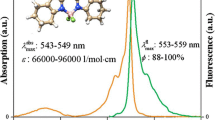
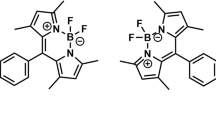
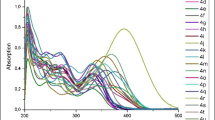
Quinoline-based BODIPY AIEgen dyes were synthesized and the structures were elucidated by 1H, 13C, 19F NMR, FT-IR spectroscopy and mass spectrometry methods. Their photophysical properties were investigated. The dyes showed fluorescence quantum yield in the range of 0.16–1.29% in MeOH. It was found that the presence of methoxy group and tetrazole moiety led to blue and red spectral shift, respectively, of the UV absorption maxima of these dyes compared to their chloroquinoline analog. Stokes shifts of the dyes were in the range of 637–955 cm–1. Aggregation-induced emission behavior of the dyes was investigated in EtOH–H2O mixture so that the dyes exhibited 1.6- to 2.3-fold fluorescence enhancement.




Similar content being viewed by others
References
Arslan, B. S.; Güzel, E.; Kaya, T.; Durmaz, V.; Keskin, M.; Avcı, D.; Nebioğlu, M.; Şişman, İ. Dyes Pigm. 2019, 164, 188.
Gupta, S. S.; Kumari, S.; Kumar, I.; Sharma, U. Chem. Heterocycl. Compd. 2020, 56, 433. [Khim. Geterotsikl. Soedin. 2020, 56, 433.]
Prajapati, S. M.; Patel, K. D.; Vekariya, R. H.; Panchal, S. N.; Patel, H. D. RSC Adv. 2014, 4, 24463.
Liu, Y.; Yang, L.; Ma, C.; Tang, A. Dyes Pigm. 2020, 173, 107981.
Wang, L.; Qian, Y. A. J. Photochem. Photobiol., A 2019, 372, 122.
Benelhadj, K.; Retailleau, P.; Massue, J.; Ulrich, G. Tetrahedron Lett. 2016, 57, 1976.
Matsuoka, R.; Nabeshima, T. Front. Chem. 2018, 6, 349.
Dudina, N. A.; Nikonova, A. Y.; Antina, Y. V.; Berezin, M. B.; Vyugin, A. I. Chem. Heterocycl. Compd. 2014, 49, 1740. [Khim. Geterotsikl. Soedin. 2013, 1878.]
Treibs, A.; Kreuzer, F. H. Justus Liebigs Ann. Chem. 1968, 718, 208.
Boens, N.; Leen, V.; Dehaen, W. Chem. Soc. Rev. 2012, 41, 1130.
Boens, N.; Verbelen, B.; Ortiz, M. J.; Jiao, L.; Dehaen, W. Coord. Chem. Rev. 2019, 399, 213024.
Loudet, A.; Burgess, K. Chem. Rev. 2007, 107, 4891.
Gao, J.; Tao, Y.; Wang, N.; He, J.; Zhang, J.; Zhao, W. Spectrochim. Acta, Part A 2018, 203, 77.
Qin, W.; Dou, W.; Leen, V.; Dehaen, W.; Van der Auweraer, M.; Boens, N. A. RSC Adv. 2016, 6, 7806.
Kamkaew, A.; Lim, S. H.; Lee, H. B.; Kiew, L. V.; Chung, L. Y.; Burgess, K. Chem. Soc. Rev. 2013, 42, 77.
Yadav, R. K.; Baeg, J. O.; Kumar, A.; Kong, K. J.; Oh, G. H.; Park, N. J. J. Mater. Chem. A 2014, 2, 5068.
Yang, H.; Zhao, M.; Zhang, J.; Ma, J.; Wu, P.; Liu, W.; Wen, L. J. Mater. Chem. A 2019, 7, 20742.
Thumuganti, G.; Gupta, V.; Singh, S. P. New J. Chem. 2019, 43, 8735.
Luo, J.; Xie, Z.; Xie, Z.; Lam, J. W. Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H. S.; Zhan, X.; Liu, Y.; Zhu, D.; Tang, B. Z. Chem. Commun. 2001, 1740.
Yamaguchi, M.; Ito, S.; Hirose, A.; Tanaka, K.; Chujo, Y. Mater. Chem. Front. 2017, 1, 1573.
Cheng, B.; Xu, J. Phosphorus, Sulfur Silicon Relat. Elem. 2017, 192, 518.
Meth-Cohn, O.; Narine, B.; Tarnowski, B.; Hayes, R.; Keyzad, A.; Rhouati, S.; Robinson, A. A. J. Chem. Soc., Perkin Trans. 1 1981, 2509.
Chandraprakash, K.; Ramesh, P.; Ravichandran, K.; Mohan, P. S.; Ponnuswamy, M. N. Acta Crystallogr., Sect. E: Struct. Rep. Online 2010, 66, o2510.
Ladani, N. K.; Patel, M. P.; Patel, R. G. ARKIVOC 2009, (vii), 292.
Singh, R. S.; Gupta, R. K.; Paitandi, R. P.; Dubey, M.; Sharma, G.; Koch, B.; Pandey, D. S. Chem. Commun. 2015, 9125.
Choi, S.; Bouffard, J.; Kim, Y. Chem. Sci. 2014, 5, 751.
Prasannan, D.; Raghav, D.; Sujatha, S.; Hareendrakrishna Kumar, H.; Rathinasamy, K.; Arunkumar, C. RSC Adv. 2016, 6, 80808.
Sørensen, M. L. H.; Vosch, T.; Laursen, B. W.; Hansen, T. Photochem. Photobiol. Sci. 2019, 18, 1315.
Patalag, L. J.; Jones, P. G.; Werz, D. B. Chem.–Eur. J. 2017, 23, 15903.
Patalag, L. J.; Loch, M.; Jones, P. G.; Werz, D. B. J. Org. Chem. 2019, 84, 7804.
Dumas-Verdes, C.; Miomandre, F.; Lépicier, E.; Galangau, O.; Vu, T. T.; Clavier, G.; Méallet-Renault, R.; Audebert, P. Eur. J. Org. Chem. 2010, 2525.
Duran-Sampedro, G.; Agarrabeitia, A. R.; Garcia-Moreno, I.; Costela, A.; Bañuelos, J.; Arbeloa, T.; López Arbeloa, I.; Chiara, J. L.; Ortiz, M. J. Eur. J. Org. Chem. 2012, 6335.
Zhang, X. F.; Zhang, I.; Liu, L. Photochem. Photobiol. 2010, 86, 492.
Rohand, T.; Baruah, M.; Qin, W.; Boens, N.; Dehaen, W. Chem. Commun. 2006, 266.
Qin, W.; Rohand, T.; Baruah, M.; Stefan, A.; Van der Auweraer, M.; Dehaen, W.; Boens, N. Chem. Phys. Lett. 2006, 420, 562.
Singh, R. S.; Kumar, A.; Mukhopadhyay, S.; Sharma, G.; Koch, B.; Pandey, D. S. J. Phys. Chem. C 2016, 120, 22605.
Qian, J.; Tang, B. Z. Chem 2017, 3, 56.
Goud, T. V.; Tutar, A.; Biellmann, J. F. Tetrahedron 2006, 62, 5084.
Neena, K. K.; Thilagar, P. ChemPlusChem 2016, 81, 955.
Benniston, A. C.; Copley, G.; Elliott, K. J.; Harrington, R. W.; Clegg, W. Eur. J. Org. Chem. 2008, 2705.
Armarego, W. L. F. Purification of Laboratory Chemicals; Elsevier: Amsterdam, 2008, 8th ed.
Swinehart, F. J. Chem. Educ. 1962, 39, 333.
Casey, K. G.; Quitevis, E. L. J. Phys. Chem. 1988, 92, 6590.
Rurack, K.; Spieles, M. Anal. Chem. 2011, 83, 1232.
Derin, Y.; Yılmaz, R. F.; Baydilek, İ. H.; Atalay, V. E.; Özdemir, A.; Tutar, A. Inorg. Chim. Acta 2018, 482, 130.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(12), 1542–1547
Supplementary Information
ESM 1
(PDF 1742 kb)
Rights and permissions
About this article
Cite this article
Derin, Y., Arslan, B.S., Mısır, B.A. et al. Synthesis and photophysical investigation of AIEgen dyes bearing quinoline and BODIPY scaffolds. Chem Heterocycl Comp 56, 1542–1547 (2020). https://doi.org/10.1007/s10593-020-02847-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02847-6