Abstract
The golden chanterelle represents one of the commonly found, edible mushrooms that is highly valued in various cuisines. The present study focused on assessing the requirements of Cantharellus cibarius such as pH, temperature, as well as the carbon and nitrogen sources for mycelial growth. Optimization of the growth parameters was carried out by one-factor-at-a-time method. The optimal pH and temperature were determined to be 6.0 and 22.5 °C, respectively. Among the various carbon sources studied, sucrose at a concentration of 2% gave maximum mycelial growth and proved to be the most suitable one. Amongst the nitrogen sources studied, peptone, ammonium sulphate, and sodium nitrate, gave the maximum mycelial growth at an optimized concentration of 0.5%. In the presence of beef extract and yeast extract, a change in colony pigmentation from yellow to dark grey was observed. Finally, the carbon to nitrogen ratio of 2:0.5 proved to be optimal for mycelial growth. This study is the first report on the optimisation of in vitro growth requirements of C. cibarius.

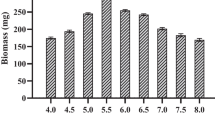
source on mycelial growth of C. cibarius. Data is given as mean of triplicates and error bars represent standard deviation. Data with the same data labels does not vary significantly (p < 0.05; Tukey’s HSD test)


source on mycelial growth of C. cibarius. Data is given as mean of triplicates and error bars represent standard deviation. Data with the same data labels does not vary significantly (p < 0.05; Tukey’s HSD test)


Similar content being viewed by others
References
Barros L, Venturini BA, Baptista P, Estevinho LM, Ferreira IC (2008) Chemical composition and biological properties of Portuguese wild mushrooms: a comprehensive study. J Agric Food Chem 56:3856–3862. https://doi.org/10.1021/jf8003114
Bellettini MB, Fiorda FA, Maieves HA, Teixeira GL, Ávila S, Hornung PS, Júnior AM, Ribani RH (2019) Factors affecting mushroom Pleurotus spp. Saudi J Biol Sci 26:633–646. https://doi.org/10.1016/j.sjbs.2016.12.005
Boonyapranai K, Tungpradit R, Lhieochaiphant S, Phutrakul S (2008) Optimization of submerged culture for the production of naphthoquinones pigment by Fusarium verticillioides. Chiang Mai J Sci 35:457–466
Chen MH, Johns MR (1994) Effect of carbon source on ethanol and pigment production by Monascus purpureus. Enzyme Microb Technol 16:584–590. https://doi.org/10.1016/0141-0229(94)90123-6
Chen MH, Johns MR (1993) Effect of pH and nitrogen source on pigment production by Monascus purpureus. Appl Microbiol Biotechnol 40:132–138. https://doi.org/10.1007/BF00170441
Dong CH, Yao YJ (2005) Nutritional requirements of mycelial growth of Cordyceps sinensis in submerged culture. J Appl Microbiol 99:483–492. https://doi.org/10.1111/j.1365-2672.2005.02640.x
Ebrahimzadeh MA, Nabavi SM, Nabavi SF, Eslami S (2010) Antioxidant and free radical scavenging activities of culinary-medicinal mushrooms, golden chanterelle Cantharellus cibarius and Angel’s wings Pleurotus porrigens. Int J Med Mushrooms. https://doi.org/10.1615/IntJMedMushr.v12.i3.50
Haxo F (1950) Carotenoids of the mushroom Cantharellus cinnabarinus. Bot Gaz 112:228–232. https://doi.org/10.1086/335653
Hoa HT, Wang CL (2015) The effects of temperature and nutritional conditions on mycelium growth of two oyster mushrooms (Pleurotus ostreatus and Pleurotus cystidiosus). Mycobiology 43:14–23. https://doi.org/10.5941/MYCO.2015.43.1.14
Itoo ZA, Reshi ZA (2014) Effect of different nitrogen and carbon sources and concentrations on the mycelial growth of ectomycorrhizal fungi under in-vitro conditions. Scand J For Res 29:619–628. https://doi.org/10.1080/02827581.2014.964756
Kalyoncu F, Oskay M, Kalyoncu M (2009) The effects of some environmental parameters on mycelial growth of six Morchella species. J Pure Appl Microbiol 3:467–472
Kim SW, Hwang HJ, Xu CP, Na YS, Song SK, Yun JW (2002) Influence of nutritional conditions on the mycelial growth and exopolysaccharide production in Paecilomyces sinclairii. Lett Appl Microbiol 34:389–393.
Koehler JF, Miller GL (2017) Impact of nitrogen source and a ph buffer on the in vitro growth and morphology of Rhizoctonia Solani AG 2–2 LP. Int Turfgrass Soc Res J 13:198–202. https://doi.org/10.2134/itsrj2016.07.0592
Kolundžić M, Stanojković T, Radović J, Tačić A, Dodevska M, Milenković M, Sisto F, Masia C, Farronato G, Nikolić V, Kundaković T (2017) Cytotoxic and antimicrobial activities of Cantharellus cibarius Fr. (Cantarellaceae). J Med Food 20:790–796. https://doi.org/10.1089/jmf.2016.0176
Kozarski M, Klaus A, Vunduk J et al (2015) Nutraceutical properties of the methanolic extract of edible mushroom Cantharellus cibarius (Fries): primary mechanisms. Food Funct 6:1875–1886. https://doi.org/10.1039/c5fo00312a
Kumari D, Reddy MS, Upadhyay RC (2011) Antioxidant activity of three species of wild mushroom genus Cantharellus collected from North-Western Himalaya, India. Int J Agric Biol 13:415–418
Lemieszek MK, Nunes FM, Cardoso C, Marques G, Rzeski W (2018) Neuroprotective properties of Cantharellus cibarius polysaccharide fractions in different in vitro models of neurodegeneration. Carbohydr Polym 197:598–607. https://doi.org/10.1016/j.carbpol.2018.06.038
Mantovani TRD, Linde GA, Colauto NB (2007) Effect of the addition of nitrogen sources to cassava fiber and carbon-to-nitrogen ratios on Agaricus brasiliensis growth. Can J Microbiol 53:139–143. https://doi.org/10.1139/W06-112
Mensah-Attipoe J, Toyinbo O (2019) Fungal Growth and Aerosolization from Various Conditions and Materials. In: Loreto ÉS and Tondolo JSM (Eds) Fungal Infection, IntechOpen.
Naraian R, Sahu RK, Kumar S, Garg SK, Singh CS, Kanaujia RS (2009) Influence of different nitrogen rich supplements during cultivation of Pleurotus florida on corn cob substrate. Environmentalist 29:1. https://doi.org/10.1007/s10669-008-9174-4
Nasim G, Malik SH, Bajwa R, Afzal M, Mian SW (2001) Effect of three different culture media on mycelial growth of oyster and Chinese Mushrooms. J Biol Sci. https://doi.org/10.3923/jbs.2001.1130.1133
Neto SLM, Matheus DR, Machado KMG (2009) Influence of pH on the growth, laccase activity and RBBR decolorization by tropical basidiomycetes. Brazilian Arch Biol Technol 52:1075–1082. https://doi.org/10.1590/S1516-89132009000500003
Nowacka-Jechalke N, Nowak R, Juda M, Malm A, Lemieszek M, Rzeski W, Kaczyński Z (2018) New biological activity of the polysaccharide fraction from Cantharellus cibarius and its structural characterization. Food Chem 268:355–361. https://doi.org/10.1016/j.foodchem.2018.06.106
Nyman AAT, Aachmann FL, Rise F, Ballance S, Samuelsen ABC (2016) Structural characterization of a branched (1 → 6)-α-mannan and β-glucans isolated from the fruiting bodies of Cantharellus cibarius. Carbohydr Polym 146:197–207. https://doi.org/10.1016/j.carbpol.2016.03.052
Palacios I, Lozano M, Moro C, D’arrigo M, Rostagno MA, Martínez JA, García-Lafuente A, Guillamón E, Villares A, (2011) Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem 128:674–678. https://doi.org/10.1016/j.foodchem.2011.03.085
Pamire M, Parawira W (2020) Spore germination for three edible Zimbabwean mushrooms using biostimulants. Tanzania J Sci 46:129–136
Pilz D (2003) Ecology and management of commercially harvested chanterelle mushrooms. USDA For Serv - Gen Tech Rep PNW. https://doi.org/10.2737/PNW-GTR-576
Sandmann G, Misawa N (2002) Fungal Carotenoids. In: Osiewacz HD (ed) Industrial Applications. Springer, Berlin, Heidelberg, pp 247–262
Schmidt CG, Furlong EB (2012) Effect of particle size and ammonium sulfate concentration on rice bran fermentation with the fungus Rhizopus oryzae. Bioresour Technol 123:36–41. https://doi.org/10.1016/j.biortech.2012.07.081
Straatsma G, Van Griensven LJLD (1986) Growth requirements of mycelial cultures of the mycorrhizal mushroom Cantharellus cibarius. Trans Br Mycol Soc 87:135–141. https://doi.org/10.1016/s0007-1536(86)80013-4
Wang Z, Wang H, Kang Z, Wu Y, Xing Y, Yang Y (2020) Antioxidant and anti-tumour activity of triterpenoid compounds isolated from Morchella mycelium. Arch Microbiol 13:1–9. https://doi.org/10.1007/s00203-020-01876-1
Wu JZ, Cheung PCK, Wong KH, Huang NL (2004) Studies on submerged fermentation of Pleurotus tuber-regium (Fr.) Singer. Part 2: Effect of carbon-to-nitrogen ratio of the culture medium on the content and composition of the mycelial dietary fibre. Food Chem 85:101–105. https://doi.org/10.1016/j.foodchem.2003.06.009
Yuan B, Chi X, Zhang R (2012) Optimization of exopolysaccharides production from a novel strain of Ganoderma lucidum CAU5501 in submerged culture. Brazilian J Microbiol 43:490–497. https://doi.org/10.1590/S1517-83822012000200009
Zervakis G, Philippoussis A, Ioannldou S, Dlamantopoulou P (2001) Mycelium growth kinetics and optimal temperature conditions for the cultivation of edible mushroom species on lignocellulosic substrates. Folia Microbiol (Praha) 46:231. https://doi.org/10.1007/BF02818539
Acknowledgement
The authors wish to thank Mr. Antti Kinnunen for administrative service and Mrs. Heli Valtonen for her technical assistance. We appreciate and are grateful for the financial support of Regional Council of Southern Savo, Finland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Deshaware, S., Marathe, S.J., Bedade, D. et al. Investigation on mycelial growth requirements of Cantharellus cibarius under laboratory conditions. Arch Microbiol 203, 1539–1545 (2021). https://doi.org/10.1007/s00203-020-02142-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-02142-0