Abstract
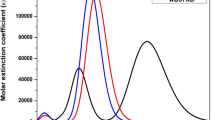
This contribution reports on the photosensitizing competence of Sn(IV)tetrakis(4-pyridyl)porphyrins (SnTP) for potential applications in dye sensitized solar cell (DSSC). The photosensitization of SnTPs is accounted on the basis of different axial ligands (OH−, Cl−, and H2O) and the pyridine protons. The qualitative information on the photovoltaic performance of the sensitizers is obtained at B3LYP/6-31+G(d) level of theory with LANL2DZ basis set for Sn atom through the calculation of parameters such as free energy change of electron injection (\(\Delta G^{\text{inject}}\)) and regeneration (\(\Delta G^{\text{reg}}\)), oxidation potentials, exciton binding energy, open-circuit voltage, light harvesting efficiency and quantum reactivity descriptors. The evaluation of solvent effects is carried out in Tetrahydrofuran (THF) and Dimethylformamide (DMF) solutions employing conductor-like polarizable continuum model (C-PCM). The overall results emphasize that the enhanced photo-sensing action can be achieved by the Sn complexes having OH− and Cl− ligands without pyridine protonation.
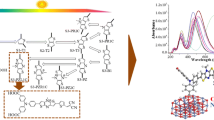
Graphic Abstract
Axial ligation and pyridine protonation significantly affect the electronic excitation properties of Sn complexes. The overall results recommend the application of [(OH−)Sn(OH−)TP] and [(Cl−)Sn(Cl−)TP] photosensitizers in dye sensitized solar cells.





Similar content being viewed by others
References
B. O’Regan, M. Grätzel, Nature 353(6346), 737–740 (1991)
P. Brogdon, H. Cheema, J.H. Delcamp, Chemsuschem 11(1), 86–103 (2018)
A. Polman, M. Knight, E.C. Garnett, B. Ehrler, W.C. Sinke, Science 352(6283), aad4424 (2016)
A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo, H. Pettersson, Chem. Rev. 110(11), 6595–6663 (2010)
H. Song, Q. Liu, Y. Xie, Chem. Commun. 54(15), 1811–1824 (2018)
L.L. Estrella, D.H. Kim, Sol. Energy 188, 1031–1040 (2019)
N. Robertson, Angew. Chem. Int. Ed. 45(15), 2338–2345 (2006)
L. Giribabu, R.K. Kanaparthi, Curr. Sci. 104(7), 847–855 (2013)
S. Karthikeyan, J.Y. Lee, J. Phys. Chem. A 117(42), 10973–10979 (2013)
W. Sang-aroon, S. Saekow, V. Amornkitbamrung, J. Photochem. Photobiol. A Chem. 236, 35–40 (2012)
H. Imahori, T. Umeyama, S. Ito, Acc. Chem. Res. 42(11), 1809–1818 (2009)
F. Arkan, M. Izadyar, Mater. Chem. Phys. 196, 142–152 (2017)
P. Jayachandran, A. Angamuthu, P. Gopalan, J. Mol. Model. 25(9), 294 (2019)
L.-L. Li, E.W.-G. Diau, Chem. Soc. Rev. 42(1), 291–304 (2013)
N.A. Sánchez-Bojorge, G. Zaragoza-Galán, N.R. Flores-Holguín, M.A. Chávez-Rojo, C. Castro-García, L.M. Rodríguez-Valdez, J. Mol. Struct. 1191, 259–270 (2019)
S. Mathew, A. Yella, P. Gao, R. Humphry-Baker, B.F.E. Curchod, N. Ashari-Astani, I. Tavernelli, U. Rothlisberger, M.K. Nazeeruddin, M. Grätzel, Nat. Chem. 6(3), 242–247 (2014)
P. Ren, C. Sun, Y. Shi, P. Song, Y. Yang, Y. Li, J. Mater. Chem. C. 7(7), 1934–1947 (2019)
P. Selvaraj, H. Baig, T.K. Mallick, J. Siviter, A. Montecucco, W. Li, M. Paul, T. Sweet, M. Gao, A.R. Knox, S. Sundaram, Sol. Energy Mater. Sol. Cells 175, 29–34 (2018)
A. Knott, O. Makarovskiy, J. O’Shea, Y. Wu, C. Tuck, Sol. Energy Mater. Sol. Cells 180, 103–109 (2018)
W. Ma, Y. Jiao, S. Meng, J. Phys. Chem. C 118(30), 16447–16457 (2014)
M.P. Balanay, D.H. Kim, Comput. Theor. Chem. 1029, 1–12 (2014)
Y.-H. Sun, C.-K. Wang, Chin. Phys. B 20(10), 104204 (2011)
H. Cheema, A. Baumann, E.K. Loya, P. Brogdon, L.E. McNamara, C.A. Carpenter, N.I. Hammer, S. Mathew, C. Risko, J.H. Delcamp, A.C.S. Appl, Mater. Interfaces 11(18), 16474–16489 (2019)
A. Yella, H.-W. Lee, H.N. Tsao, C. Yi, A.K. Chandiran, M.K. Nazeeruddin, E.W.-G. Diau, C.-Y. Yeh, S.M. Zakeeruddin, M. Grätzel, Science 334(6056), 629 (2011)
F. Gao, C.-L. Yang, M.-S. Wang, X.-G. Ma, Y.-G. Yi, J. Photochem. Photobiol. A Chem. 379, 24–31 (2019)
M. Adineh, P. Tahay, A. Shahrjerdi, S.S.H. Davarani, N. Safari, J. Iran. Chem. Soc. 13(7), 1357–1365 (2016)
Z. Parsa, P. Tahay, N. Safari, J. Iran. Chem. Soc. 17(2), 453–459 (2020)
H. Song, W. Tang, S. Zhao, Q. Liu, Y. Xie, Dyes Pigm. 155, 323–331 (2018)
M. Xie, F.-Q. Bai, J. Wang, Y.-Q. Zheng, Z. Lin, Phys. Chem. Chem. Phys. 20(5), 3741–3751 (2018)
A.S. Shalabi, A.M. El Mahdy, H.O. Taha, J. Nanopart. Res. 15(6), 1696 (2013)
V.S. Shetti, M. Ravikanth, J. Porphyrins Phthalocyanines 14(04), 361–370 (2010)
B. Babu, E. Amuhaya, D. Oluwole, E. Prinsloo, J. Mack, T. Nyokong, MedChemComm 10(1), 41–48 (2019)
D.P. Arnold, J. Blok, Coord. Chem. Rev. 248(3), 299–319 (2004)
F. Gao, C.-L. Yang, M.-S. Wang, X.-G. Ma, W.-W. Liu, Spectrochim. Acta A Mol. Biomol. Spectrosc. 195, 176–183 (2018)
T. Hashimoto, Y.-K. Choe, H. Nakano, K. Hirao, J. Phys. Chem. A 103(12), 1894–1904 (1999)
J. Rani, A. Raveendran, Sushila, A. Chaudhary, M.K. Panda, R. Patra, Cryst. Growth Des. 18(3), 1437–1447 (2018)
Z. Wang, C.J. Medforth, J.A. Shelnutt, J. Am. Chem. Soc. 126(49), 15954–15955 (2004)
O. Bajjou, A. Bakour, M. Khenfouch, M. Baitoul, B. Mothudi, M. Maaza, E. Faulques, J. Phys. Conf. Ser. 984, 012004 (2018)
D.-M. Ren, Z. Guo, F. Du, Z.-F. Liu, Z.-C. Zhou, X.-Y. Shi, Y.-S. Chen, J.-Y. Zheng, Int. J. Mol. Sci. 9(1), 45–55 (2008)
T. Lazarides, S. Kuhri, G. Charalambidis, M.K. Panda, D.M. Guldi, A.G. Coutsolelos, Inorg. Chem. 51(7), 4193–4204 (2012)
M.-Y. Duan, J. Li, G. Mele, C. Wang, X.-F. Lü, G. Vasapollo, F.-X. Zhang, J. Phys. Chem. C 114(17), 7857–7862 (2010)
S. Zargari, R. Rahimi, A. Yousefi, RSC Adv. 6(29), 24218–24228 (2016)
M. Ravikumar, D. Raghav, K. Rathinasamy, A. Kathiravan, E.M. Mothi, A.C.S. Appl, Bio Mater. 1(5), 1705–1716 (2018)
M. Ravikumar, A. Kathiravan, A. Neels, E.M. Mothi, Eur. J. Inorg. Chem. 2018(34), 3868–3877 (2018)
A.-M. Manke, K. Geisel, A. Fetzer, P. Kurz, Phys. Chem. Chem. Phys. 16(24), 12029–12042 (2014)
P. Jayachandran, A. Angamuthu, P. Gopalan, J. Chin. Chem. Soc. 65(8), 908–917 (2018)
A.D. Becke, J. Chem. Phys. 98(7), 5648–5652 (1993)
C. Lee, W. Yang, R.G. Parr, Phys. Rev. B. 37(2), 785–789 (1988)
P.J. Hay, W.R. Wadt, J. Chem. Phys. 82(1), 270–283 (1985)
N. Santhanamoorthi, C.-M. Lo, J.-C. Jiang, J. Phys. Chem. Lett. 4(3), 524–530 (2013)
M. Cossi, N. Rega, G. Scalmani, V. Barone, J. Comput. Chem. 24(6), 669–681 (2003)
M. Szafran, M.M. Karelson, A.R. Katritzky, J. Koput, M.C. Zerner, J. Comput. Chem. 14(3), 371–377 (1993)
S. Miertuš, E. Scrocco, J. Tomasi, Chem. Phys. 55(1), 117–129 (1981)
G.W.T.M.J. Frisch, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian 09, Revision B. 01. Gaussian, Inc., Wallingford CT (2009)
I.N. Obotowo, I.B. Obot, U.J. Ekpe, J. Mol. Struct. 1122, 80–87 (2016)
F. Zanjanchi, J. Beheshtian, J. Iran. Chem. Soc. 16(4), 795–805 (2019)
A. Hagfeldt, M. Grätzel, Acc. Chem. Res. 33(5), 269–277 (2000)
A. Pramanik, S. Sarkar, S. Pal, P. Sarkar, Phys. Lett. A 379(14), 1036–1042 (2015)
C.-R. Zhang, Z.-J. Liu, Y.-H. Chen, H.-S. Chen, Y.-Z. Wu, W. Feng, D.-B. Wang, Curr. Appl. Phys. 10(1), 77–83 (2010)
J. Feng, Y. Jiao, W. Ma, M.K. Nazeeruddin, M. Grätzel, S. Meng, J. Phys. Chem. C 117(8), 3772–3778 (2013)
R. Katoh, A. Furube, T. Yoshihara, K. Hara, G. Fujihashi, S. Takano, S. Murata, H. Arakawa, M. Tachiya, J. Phys. Chem. B. 108(15), 4818–4822 (2004)
D. Rehm, A. Weller, Isr. J. Chem. 8(2), 259–271 (1970)
J.L. Goodman, K.S. Peters, J. Am. Chem. Soc. 108(7), 1700–1701 (1986)
J.B. Asbury, Y.-Q. Wang, E. Hao, H.N. Ghosh, T. Lian, Res. Chem. Intermed. 27(4), 393–406 (2001)
D. Cahen, G. Hodes, M. Grätzel, J.F. Guillemoles, I. Riess, J. Phys. Chem. B 104(9), 2053–2059 (2000)
Y. Fu, T. Lu, Y. Xu, M. Li, Z. Wei, H. Liu, W. Lu, Dyes Pigm. 155, 292–299 (2018)
J.-C. Lee, J.-D. Chai, S.-T. Lin, RSC Adv. 5(123), 101370–101376 (2015)
P.K. Nayak, N. Periasamy, Org. Electron. 10(7), 1396–1400 (2009)
R. Meenakshi, J. Mol. Struct. 1127, 694–707 (2017)
K. Chaitanya, X.-H. Ju, B.M. Heron, RSC Adv. 4(51), 26621–26634 (2014)
A. Calborean, F. Graur, V. Bintintan, J. Mol. Model. 23(6), 181 (2017)
H.Y. Ammar, H.M. Badran, Heliyon 5(10), e02545 (2019)
A.L. Arokiyanathan, S. Lakshmipathi, New J. Chem. 42(5), 3465–3472 (2018)
M.-S. Liao, P. Bonifassi, J. Leszczynski, M.-J. Huang, Mol. Phys. 106(1), 147–160 (2008)
F. Pichierri, A. Sekine, T. Yamamoto, Mater. Trans. 56(9), 1425–1427 (2015)
M. Cao, A. Gao, Y. Liu, Y. Zhou, Z. Sun, Y. Li, F. He, L. Li, L. Mo, R. Liu, Y. Han, Y. Yang, Catalysts 10(2), 224 (2020)
M.D. Davari, H. Bahrami, M. Zahedi, N. Safari, J. Mol. Model. 15(11), 1299–1315 (2009)
G.V. Girichev, N.I. Giricheva, O.I. Koifman, Y.V. Minenkov, A.E. Pogonin, A.S. Semeikin, S.A. Shlykov, Dalton Trans. 41(25), 7550–7558 (2012)
X. Wang, S. Li, L. Zhao, C. Xu, J. Gao, Chin. J. Chem. Eng. 28(2), 532–540 (2020)
K. Sharma, V. Sharma, S.S. Sharma, Nanoscale Res. Lett. 13(1), 381 (2018)
J. Preat, C. Michaux, D. Jacquemin, E.A. Perpète, J. Phys. Chem. C 113(38), 16821–16833 (2009)
L.-J. He, J. Chen, F.-Q. Bai, R. Jia, J. Wang, H.-X. Zhang, Dyes Pigm. 141, 251–261 (2017)
Z.-H. Yang, C.A. Ullrich, Phys. Rev. B. 87(19), 195204 (2013)
B.A. Gregg, J. Phys. Chem. B. 107(20), 4688–4698 (2003)
B.-G. Kim, K. Chung, J. Kim, Chem. Eur. J. 19(17), 5220–5230 (2013)
B.-G. Kim, C.-G. Zhen, E.J. Jeong, J. Kieffer, J. Kim, Adv. Funct. Mater. 22(8), 1606–1612 (2012)
B. Ali, N. Allam, Spectrochim. Acta A Mol. Biomol. Spectrosc. 188, 237 (2018)
T. Delgado-Montiel, R. Soto-Rojo, J. Baldenebro-López, D. Glossman-Mitnik, Molecules 24(21), 3897 (2019)
Acknowledgements
The authors are thankful to “Bioinformatics Resources and Applications Facility (BRAF), C-DAC, Pune for offering the computational facilities to carry out this work. PG is grateful to the Science and Engineering Research Board (SERB), India.
Funding
This work received FastTrack Research Grant (Project No. SB/FTP/PS-096/2013) from the Science and Engineering Research Board (SERB), India.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jayachandran, P., Angamuthu, A. & Gopalan, P. Dye sensitized solar cell action of Sn(IV)tetrakis(4-pyridyl) porphyrins as a function of axial ligation and pyridine protonation. J IRAN CHEM SOC 18, 1523–1536 (2021). https://doi.org/10.1007/s13738-020-02129-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-020-02129-0