rodriguezc, M., Tarasov, V. V., & Aliev, G. (2017). Is VEGF a key target of cotinine and other potential therapies against Alzheimer disease? Current Alzheimer Research, 14(11), 1155–1163. https://doi.org/10.2174/1567205014666170329113007.
Ehrlichman, R. S., Gandal, M. J., Maxwell, C. R., Lazarewicz, M. T., Finkel, L. H., Contreras, D., et al. (2009). N-methyl-d-aspartic acid receptor antagonist-induced frequency oscillations in mice recreate pattern of electrophysiological deficits in schizophrenia. Neuroscience, 158(2), 705–712. https://doi.org/10.1016/j.neuroscience.2008.10.031.
Fardo, F., Vinding, M. C., Allen, M., Jensen, T. S., & Finnerup, N. B. (2017). Delta and gamma oscillations in operculo-insular cortex underlie innocuous cold thermosensation. Journal of Neurophysiology, 117(5), 1959–1968. https://doi.org/10.1152/jn.00843.2016.
Guo, S., Xu, J. J., Wei, N., Han, J. Y., Xue, R., Xu, P. S., et al. (2019). Honokiol attenuates the memory impairments, oxidative stress, neuroinflammation, and GSK-3beta activation in vascular dementia rats. Journal of Alzheimers Disease. https://doi.org/10.3233/JAD-190324.
Han, X. J., Shi, Z. S., Xia, L. X., Zhu, L. H., Zeng, L., Nie, J. H., et al. (2016). Changes in synaptic plasticity and expression of glutamate receptor subunits in the CA1 and CA3 areas of the hippocampus after transient global ischemia. Neuroscience, 327, 64–78. https://doi.org/10.1016/j.neuroscience.2016.04.011.
Hasselmo, M. E. (2005). What is the function of hippocampal theta rhythm?–Linking behavioral data to phasic properties of field potential and unit recording data. Hippocampus, 15(7), 936–949. https://doi.org/10.1002/hipo.20116.
He, J. W., Rabiller, G., Nishijima, Y., Akamatsu, Y., Khateeb, K., Yazdan-Shahmorad, A., et al. (2019). Experimental cortical stroke induces aberrant increase of sharp-wave-associated ripples in the hippocampus and disrupts cortico-hippocampal communication. Journal of Cerebral Blood Flow and Metabolism. https://doi.org/10.1177/0271678X19877889.
Hudson, M. R., Sokolenko, E., O’Brien, T. J., & Jones, N. C. (2020). NMDA receptors on parvalbumin-positive interneurons and pyramidal neurons both contribute to MK-801 induced gamma oscillatory disturbances: Complex relationships with behaviour. Neurobiology of Disease, 134, 104625. https://doi.org/10.1016/j.nbd.2019.104625.
Hutchison, I. C., & Rathore, S. (2015). The role of REM sleep theta activity in emotional memory. Frontiers in Psychology, 6, 1439. https://doi.org/10.3389/fpsyg.2015.01439.
Lara, G. A., Alekseichuk, I., Turi, Z., Lehr, A., Antal, A., & Paulus, W. (2018). Perturbation of theta-gamma coupling at the temporal lobe hinders verbal declarative memory. Brain Stimulation, 11(3), 509–517. https://doi.org/10.1016/j.brs.2017.12.007.
Latzer, P., Shchyglo, O., Hartl, T., Matschke, V., Schlegel, U., Manahan-Vaughan, D., et al. (2019). Blocking VEGF by bevacizumab compromises electrophysiological and morphological properties of hippocampal neurons. Frontiers in Cellular Neuroscience, 13, 113. https://doi.org/10.3389/fncel.2019.00113.
Levesque, M., Langlois, J. M., Lema, P., Courtemanche, R., Bilodeau, G. A., & Carmant, L. (2009). Synchronized gamma oscillations (30–50 Hz) in the amygdalo-hippocampal network in relation with seizure propagation and severity. Neurobiology of Disease, 35(2), 209–218. https://doi.org/10.1016/j.nbd.2009.04.011.
Li, W., Wei, D., Liang, J., Xie, X., Song, K., & Huang, L. (2019a). Comprehensive evaluation of white matter damage and neuron death and whole-transcriptome analysis of rats with chronic cerebral hypoperfusion. Frontiers in Cellular Neuroscience, 13, 310. https://doi.org/10.3389/fncel.2019.00310.
Li, W., Wei, D., Lin, J., Liang, J., Xie, X., Song, K., et al. (2019b). Dl-3-n-butylphthalide reduces cognitive impairment induced by chronic cerebral hypoperfusion through GDNF/GFRalpha1/Ret signaling preventing hippocampal neuron apoptosis. Frontiers in Cellular Neuroscience, 13, 351. https://doi.org/10.3389/fncel.2019.00351.
Lisman, J. E., & Jensen, O. (2013). The theta-gamma neural code. Neuron, 77(6), 1002–1016. https://doi.org/10.1016/j.neuron.2013.03.007.
Luck, R., Urban, S., Karakatsani, A., Harde, E., Sambandan, S., Nicholson, L., et al. (2019). VEGF/VEGFR2 signaling regulates hippocampal axon branching during development. eLife. https://doi.org/10.7554/eLife.49818.
Manoonkitiwongsa, P. S., Schultz, R. L., McCreery, D. B., Whitter, E. F., & Lyden, P. D. (2004). Neuroprotection of ischemic brain by vascular endothelial growth factor is critically dependent on proper dosage and may be compromised by angiogenesis. Journal of Cerebral Blood Flow and MetabolisM, 24(6), 693–702. https://doi.org/10.1097/01.WCB.0000126236.54306.21.
Pinault, D. (2008). N-methyl d-aspartate receptor antagonists ketamine and MK-801 induce wake-related aberrant gamma oscillations in the rat neocortex. Biological Psychiatry, 63(8), 730–735. https://doi.org/10.1016/j.biopsych.2007.10.006.
Scheepers, L., Jacobsson, L. T. H., Kern, S., Johansson, L., Dehlin, M., & Skoog, I. (2019). Urate and risk of Alzheimer’s disease and vascular dementia: A population-based study. Alzheimers Dement, 15(6), 754–763. https://doi.org/10.1016/j.jalz.2019.01.014.
Scheffer-Teixeira, R., & Tort, A. B. (2016). On cross-frequency phase-phase coupling between theta and gamma oscillations in the hippocampus. eLife. https://doi.org/10.7554/eLife.20515.
Schulz, S. B., Klaft, Z. J., Rosler, A. R., Heinemann, U., & Gerevich, Z. (2012). Purinergic P2X, P2Y and adenosine receptors differentially modulate hippocampal gamma oscillations. Neuropharmacology, 62(2), 914–924. https://doi.org/10.1016/j.neuropharm.2011.09.024.
Shingo, T., Date, I., Yano, A., Kobayashi, K., & Ohmoto, T. J. I. C. (2003). Cell therapy for cerebral ischemia using vascular endothelial growth factor-secreting cells. International Congress Series, 1252(03), 483–487.
Sokolenko, E., Hudson, M. R., Nithianantharajah, J., & Jones, N. C. (2019). The mGluR2/3 agonist LY379268 reverses NMDA receptor antagonist effects on cortical gamma oscillations and phase coherence, but not working memory impairments, in mice. Journal of Psychopharmacology, 33(12), 1588–1599. https://doi.org/10.1177/0269881119875976.
Trimper, J. B., Stefanescu, R. A., & Manns, J. R. (2014). Recognition memory and theta-gamma interactions in the hippocampus. Hippocampus, 24(3), 341–353. https://doi.org/10.1002/hipo.22228.
Wang, L., Wang, F., Liu, S., Yang, X., Yang, J., & Ming, D. (2018). VEGF attenuates 2-VO induced cognitive impairment and neuronal injury associated with the activation of PI3K/Akt and Notch1 pathway. Experimental Gerontology, 102, 93–100. https://doi.org/10.1016/j.exger.2017.12.010.
Wang, L., Wang, J., Wang, F., Liu, C., Yang, X., Yang, J., et al. (2017). VEGF-mediated cognitive and synaptic improvement in chronic cerebral hypoperfusion rats involves autophagy process. Neuromolecular Medicine, 19(2–3), 423–435. https://doi.org/10.1007/s12017-017-8458-6.
Wang, Y., Galvan, V., Gorostiza, O., Ataie, M., Jin, K., & Greenberg, D. A. (2006). Vascular endothelial growth factor improves recovery of sensorimotor and cognitive deficits after focal cerebral ischemia in the rat. Brain Research, 1115(1), 186–193. https://doi.org/10.1016/j.brainres.2006.07.060.
Xu, X., Li, Z., Yang, Z., & Zhang, T. (2012). Decrease of synaptic plasticity associated with alteration of information flow in a rat model of vascular dementia. Neuroscience, 206, 136–143. https://doi.org/10.1016/j.neuroscience.2011.12.050.
Xu, X., Liu, C., Li, Z., & Zhang, T. (2015). Effects of hydrogen sulfide on modulation of theta-gamma coupling in hippocampus in vascular dementia rats. Brain Topography, 28(6), 879–894. https://doi.org/10.1007/s10548-015-0430-x.
Xu, X., Zheng, C., & Zhang, T. (2013). Reduction in LFP cross-frequency coupling between theta and gamma rhythms associated with impaired STP and LTP in a rat model of brain ischemia. Frontiers in Computational Neuroscience, 7, 27. https://doi.org/10.3389/fncom.2013.00027.
Yang, J., Yao, Y., Chen, T., & Zhang, T. (2014). VEGF ameliorates cognitive impairment in in vivo and in vitro ischemia via improving neuronal viability and function. Neuromolecular Medicine, 16(2), 376–388. https://doi.org/10.1007/s12017-013-8284-4.
Zheng, C., Bieri, K. W., Hsiao, Y. T., & Colgin, L. L. (2016). Spatial sequence coding differs during slow and fast gamma rhythms in the hippocampus. Neuron, 89(2), 398–408. https://doi.org/10.1016/j.neuron.2015.12.005.
Zheng, C., & Zhang, T. (2015). Synaptic plasticity-related neural oscillations on hippocampus-prefrontal cortex pathway in depression. Neuroscience, 292, 170–180. https://doi.org/10.1016/j.neuroscience.2015.01.071.
Zheng, L., Yu, M., Lin, R., Wang, Y., Zhuo, Z., Cheng, N., et al. (2020). Rhythmic light flicker rescues hippocampal low gamma and protects ischemic neurons by enhancing presynaptic plasticity. Nature Communications, 11(1), 3012. https://doi.org/10.1038/s41467-020-16826-0.
Acknowledgements
The study was funded by grants from the National Natural Science Foundation of China (81871517, 31800889, 81870847), the National Key R&D Program of China (2017YFB1300302), and the Doctoral Fund of Ministry of Education of China (2020M670645). The authors are grateful to Professor Zhuo Yang and Tao Zhang from Nankai University for providing the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors acknowledged that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, L., Yang, J., Xiao, X. et al. VEGF Modulates the Neural Dynamics of Hippocampal Subregions in Chronic Global Cerebral Ischemia Rats. Neuromol Med 23, 416–427 (2021). https://doi.org/10.1007/s12017-020-08642-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-020-08642-y