Abstract
Purpose
Antibiotics often coexist with heavy metals in contaminated soils, making their behaviors more complicated than their individual presences. Sorption to soil components deeply affects the mobility, bioavailability, and fate of contaminants. However, limited studies have investigated the cosorption characteristics of tetracycline (TC) and heavy metals to typical soil minerals, particularly phyllosilicates.
Materials and methods
This study investigates the effect of Cd(II) on TC sorption by kaolinite (Kao, 1:1-type) and montmorillonite (Mont, 2:1-type) via isothermal adsorption, coupled with X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR).
Results and discussion
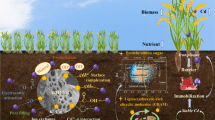
Isothermal results show that TC sorption on both kaolinite (Kao, 1:1-type) and montmorillonite (Mont, 2:1-type) is little affected by Cd(II) at pH 4.5 but increases significantly at pH 8.5. The overall effect of Cd(II) on TC sorption depends on the intensities of two positive processes, i.e., (1) the formation of soluble Cd-TC complex increases sorption compared to free TC molecules and (2) the formation of a ternary clay-Cd-TC complex at clay variable edge sites increases sorption, and one negative process, i.e., competition of Cd(II) with cationic and zwitterionic TC species at the permanently charged sites decreases sorption. XRD analyses demonstrate that TC intercalates into the interlayer (d001 = 18.7 Å) of Mont as one monolayer parallel molecule both with and without Cd(II) but adsorbs predominately on external surface of Kao. FTIR results suggest a flat-lying horizontal configuration of adsorbed TC molecules on clays, accompanied by a strong complexation between the carbonyl and amino groups of TC molecules and the surfaces. Besides, the presence of Cd(II) more or less facilitates this complexation.
Conclusions
We propose that Cd(II) can significantly promote TC adsorption on clay minerals at alkaline pH rather than acidic pH. TC and Cd(II) interact intensely and form strong surface complexation on clay surfaces. Hence, coexistence with Cd(II) may lower the mobility and thus the bioavailability of TC in alkaline soils, which provides a new direction for soil remediation by regulating soil pH using alkaline materials.
Graphical abstract





Similar content being viewed by others
References
Acosta R, Fierro V, Martinez de Yuso A, Nabarlatz D, Celzard A (2016) Tetracycline adsorption onto activated carbons produced by KOH activation of tyre pyrolysis char. Chemosphere 149:168–176
Aristilde L, Marichal C, Miéhé-Brendlé J, Lanson B, Charlet L (2010) Interactions of oxytetracycline with a smectite clay: a spectroscopic study with molecular simulations. Environ Sci Technol 44:7839–7845
Binh VN, Dang N, Anh NTK, Ky LX, Thai PK (2018) Antibiotics in the aquatic environment of Vietnam: sources, concentrations, risk and control strategy. Chemosphere 197:438–450
Caminati G, Focardi C, Gabrielli G, Gambinossi F, Mecheri B, Nocentini M, Puggelli M (2002) Spectroscopic investigation of tetracycline interaction with phospholipid Langmuir–Blodgett films. Mat Sci Eng C 22:301–305
Chang PH, Li Z, Jiang WT, Jean JS (2009a) Adsorption and intercalation of tetracycline by swelling clay minerals. Appl Clay Sci 46:27–36
Chang PH, Jean JS, Jiang WT, Li Z (2009b) Mechanism of tetracycline sorption on rectorite. Colloid Surface A 339:94–99
Chang PH, Li Z, Yu TL, Munkhbayer S, Kuo TH, Hung YC, Jean JS, Lin KH (2009c) Sorptive removal of tetracycline from water by palygorskite. J Hazard Mater 165:148–155
Chang PH, Li Z, Jean JS, Jiang WT, Wang CJ, Lin KH (2012) Adsorption of tetracycline on 2:1 layered non-swelling clay mineral illite. Appl Clay Sci 67–68:158–163
Chen CQ, Zheng L, Zhou JL, Zhao H (2017) Persistence and risk of antibiotic residues and antibiotic resistance genes in major mariculture sites in Southeast China. Sci Total Environ 580:1175–1184
Daghrir R, Drogui O (2013) Tetracycline antibiotics in the environment: a review. Environ Chem Lett 11:209–227
Du H, Huang Q, Lei M, Tie B (2018) Sorption of Pb(II) by nanosized ferrihydrite organo-mineral composites formed by adsorption versus coprecipitation. ACS Earth Space Chem 2:556–564
Du H, Qu C, Ma M, Lei M, Tie B, Liu X, Wei X, Yang Y (2019) Insights into Pb(II) binding by Fe/Al hydroxide–microbe composite: XAFS spectroscopy and isothermal titration calorimetry study. Chem Geol 510:84–90
Du H, Xu Z, Hu M, Zhang H, Peacock C, Liu X, Nie N, Xue Q, Lei M, Tie B (2020) Natural organic matter decreases uptake of W(VI), and reduces W(VI) to W(V), during adsorption to ferrihydrite. Chem Geol 540:119567
Figueroa RA, Leonard A, MacKay AA (2004) Modeling tetracycline antibiotic sorption to clays. Environ Sci Technol 38:476–483
Gambinossi F, Mecheri B, Nocentini M, Puggelli M, Caminati G (2004) Effect of the phospholipid head group in antibiotic-phospholipid association at water-air interface. Biophys Chem 110:101–117
Grafe M, Singhb B, Balasubramanian M (2007) Surface speciation of Cd(II) and Pb(II) on kaolinite by XAFS spectroscopy. J Colloid Interface Sci 315:21–32
Graouer-Bacart M, Sayen S, Guillon E (2013) Macroscopic and molecular approaches of enrofloxacin retention in soils in presence of Cu(II). J Colloid Interface Sci 408:191–199
Gu X, Evans LJ (2008) Surface complexation modelling of Cd(II), Cu(II), Ni(II), Pb(II) and Zn(II) adsorption onto kaolinite. Geochim Cosmochim Ac 72:267–276
Jackson ML (1957) Frequency distribution of clay minerals in major great soil groups as related to the factors of soil formation. Clays Clay Miner 6:133–143
Jia DA, Zhou DM, Wang YJ, Zhu HW, Chen JL (2008) Adsorption and cosorption of Cu(II) and tetracycline on two soils with different characteristics. Geoderma 146:224–230
Kleber M, Eusterhues K, Keiluweit M, Mikutta C, Mikutta R, Nico PS (2015) Mineral-organic associations: formation, properties, and relevance in soil environments. Adv Agron 130:1–140
Kong WD, Zhu YG, Fu BJ, Marschner P, He JZ (2006) The veterinary antibiotic oxytetracycline and Cu influence functional diversity of the soil microbial community. Environ Pollut 143:129–137
Levin-Reisman I, Ronin I, Gefen O, Braniss I, Shoresh N, Balaban NQ (2017) Antibiotic tolerance facilitates the evolution of resistance. Science 355:826–830
Li Z, Schulz L, Ackley C, Fenske N (2010) Adsorption of tetracycline on kaolinite with pH-dependent surface charges. J Colloid Interface Sci 351:254–260
Li X, Liu N, Tang L (2020) Zhang J (2020) Specific elevated adsorption and stability of cations in the interlayer compared with at the external surface of clay minerals. Appl Clay Sci 198:105814
Parolo ME, Savini MC, Vallés JM, Baschini MT, Avena MJ (2008) Tetracycline adsorption on montmorillonite: pH and ionic strength effects. Appl Clay Sci 40:179–186
Parolo ME, Avena MJ, Pettinari GR, Zajonkovsky I, Valles JM, Baschini MT (2010) Antimicrobial properties of tetracycline and minocycline-montmorillonites. Appl Clay Sci 49:194–199
Parolo ME, Avena MJ, Pettinari GR, Baschini MT (2012) Influence of Ca2+ on tetracycline adsorption on montmorillonite. J Colloid Interface Sci 368:420–426
Pei ZG, Shan XQ, Kong JJ, Wen B, Owens G (2010) Coadsorption of ciprofloxacin and Cu(II) on montmorillonite and kaolinite as affected by solution pH. Environ Sci Technol 44(3):915–920
Pei ZG, Shan XQ, Zhang SZ, Kong JJ, Wen B, Zhang J, Zheng LR, Xie YN, Janssens K (2011) Insight to ternary complexes of co-adsorption of norfloxacin and Cu(II) onto montmorillonite at different pH using EXAFS. J Hazard Mater 186:842–848
Porubcan LS, Serna CJ, White JL, Hem SL (1978) Mechanism of adsorption of clindamycin and tetracycline by montmorillonite. J Pharm Sci 67:1081–1087
Priya SS, Radha KV (2017) A review on the adsorption studies of tetracycline onto various types of adsorbents. Chem Eng Commun 204:821–839
Rong X, Huang Q, Chen W (2007) Microcalorimetric investigation on the metabolic activity of Bacillus thuringiensis as influenced by kaolinite, montmorillonite and goethite. Appl Clay Sci 38:97–103
Schmitt H, Stoob K, Hamscher G, Smit E, Seinen W (2006) Tetracyclines and tetracycline resistance in agricultural soils: microcosm and field studies. Microb Ecol 51:267–276
Sposito G, Prost R (1982) Structure of water adsorbed on smectites. Chem Rev 82:553–573
Sun J, Zeng Q, Tsang DCW, Zhu LZ, Li XD (2017) Antibiotics in the agricultural soils from the Yangtze River Delta, China. Chemosphere 189:301–308
Tolls J (2001) Sorption of veterinary pharmaceuticals in soils: a review. Environ Sci Technol 35:3397–3406
Vasconcelos IF, Haack EA, Maurice PA, Bunker BA (2008) EXAFS analysis of cadmium(II) adsorption to kaolinite. Chem Geol 249:237–249
Wan Y, Bao Y, Zhou Q (2010) Simultaneous adsorption and desorption of cadmium and tetracycline on cinnamon soil. Chemosphere 80:807–812
Wang YJ, Jia DA, Sun RJ, Zhu HW, Zhou DM (2008) Adsorption and cosorption of tetracycline and copper(II) on montmorillonite as affected by solution pH. Environ Sci Technol 42:3254–3259
Wu L, Pan X, Chen L, Huang Y, Teng Y, Luo Y, Christie P (2013) Occurrence and distribution of heavy metals and tetracyclines in agricultural soils after typical land use change in east China. Environ Sci Pollut Res 20:8342–8354
Wu HL, Xie HR, He GP, Guan YF, Zhang YL (2016) Effects of the pH and anions on the adsorption of tetracycline on iron-montmorillonite. Appl Clay Sci 119:161–169
Xu Y, Yu W, Ma Q, Zhou H (2015) Interactive effects of sulfadiazine and Cu(II) on their sorption and desorption on two soils with different characteristics. Chemosphere 138:701–707
Xue Q, Ran Y, Tan Y, Peacock CL, Du H (2019) Arsenite and arsenate binding to ferrihydrite organo-mineral coprecipitate: Implications for arsenic mobility and fate in natural environments. Chemosphere 224:103–110
Zhang Z, Sun K, Gao B, Zhang G, Liu X, Zhao Y (2011) Adsorption of tetracycline on soil and sediment: effects of pH and the presence of Cu(II). J Hazard Mater 190:856–862
Zhao Y, Geng J, Wang X, Gu X, Gao S (2011) Tetracycline adsorption on kaolinite: pH, metal cations and humic acid effects. Ecotoxicology 20:1141–1147
Zhao Y, Gu X, Gao S, Geng J, Wang X (2012) Adsorption of tetracycline (TC) onto montmorillonite: cations and humic acid effects. Geoderma 183–184:12–18
Zhao Y, Tan Y, Guo Y, Gu X, Wang X, Zhang Y (2013) Interactions of tetracycline with Cd(II), Cu(II) and Pb(II) and their cosorption behavior in soils. Environ Pollut 180:206–213
Zhao Y, Gu X, Li S, Han R, Wang G (2015) Insights into tetracycline adsorption onto kaolinite and montmorillonite: experiments and modeling. Environ Sci Pollut Res 22:17031–17040
Funding
We acknowledge the National Natural Science Foundation of China (41907015), Hunan Provincial Natural Science Foundation of China (2018JJ3239), and the Scientific Research Fund of Hunan Provincial Education Department, China (18B120), for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible Editor: Xilong Wang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1.
Supplemental materials contain the Freundlich and Langmuir equation description, molecular structures of TC on a planar view (Figure S1); Cd solution speciation analyses (Figure S2); standard curves for TC with or without Cd at a wavelength of 275 nm using colorimetric method (Figure S3); FTIR spectra of TC and TC-loaded montmorillonite and kaolinite in the presence and absence of Cd at different pHs (Figure S4); and the FTIR band positions (cm-1) for crystalline TC and TC adsorbed on Mont and Kao in the presence and absence of Cd (Table S1). (DOCX 997 kb)
Rights and permissions
About this article
Cite this article
Du, H., Du, J., Liu, F. et al. Binding of tetracycline on soil phyllosilicates with Cd(II) as affected by pH and mineral type. J Soils Sediments 21, 775–783 (2021). https://doi.org/10.1007/s11368-020-02867-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-020-02867-x