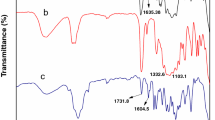
Abstract
Waterborne epoxy curing agent plays a key part in the waterborne epoxy resin coatings, directly affecting the physical and chemical properties of the coating film. Here, we present triethylene tetramine (TETA), polypropylene glycol diglycidyl ether (PPGDGE), epoxy resin E20 and phenyl glycidyl ether (PGE) as reactants to fabricate a novel nonionic self-emulsifiable waterborne epoxy curing agent for oily epoxy resin through three steps of chain extension reaction, addition reaction, and capping reaction. The curing agent achieved excellent emulsifying and curing properties under the condition that TETA reacted with PPGDGE at 65°C for 4 h with the molar ratio of 2.2:1, 40% primary amine was added with E20 at 65°C for 3 h and 125% residual primary amine was capped with PGE at 65°C for 3 h. The as-prepared waterborne epoxy curing agent was characterized by Fourier transform-infrared spectroscopy and surface tension. The cured film of emulsified oily epoxy resin (E51) prepared by the as-prepared curing agent indirectly was characterized by scanning electron microscopy, thermogravimetric analysis and differential scanning calorimetry tests. The 2K waterborne epoxy coating film prepared by the as-prepared nonionic self-emulsifiable curing agent and E51 displayed better thermal property, surface appearance, flexibility, impact resistance, adhesion, and chemical corrosion resistance.







Similar content being viewed by others

References
Peiliang, C, Jianying, Y, Shuanfa, C, “Effects of Epoxy Resin Contents on the Rheological Properties of Epoxy-Asphalt Blends.” J. Appl. Polym. Sci., 118 (6) 3678–3684 (2010)
Li, H, Zhang, Z, Ma, X, et al., “Synthesis and Characterization of Epoxy Resin Modified with Nano-SiO2 and γ -Glycidoxypropyltrimethoxy Silane.” Surf. Coat. Technol., 201 (9) 5269–5272 (2007)
Kornmann, X, Lindberg, H, Berglund, LA, “Synthesis of Epoxy–Clay Nanocomposites. Influence of the Nature of the Curing Agent on Structure.” Polymer, 42 (10) 4493–4499 (2001)
Han, S, Meng, Q, Pan, X, et al., “Synergistic Effect of Graphene and Carbon Nanotube on Lap Shear Strength and Electrical Conductivity of Epoxy Adhesives.” J. Appl. Polym. Sci., 136 48056 (2019)
Liu, X, Wang, Y, Cao, Y, et al., “Study of Dextrin-Derived Curing Agent for Waterborne Epoxy Adhesive.” Carbohydr. Polym., 83 (3) 1180–1184 (2011)
Shi, X, Nguyen, TA, Suo, Z, et al., “Effect of Nanoparticles on the Anticorrosion and Mechanical Properties of Epoxy Coating.” Surf. Coat. Technol., 204 (3) 237–245 (2009)
Que, X, Yan, Y, Qiu, Z, et al., “Synthesis and Characterization of Trifluoromethyl-Containing Polyimide-Modified Epoxy Resins.” J. Mater. Sci., 51 (24) 1–16 (2016)
Naderi, R, Attar, MM, “The Role of Zinc Aluminum Phosphate Anticorrosive Pigment in Protective Performance and Cathodic Disbondment of Epoxy Coating.” Corros. Sci., 52 (4) 1291–1296 (2010)
Sharmin, E, Imo, L, Ashraf, SM, et al., “Acrylic-Melamine Modified DGEBA-Epoxy Coatings and Their Anticorrosive Behavior.” Prog. Org. Coat., 50 (1) 47–54 (2004)
Papalos JG, Shah S, Grinstein RH, et al., “Self-dispersing Curable Epoxy Resins, Dispersions Made Therewith, and Coating Compositions Made Therefrom.” U.S. Patent 6, 303, 672, 2001
Walker FH, Amide-containing Self-emulsifying Epoxy Curing Agent.” U.S. Patent 5, 596, 030, 1997
Liu, S, Gu, L, Zhao, H, et al., “Corrosion Resistance of Graphene-Reinforced Waterborne Epoxy Coatings.” J. Mater. Sci. Technol., 32 (5) 425–431 (2016)
Wang, N, Zhang, Y, Chen, J, et al., “Dopamine Modified Metal-Organic Frameworks on Anti-corrosion Properties of Waterborne Epoxy Coatings.” Prog. Org. Coat., 109 126–134 (2017)
Li, SQ, Wang, F, Wang, Y, et al., “Effect of Acid and TETA Modification on Mechanical Properties of MWCNTs/Epoxy Composites.” J. Mater. Sci., 43 (8) 2653–2658 (2008)
Jian, R, Wang, P, Xia, L, et al., “Low-Flammability Epoxy Resins with Improved Mechanical Properties Using a Lewis Base Based on Phosphaphenanthrene and 2-Aminothiazole.” J. Mater. Sci., 52 (16) 1–15 (2017)
Ruiz, MM, Cavaillé, JY, Dufresne, A, et al., “New Waterborne Epoxy Coatings Based on Cellulose Nanofillers.” Macromol. Symp., 169 (1) 211–222 (2001)
Wang, F, Liu, Y, Xu, L, et al., “Progress of Curing Agents for Waterborne Epoxy Coatings.” Paint Coat. Ind., 8 008 (2004)
Zhang, J, Zhang, W, Lu, J, et al., “Aqueous Epoxy-Based Superhydrophobic Coatings: Fabrication and Stability in Water.” Prog. Org. Coat., 121 201–208 (2018)
Shimp DA, Hicks DD, Graver RB, “Two Component Aqueous Coating Composition Based on an Epoxy-Polyamine Adduct and a Polyepoxide.” U.S. Patent 4, 246, 148, 1981
Zhang, K, Huang, C, Fang, Q, et al., “Synthesis of a Self-emulsifiable Waterborne Epoxy Curing Agent Based on Glycidyl Tertiary Carboxylic Ester and Its Cure Characteristics.” J. Appl. Polym. Sci., 134 6 (2017)
Ai, D, Mo, R, Wang, H, Lai, Y, Jia, X, Zhang, X, “Preparation of Waterborne Epoxy Dispersion and Its Application in 2 K Waterborne Epoxy Coatings.” Prog. Org. Coat., 136 105258 (2019)
Walker FH, “Self-emulsifying Epoxy Curing Agent Based on the Reaction Product of Epoxy Resin and Polyether Polyol.” U.S. Patent 5, 527, 839, 1996
Rodrıguez, MT, Gracenea, JJ, Saura, JJ, et al., “The Influence of the Critical Pigment Volume Concentration (CPVC) on the Properties of an Epoxy Coating: Part II. Anticorrosion and Economic Properties.” Prog. Org. Coat., 50 (1) 68–74 (2004)
Funding
This study was funded by the National Natural Science Foundation of China (51576070).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, X., Wang, L., Lai, Y. et al. Fabrication of a nonionic self-emulsifiable waterborne epoxy curing agent with high cure properties. J Coat Technol Res 18, 549–558 (2021). https://doi.org/10.1007/s11998-020-00423-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-020-00423-3