Abstract
The efficient performance of cell-immobilized biochar in removing paraquat (PQ) from contaminated water is described in this work. Pseudomonas putida was immobilized onto coconut fiber-derived biochar (BC) using the adsorption method and the covalent binding method. The FTIR spectrum analysis revealed that the main adsorption mechanism of bacteria and BC relates to the interaction between cell surface proteins and the functional group on biochar. The pseudo-second-order model could explain the kinetics of simultaneous adsorption and biodegradation of cell immobilization on BC. Cell-immobilized biochar using the covalent bonding method and adsorption method exhibited superior PQ removal capacity with 30–35% increases in efficiency compared with simple biochar. After 48 h using cell-immobilized biochar, the PQ was transformed to 4,4-bipyridyl and malic acid as detected by GC/MS. From the PQ kinetic removal rate and bacterial retention, the covalent binding method with 16.79 mg/g paraquat adsorption capacity and 0.626 g/mg/h kinetic rate constant is considered as the best cell-immobilization method for the immobilization technique. The cell immobilization on biochar using both the adsorption method and the covalent binding method can support bacteria to degrade PQ into small molecular substances in water treatment processes.
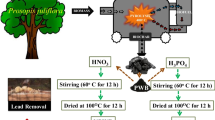
Graphic abstract











Similar content being viewed by others
Availability of data and materials
The authors declare the availability of data and material is appeared as it is shown in this manuscript.
Code availability
Not applicable.
References
Agarry SE, Aremu MO (2012) Batch equilibrium and kinetic studies of simultaneous adsorption and biodegradation of naphthalene by orange peels immobilized Pseudomonas aeruginosa NCIB 950. J Bioremed Biodegrad 3:138. https://doi.org/10.4172/2155-6199.1000138
Amondham W, Parkpian P, Polprasert C, Delaune RD, Jugsujinda A (2006) Paraquat adsorption, degradation, and remobilization in tropical soils of Thailand. J Environ Sci Health, Part B 41:485–507. https://doi.org/10.1080/03601230600701635
Angalaeeswari K, Kamaludeen SPB (2017) Production and characterization of coconut shell and mesquite wood biochar. Int J Chem Stud 5(4):442–446
Banerjee P, Barman SR, Swarnakar S, Mukhopadhyay A, Das P (2018) Treatment of textile effluent using bacteria-immobilized graphene oxide nanocomposites: evaluation of effluent detoxification using Bellamya bengalensis. Clean Technol Environ Policy 20:2287–2298. https://doi.org/10.1007/s10098-018-1602-7
Camacho-Morales RL, Gerardo-Gerardo Jl, Sánchez KGNJE (2017). Producción de enzimas igninolíticas durante la degradación del herbicida paraquat por hongos de la pudrición blanca. Rev Argent Microbiol 49(2):189–196. https://doi.org/10.1016/j.ram.2016.11.004
Carr RJG, Bilton RF, Atkinson T (1985) Mechanism of biodegradation of paraquat by Lipomyces starkeyi. Appl Environ Microbiol 49:1290–1294
Chen Y, Yu B, Lin J, Naidu R, Chen Z (2016) Simultaneous adsorption and biodegradation (SAB) of diesel oil using immobilized Acinetobacter venetianus on porous material. Chem Eng 289:463–470. https://doi.org/10.1016/j.cej.2016.01.010
Cheng C, Lehmann J, Thies JE, Burton SD, Engelhard MH (2006) Oxidation of black carbon by biotic and abiotic processes. Org Geochem 37:1477–1488. https://doi.org/10.1016/j.orggeochem.2006.06.022
Dean CR (1999) The microbial biodegradation of paraquat in soil. Pestic Sci 55:566–614
Dhaouadi A, Adhoum N (2009) Degradation of paraquat herbicide by electrochemical advanced oxidation methods. J Electroanal Chem 637:33–42. https://doi.org/10.1016/j.jelechem.2009.09.027
Dinis-Oliveira RJ, Duarte JA, Sánchez-Navarro A, Remião F, Carvalho F (2008) Paraquat poisonings: mechanisms of lung toxicity, clinical features, and treatment. Crit Rev Toxicol 38:13–71. https://doi.org/10.1080/10408440701669959
Donnison AM, Ross CM, Russell JM (1993) Quality control of bacterial enumeration. Appl Environ Microbiol 59:922–923. https://doi.org/10.1128/AEM.59.3.922-923.1993
Elzinga EJ, Huang JH, Chorover J, Kretzschmar R (2012) ATR-FTIR spectroscopy study of the influence of pH and contact time on the adhesion of Shewanella putrefaciens bacterial cells to the surface of hematite. Environ Sci Technol 46:12848–12855. https://doi.org/10.1021/es303318y
Fang L, Cao Y, Huang Q, Walker SL, Cai P (2012) Reactions between bacterial exopolymers and goethie: a combined macroscopic and spectroscopic investigation. Water Res 46(17):5613–5620. https://doi.org/10.1016/j.watres.2012.07.046
Fu Y, Xu X, Huang Y, Hu J, Chen Q, Wu Y (2017) Preparation of new diatomite-chitosan composite materials and their adsorption properties and mechanism of Hg(II). R Soc Open Sci 4:1–10. https://doi.org/10.1098/rsos.170829
Gulrez SKH, Al-Assaf S, Philips GO (2011) Hydrogels: methods of preparation, characterisation and applications. In: Carpi A (ed) Progress in molecular and environmental bioengineering—from analysis and modeling to technology applications. InTech, London, pp 117–150. https://doi.org/10.5772/24553
Hou D, Shen X, Luo Q, He Y, Wang Q, Liu Q (2013) Enhancement of the diesel oil degradation ability of a marine bacterial strain by immobilization on a novel compound carrier material. Mar Pollut Bull 67:146–151. https://doi.org/10.1016/j.marpolbul.2012.11.021
Huang Y, Zhan H, Bhatt P, Chen S (2019) Paraquat degradation from contaminated environments: current achievements and perspectives. Front Microbiol 10:1754. https://doi.org/10.3389/fmicb.2019.01754
Jimenez BJ, Rivas PR, Lopez JG, Pesciaroli C, Barghini P, Fenice M (2012) Immobilization of Delftia tsuruhatensis in macro-porous cellulose and biodegradation of phenolic compounds in repeated batch process. J Biotechnol 157:148–153. https://doi.org/10.1016/j.jbiotec.2011.09.026
Kilonzo P, Bergougnou M (2012) Surface modifications for controlled and optimized cell immobilization by adsorption: applications in fibrous bed bioreactors containing recombinant cells. J Microb Biochem Technol 4:22–30. https://doi.org/10.4172/1948-5948.s8-001
Kopytko M, Chalela G, Zauscher F (2002) Biodegradation of two commercial herbicides (Gramoxone and Matancha) by the bacteria Pseudomonas putida. Electron J Biotechnol 5(2):182–195. https://doi.org/10.2225/vol5-issue2-fulltext-1
Lasch P, Boese M, Pacifico A, Diem M (2002) FT-IR spectroscopic investigations of single cells on the subcellular level. Vib Spectrosc 28:147–157. https://doi.org/10.1016/S0924-2031(01)00153-9
Lin Q, Donghui W, Jianlong W (2010) Biodegradation of pyridine by Paracoccus sp. KT-5 immobilized on bamboo-based activated carbon. Bioresour Technol 101(14):5229–5234. https://doi.org/10.1016/j.biortech.2010.02.059
Liu J, Duan C, Zhou J, Li X, Qian G, Xu ZP (2013) Adsorption of bacteria onto layered double hydroxide particles to form biogranule-like aggregates. Appl Clay Sci 75–76:39–45. https://doi.org/10.1016/j.clay.2013.02.007
Lou L, Huang Q, Lou Y, Lu J, Hu B, Lin Q (2019) Adsorption and degradation in the removal of nonylphenol from water by cells immobilized on biochar. Chemosphere 228:676–684. https://doi.org/10.1016/j.chemosphere.2019.04.151
Ma X, Li N, Jiang J, Xu Q, Li H, Wang L, Lu J (2013) Adsorption-synergic biodegradation of high-concentrated phenolic water by Pseudomonas putida immobilized on activated carbon fiber. J Environ Chem Eng 1:466–472. https://doi.org/10.1016/j.jece.2013.06.014
Margino S, Martani E, Magdalena M (2007) Superoxide dismutase of Micrococcus sp. S2 and its involve in paraquat detoxification. Indones J Biotechnol 12(1):973–979. https://doi.org/10.22146/IJBIOTECH.7768
Najafpour G (1990) Immobilization of microbial cells for the production of organic acids. J Sci I R Iran 1(3):172–176
Nguyen TC, Quach AB, Tungtakanpoung D, Kajitvichyanukul K (2020) Kinetic, isotherm and mechanism in paraquat removal by adsorption processes using different biochars. Lowel Technol Int 22(2):304–317
Nikolajeva V, Griba T, Petriņa Z (2012) Factors influencing adhesion of Pseudomonas putida on porous clay ceramic granules. Environ Exp Bot 10:77–80. https://doi.org/10.22364/EER
Ojeda JJ, Romero-Gonzalez ME, Pouran HM, Banwart SA (2008) In situ monitoring of the biofilm formation of Pseudomonas putida on hematite using flow-cell ATR-FTIR spectroscopy to investigate the formation of inner-sphere bonds between the bacteria and the mineral. Miner Mag 72(1):101–106. https://doi.org/10.1180/minmag.2008.072.1.101
Omoike A, Chorover J (2006) Adsorption to goethite of extracellular polymeric substances from Bacillus subtilis. Geochim Cosmochim Acta 70:827–838. https://doi.org/10.1016/j.gca.2005.10.012
Quach AB, Kajitvichyanukul P (2019) Adsorption mechanism of dichlorvos onto coconut fibre biochar: the significant dependence of H-bonding and the pore-filling mechanism. Water Sci Technol 79(5):866–876. https://doi.org/10.2166/wst.2018.529
Ren LY, Hong ZN, Qian W, Li JY, Xu RK (2018) Adsorption mechanism of extracellular polymeric subtances from two bacteria on Ultisol and Alfisol. Environ Pollut 237:39–49. https://doi.org/10.1016/j.envpol.2018.01.075
Rong X, Huang Q, He X, Chen H, Cai P, Liang W (2008) Interaction of Pseudomonas putida with kaolinite and montmorillonite: a combination study by equilibrium adsorption, ITC, SEM and FTIR. Colloids Surf B Biointerfaces 64:49–55. https://doi.org/10.1016/j.colsurfb.2008.01.008
Sam AT (2014) The use of biochar to remediate two coastal savannah soils contaminated with Atrazine and Paraquat. Dissertation, University of Ghana
Santos MSF, Alves A, Madeira LM (2011) Paraquat removal from water by oxidation with Fenton’s reagent. Chem Eng J 175:279–290. https://doi.org/10.1016/j.cej.2011.09.106
Santos MSF, Schaule G, Alves A, Madeira LM (2013) Adsorption of paraquat herbicide on deposits from drinking water networks. Chem Eng J 229:324–333. https://doi.org/10.1016/j.cej.2013.06.008
Shahee SM, Niazi NK, Hassan NEE, Bibi I, Wang H, Tsang DCW, Ok YS, Bolan N, Rinklebe J (2019) Wood-based biochar for the removal of potentially toxic elements in water and wastewater: a critical review. Int Mater Rev 64(4):216–247. https://doi.org/10.1080/09506608.2018.1473096
Tsai WT, Chen HR (2013) Adsorption kinetics of herbicide paraquat in aqueous solution onto a low-cost adsorbent, swine-manure-derived biochar. Int J Environ Sci Technol 10:1349–1356. https://doi.org/10.1007/s13762-012-0174-z
Tu CM, Bollen WB (1968) Interaction between paraquat and microbes in soil. Weed Res 8:38–45
Ueshima M, Ginn BR, Haack EA, Szymanowski JES, Fein JB (2008) Cd adsorption onto Pseudomonas putida in the presence and absence of extracellular polymeric substances. Geochim Cosmochim Acta 72:5885–5895. https://doi.org/10.1016/j.gca.2008.09.014
Watts M (2012) Paraquat. http://dev.panap.net/sites/default/files/pesticides-factsheethhps-paraquat.pdf. Accessed 30 Aug 2020
Yavari S, Malakahmad A, Sapari NB (2015) Biochar efficiency in pesticides sorption as a function of production variables - a review. Environ Sci Pollut Res 22:13824–13841. https://doi.org/10.1007/s11356-015-5114-2
Yee N, Fein JB, Daughney CJ (2000) Experimental study of the pH, ionic strength, and reversibility behavior of bacteria-mineral adsorption. Geochim Cosmochim Acta 64(4):609–617. https://doi.org/10.1016/S0016-7037(99)00342-7
Youngwilai T, Kidkhunthod P, Jearanaikoon N, Chaiprapa J, Supanchaiyamat N, Hunt AJ, Ngernyen Y, Ratpukdi T, Khan E, Ratpukdi SS (2020) Simultaneous manganese adsorption and biotransformation by Streptomyces violarus strain SBP1 cell-immobilized biochar. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.136708
Acknowledgements
The authors would like to thank you Thailand Research Fund (Grant No. BRG6180009 and Grant No. IRN62W0005) and Program Management Unit C (Grant No. C16F630255) for the funding support for this work. This research work was also partially supported by Naresuan University and the Center of Excellence in Materials Science and Technology, Chiang Mai University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest or competing interest that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ha, N.T.H., Toan, N.C. & Kajitvichyanukul, P. Enhanced paraquat removal from contaminated water using cell-immobilized biochar. Clean Techn Environ Policy 24, 1073–1085 (2022). https://doi.org/10.1007/s10098-020-01996-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-020-01996-8