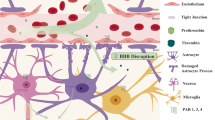
Abstract
Cerebral ischemia is a cerebrovascular disease with high morbidity and mortality that poses a significant burden on society and the economy. About 60% of cerebral ischemia is caused by thrombus, and the formation of thrombus proceeds from insoluble fibrin, following its transformation from liquid fibrinogen. In thrombus-induced ischemia, increased permeability of the blood–brain barrier (BBB), followed by the extravasation of blood components into the brain results in an altered brain microenvironment. Changes in the brain microenvironment affect brain function and the neurovascular unit (NVU), the working unit of the brain. Recent studies have reported that coagulation factors interact with the NVU and its components, but the specific function of this interaction is highly speculative and warrants further investigations. In this article, we reviewed the role of coagulation factors in cerebral ischemia and the role of coagulation factors in thrombosis. Additionally, the influence of thrombin on the NVU is introduced, as well as in the function of NVU, which may help to explore part of brain injury mechanism during ischemia. Lastly, we propose some novel therapeutic approaches on ischemic stroke by reducing the risk of coagulation.



Similar content being viewed by others
Data Availability
Not applicable.
Code availability
Not applicable.
References
Amantea D, Micieli G, Tassorelli C et al (2015) Rational modulation of the innate immune system for neuroprotection in ischemic stroke. Front Neurosci 2015(9):147. https://doi.org/10.3389/fnins.2015.00147
Arumugam TV, Baik SH, Balaganapathy P et al (2018) Notch signaling and neuronal death in stroke. Prog Neurobiol 165:103–116. https://doi.org/10.1016/j.pneurobio.2018.03.002
Bao L, Zu J, He Q et al (2017) Thrombin-induced apoptosis in neurons through activation of c-Jun-N-terminal kinase. Toxicol Mech Methods 27(1):18–23. https://doi.org/10.3109/15376516.2016.1172691
Bao X, Hua Y, Keep RF et al (2018) Thrombin-induced tolerance against oxygen-glucose deprivation in astrocytes: role of protease-activated receptor-1. Conditioning medicine 1(2):57–63
Becerra-Calixto A, Cardona-Gómez GP (2017) The Role of Astrocytes in Neuroprotection after Brain Stroke: Potential in Cell Therapy. Frontiers in molecular neuroscience 10:88. https://doi.org/10.3389/fnmol.2017.00088
Ben Shimon M, Lenz M, Ikenberg B et al (2015) Thrombin regulation of synaptic transmission and plasticity: implications for health and disease. Frontiers in cellular neuroscience 9:151. https://doi.org/10.3389/fncel.2015.00151
Bi M, Guo A, Zhao H et al (2015) Role of the extracellular signal-regulated kinase 1/2 signaling pathway in the process of thrombin-promoting airway remodeling in ovalbumin-allergic rats. Immunopharmacol Immunotoxicol 37(1):26–34. https://doi.org/10.3109/08923973.2014.993083
Blanchette M, Daneman R (2015) Formation and maintenance of the BBB. Mech Dev 138(Pt 1):8–16. https://doi.org/10.1016/j.mod.2015.07.007
Bochenek ML, Schäfer K (2019) Role of Endothelial Cells in Acute and Chronic Thrombosis. Hamostaseologie 39(2):128–139. https://doi.org/10.1055/s-0038-1675614
Bunch ME, Nunziato EC, Labovitz DL (2012) Barriers to the use of intravenous tissue plasminogen activator for in-hospital strokes. Journal of stroke and cerebrovascular diseases: the official journal of National Stroke Association 21(8):808–811. https://doi.org/10.1016/j.jstrokecerebrovasdis.2011.04.012
Bushi D, Ben Shimon M, Shavit Stein E et al (2015) Increased thrombin activity following reperfusion after ischemic stroke alters synaptic transmission in the hippocampus. J Neurochem 135(6):1140–1148. https://doi.org/10.1111/jnc.13372
Bushi D, Gera O, Kostenich G et al (2016) A novel histochemical method for the visualization of thrombin activity in the nervous system. Neuroscience 320:93–104. https://doi.org/10.1016/j.neuroscience.2016.01.065
Bushi D, Chapman J, Wohl A et al (2018) Apixaban decreases brain thrombin activity in a male mouse model of acute ischemic stroke. J Neurosci Res 96(8):1406–1411. https://doi.org/10.1002/jnr.24253
Cai W, Zhang K, Li P et al (2017) Dysfunction of the neurovascular unit in ischemic stroke and neurodegenerative diseases: An aging effect. Ageing research reviews 34:77–87. https://doi.org/10.1016/j.arr.2016.09.006
Canfield SG, Stebbins MJ, Morales BS et al (2017) An isogenic blood-brain barrier model comprising brain endothelial cells, astrocytes, and neurons derived from human induced pluripotent stem cells. J Neurochem 140(6):874–888. https://doi.org/10.1111/jnc.13923
Carrim N, Arthur JF, Hamilton JR et al (2015) Thrombin-induced reactive oxygen species generation in platelets: A novel role for protease-activated receptor 4 and GPIbα. Redox biology 6:640–647. https://doi.org/10.1016/j.redox.2015.10.009
Cassella CR, Jagoda A (2017) Ischemic Stroke: Advances in Diagnosis and Management. Emerg Med Clin North Am 35(4):911–930. https://doi.org/10.1016/j.emc.2017.07.007
Chapin JC, Hajjar KA (2015) Fibrinolysis and the control of blood coagulation. Blood Rev 29(1):17–24. https://doi.org/10.1016/j.blre.2014.09.003
Chen B, Friedman B, Whitney MA et al (2012) Thrombin activity associated with neuronal damage during acute focal ischemia. The Journal of neuroscience: the official journal of the Society for Neuroscience 32(22):7622–7631. https://doi.org/10.1523/JNEUROSCI.0369-12.2012
Chen ZQ, Mou RT, Feng DX et al (2017) The role of nitric oxide in stroke. Medical gas research 7(3):194–203. https://doi.org/10.4103/2045-9912.215750
Chen S, Chen Z, Cui J et al (2018) Early Abrogation of Gelatinase Activity Extends the Time Window for tPA Thrombolysis after Embolic Focal Cerebral Ischemia in Mice. eNeuro. https://doi.org/10.1523/ENEURO.0391-17.2018
Choi MS, Kim YE, Lee WJ et al (2008) Activation of protease-activated receptor1 mediates induction of matrix metalloproteinase-9 by thrombin in rat primary astrocytes. Brain Res Bull 76(4):368–375. https://doi.org/10.1016/j.brainresbull.2008.02.031
Choi HJ, Kim NE, Kim J et al (2018) Dabigatran reduces endothelial permeability through inhibition of thrombin-induced cytoskeleton reorganization. Thromb Res S0049–3848(18):30324–30334. https://doi.org/10.1016/j.thromres.2018.04.019
Choudhury GR, Ding S (2016) Reactive astrocytes and therapeutic potential in focal ischemic stroke. Neurobiology of disease 85:234–244. https://doi.org/10.1016/j.nbd.2015.05.003
Clark VD, Layson A, Charkviani M et al (2018) Hyperfibrinogenemia-mediated astrocyte activation. Brain Res 1699:158–165. https://doi.org/10.1016/j.brainres.2018.08.023
Colombo E, Farina C (2016) Astrocytes: Key Regulators of Neuroinflammation. Trends Immunol 37(9):608–620. https://doi.org/10.1016/j.it.2016.06.006
Cuartero MI, de la Parra J, García-Culebras A et al (2016) The Kynurenine Pathway in the Acute and Chronic Phases of Cerebral Ischemia. Curr Pharm Des 22(8):1060–1073. https://doi.org/10.2174/1381612822666151214125950
Dalkara T, Alarcon-Martinez L, Yemisci M (2019) Pericytes in Ischemic Stroke. Adv Exp Med Biol 1147:189–213. https://doi.org/10.1007/978-3-030-16908-4_9
De Luca C, Virtuoso A, Maggio N et al (2017) Neuro-Coagulopathy: Blood Coagulation Factors in Central Nervous System Diseases. Int J Mol Sci 18(10):2128. https://doi.org/10.3390/ijms18102128
De Luca C, Colangelo AM, Alberghina L et al (2018) Neuro-Immune Hemostasis: Homeostasis and Diseases in the Central Nervous System. Frontiers in cellular neuroscience 12:459. https://doi.org/10.3389/fncel.2018.00459
Dhar A, Sadiq F, Anstee QM et al (2018) Thrombin and factor Xa link the coagulation system with liver fibrosis. BMC gastroenterology 18(1):60. https://doi.org/10.1186/s12876-018-0789-8
Ding H, Gao S, Wang L et al (2019) Overexpression of miR-582-5p Inhibits the Apoptosis of Neuronal Cells after Cerebral Ischemic Stroke Through Regulating PAR-1/Rho/Rho Axis. Journal of stroke and cerebrovascular diseases: the official journal of National Stroke Association 28(1):149–155. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.09.023
Dittmeier M, Wassmuth K, Schuhmann K, M, et al (2016) Dabigatran Etexilate Reduces Thrombin-Induced Inflammation and Thrombus Formation in Experimental Ischemic Stroke. Current neurovascular research 13(3):199–206. https://doi.org/10.2174/1567202613666160517122605
Dong T, Chen N, Ma X et al (2018) The protective roles of L-borneolum, D-borneolum and synthetic borneol in cerebral ischaemia via modulation of the neurovascular unit. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 102:874–883. https://doi.org/10.1016/j.biopha.2018.03.087
Ebrahimi S, Jaberi N, Avan A et al (2017) Role of thrombin in the pathogenesis of central nervous system inflammatory diseases. J Cell Physiol 232(3):482–485. https://doi.org/10.1002/jcp.25501
ElAli A (2016) The implication of neurovascular unit signaling in controlling the subtle balance between injury and repair following ischemic stroke. Neural regeneration research 11(6):914–915. https://doi.org/10.4103/1673-5374.184485
Fan F, Yang J, Xu Y et al (2018) MiR-539 Targets MMP-9 to Regulate the Permeability of Blood-Brain Barrier in Ischemia/Reperfusion Injury of Brain. Neurochem Res 43(12):2260–2267. https://doi.org/10.1007/s11064-018-2646-0
Festoff BW, Citron BA (2019) Thrombin and the Coag-Inflammatory Nexus in Neurotrauma, ALS, and Other Neurodegenerative Disorders. Frontiers in neurology 10:59. https://doi.org/10.3389/fneur.2019.00059
Festoff BW, Sajja RK, van Dreden P et al (2016) HMGB1 and thrombin mediate the blood-brain barrier dysfunction acting as biomarkers of neuroinflammation and progression to neurodegeneration in Alzheimer’s disease. Journal of neuroinflammation 13(1):194. https://doi.org/10.1186/s12974-016-0670-z
Fisher M, Saver JL (2015) Future directions of acute ischaemic stroke therapy. The Lancet Neurology 14(7):758–767. https://doi.org/10.1016/S1474-4422(15)00054-X
Gera O, Shavit-Stein E, Bushi D et al (2016) Thrombin and protein C pathway in peripheral nerve Schwann cells. Neuroscience 339:587–598. https://doi.org/10.1016/j.neuroscience.2016.10.034
Geranmayeh MH, Rahbarghazi R, Farhoudi M (2019) Targeting pericytes for neurovascular regeneration. Cell communication and signaling: CCS 17(1):26. https://doi.org/10.1186/s12964-019-0340-8
Gerard ILLR, Palomer E, Ramos-Fernández E et al (2015) Fibrinogen nitrotyrosination after ischemic stroke impairs thrombolysis and promotes neuronal death. Biochem Biophys Acta 1852(3):421–428. https://doi.org/10.1016/j.bbadis.2014.12.007
Gleeson EM, Dichiara MG, Salicio A et al (2015) Activated protein C β-glycoform promotes enhanced noncanonical PAR1 proteolysis and superior resistance to ischemic injury. Blood 126(7):915–919. https://doi.org/10.1182/blood-2015-03-632877
Göbel K, Eichler S, Wiendl H et al (2018) The Coagulation Factors Fibrinogen, Thrombin, and Factor XII in Inflammatory Disorders-A Systematic Review. Frontiers in immunology 9:1731. https://doi.org/10.3389/fimmu.2018.01731
Gofrit SG, Shavit-Stein E (2019) The neuro-glial coagulonome: the thrombin receptor and coagulation pathways as major players in neurological diseases. Neural regeneration research 14(12):2043–2053. https://doi.org/10.4103/1673-5374.262568
Golanov EV, Sharpe MA, Regnier-Golanov AS et al (2019) Fibrinogen Chains Intrinsic to the Brain. Frontiers in neuroscience 13:541. https://doi.org/10.3389/fnins.2019.00541
Goto M, Miura S, Suematsu Y et al (2016) Rivaroxaban, a factor Xa inhibitor, induces the secondary prevention of cardiovascular events after myocardial ischemia reperfusion injury in mice. Int J Cardiol 220:602–607. https://doi.org/10.1016/j.ijcard.2016.06.212
Griffin JH, Zlokovic BV, Mosnier LO (2015) Activated protein C: biased for translation. Blood 125(19):2898–2907. https://doi.org/10.1182/blood-2015-02-355974
Grover SP, Mackman N (2018) Tissue Factor: An Essential Mediator of Hemostasis and Trigger of Thrombosis. Arterioscler Thromb Vasc Biol 38(4):709–725. https://doi.org/10.1161/ATVBAHA.117.309846
Grover SP, Mackman N (2019) Intrinsic Pathway of Coagulation and Thrombosis. Arterioscler Thromb Vasc Biol 39(3):331–338. https://doi.org/10.1161/ATVBAHA.118.312130
Haim LB, Rowitch DH (2017) Functional diversity of astrocytes in neural circuit regulation. Nat Rev Neurosci 18(1):31–41. https://doi.org/10.1038/nrn.2016.159
Han JY, Li Q, Ma ZZ et al (2017) Effects and mechanisms of compound Chinese medicine and major ingredients on microcirculatory dysfunction and organ injury induced by ischemia/reperfusion. Pharmacol Ther 177:146–173. https://doi.org/10.1016/j.pharmthera.2017.03.005
Hawkins BT, Gu YH, Izawa Y et al (2015) Dabigatran abrogates brain endothelial cell permeability in response to thrombin. Journal of cerebral blood flow and metabolism: official journal of the International Society of Cerebral Blood Flow and Metabolism 35(6):985–992. https://doi.org/10.1038/jcbfm.2015.9
He Q, Bao L, Zimering J et al (2015) The protective role of (-)-epigallocatechin-3-gallate in thrombin-induced neuronal cell apoptosis and JNK-MAPK activation. NeuroReport 26(7):416–423. https://doi.org/10.1097/WNR.0000000000000363
Hirt L, Price M, Mastour N et al (2018) Increase of aquaporin 9 expression in astrocytes participates in astrogliosis. J Neurosci Res 96(2):194–206. https://doi.org/10.1002/jnr.24061
Hosoo H, Marushima A, Nagasaki Y et al (2017) Neurovascular Unit Protection From Cerebral Ischemia-Reperfusion Injury by Radical-Containing Nanoparticles in Mice. Stroke 48(8):2238–2247. https://doi.org/10.1161/STROKEAHA.116.016356
Hu H, Yamashita S, Hua Y et al (2010) Thrombin-induced neuronal protection: role of the mitogen activated protein kinase/ribosomal protein S6 kinase pathway. Brain Res 1361:93–101. https://doi.org/10.1016/j.brainres.2010.09.025
Hu S, Wu G, Ding X et al (2016) Thrombin preferentially induces autophagy in glia cells in the rat central nervous system. Neurosci Lett 630:53–58. https://doi.org/10.1016/j.neulet.2016.07.023
Hultman K, Cortes-Canteli M, Bounoutas A et al (2014) Plasmin deficiency leads to fibrin accumulation and a compromised inflammatory response in the mouse brain. Journal of thrombosis and haemostasis : JTH 12(5):701–712. https://doi.org/10.1111/jth.12553
Iadecola C (2017) The Neurovascular Unit Coming of Age: A Journey through Neurovascular Coupling in Health and Disease. Neuron 96(1):17–42. https://doi.org/10.1016/j.neuron.2017.07.030
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17(7):796–808. https://doi.org/10.1038/nm.2399
Isermann B (2017) Homeostatic effects of coagulation protease-dependent signaling and protease activated receptors. Journal of thrombosis and haemostasis : JTH 15(7):1273–1284. https://doi.org/10.1111/jth.13721
Jiang X, Andjelkovic AV, Zhu L et al (2018) Blood-brain barrier dysfunction and recovery after ischemic stroke. Prog Neurobiol 163–164:144–171. https://doi.org/10.1016/j.pneurobio.2017.10.001
Jin X, Yamashita T (2016) Microglia in central nervous system repair after injury. J Biochem 159(5):491–496. https://doi.org/10.1093/jb/mvw009
Johnson CO, Nguyen M, Roth GA et al (2019) Global, regional, and national burden of stroke, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet Neurology 18(5):439–458. https://doi.org/10.1016/S1474-4422(19)30034-1
Kaminari A, Tsilibary EC, Tzinia A (2018) A New Perspective in Utilizing MMP-9 as a Therapeutic Target for Alzheimer’s Disease and Type 2 Diabetes Mellitus. Journal of Alzheimer’s disease: JAD 64(1):1–16. https://doi.org/10.3233/JAD-180035
Kattula S, Byrnes JR, Wolberg AS (2017) Fibrinogen and Fibrin in Hemostasis and Thrombosis. Arterioscler Thromb Vasc Biol 37(3):e13–e21. https://doi.org/10.1161/ATVBAHA.117.308564
Knowland D, Arac A, Sekiguchi KJ et al (2014) Stepwise recruitment of transcellular and paracellular pathways underlies blood-brain barrier breakdown in stroke. Neuron 82(3):603–617. https://doi.org/10.1016/j.neuron.2014.03.003
Krenzlin H, Lorenz V, Danckwardt S et al (2016) The Importance of Thrombin in Cerebral Injury and Disease. Int J Mol Sci 17(1):84. https://doi.org/10.3390/ijms17010084
Krenzlin H, Gresser E, Jussen D et al (2020) The Cerebral Thrombin System Is Activated after Intracerebral Hemorrhage and Contributes to Secondary Lesion Growth and Poor Neurological Outcome in C57Bl/6 Mice. J Neurotrauma 37(12):1481–1490. https://doi.org/10.1089/neu.2019.6582
Krupka J, May F, Weimer T et al (2016) The Coagulation Factor XIIa Inhibitor rHA-Infestin-4 Improves Outcome after Cerebral Ischemia/Reperfusion Injury in Rats. PLoS ONE 11(1):e0146783. https://doi.org/10.1371/journal.pone.0146783
Kubes P, Suzuki M, Granger DN (1991) Nitric oxide: an endogenous modulator of leukocyte adhesion. Proc Natl Acad Sci USA 88(11):4651–4655. https://doi.org/10.1073/pnas.88.11.4651
Lee JH, Won S, Stein DG (2015) Progesterone attenuates thrombin-induced endothelial barrier disruption in the brain endothelial cell line bEnd. 3: The role of tight junction proteins and the endothelial protein C receptor. Brain Res 1613:73–80. https://doi.org/10.1016/j.brainres.2015.04.002
Li G, Wang Q, Lin T et al (2019) Thrombin Induces Cerebral Vasospasm Effect via Proteinase-Activated Receptor-1 (PAR-1)/Tumor Necrosis Factor-α (TNF-α) Inflammatory Pathways in Subarachnoid Hemorrhage Rat Model. Journal of Biomaterials and Tissue Engineering 9(5):599–606. https://doi.org/10.1166/jbt.2019.2024
Li Y, Zhong W, Jiang Z et al (2019) New progress in the approaches for blood-brain barrier protection in acute ischemic stroke. Brain Res Bull 144:46–57. https://doi.org/10.1016/j.brainresbull.2018.11.006
Liddelow SA, Guttenplan KA, Clarke LE et al (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541(7638):481–487. https://doi.org/10.1038/nature21029
Lin CC, Lee IT, Wu WB et al (2013) Thrombin mediates migration of rat brain astrocytes via PLC, Ca2+, CaMKII, PKCα, and AP-1-dependent matrix metalloproteinase-9 expression. Mol Neurobiol 48(3):616–630. https://doi.org/10.1007/s12035-013-8450-6
Lin R, Yu K, Li X et al (2016) Electroacupuncture ameliorates post-stroke learning and memory through minimizing ultrastructural brain damage and inhibiting the expression of MMP-2 and MMP-9 in cerebral ischemia-reperfusion injured rats. Molecular medicine reports 14(1):225–233. https://doi.org/10.3892/mmr.2016.5227
Lindsay MP, Norrving B, Sacco RL et al (2019) World Stroke Organization (WSO): Global Stroke Fact Sheet 2019. International journal of stroke: official journal of the International Stroke Society 14(8):806–817. https://doi.org/10.1177/1747493019881353
Linville RM, DeStefano JG, Sklar MB et al (2019) Human iPSC-derived blood-brain barrier microvessels: validation of barrier function and endothelial cell behavior. Biomaterials 190–191:24–37. https://doi.org/10.1016/j.biomaterials.2018.10.023
Liu Z, Chopp M (2016) Astrocytes, therapeutic targets for neuroprotection and neurorestoration in ischemic stroke. Prog Neurobiol 144:103–120. https://doi.org/10.1016/j.pneurobio.2015.09.008
Lu Y, Li C, Chen Q et al (2019) Microthrombus-Targeting Micelles for Neurovascular Remodeling and Enhanced Microcirculatory Perfusion in Acute Ischemic Stroke. Advanced materials (Deerfield Beach, Fla.) 31(21):e1808361. https://doi.org/10.1002/adma.201808361
Lyden P, Pereira B, Chen B et al (2014) Direct thrombin inhibitor argatroban reduces stroke damage in 2 different models. Stroke 45(3):896–899. https://doi.org/10.1161/STROKEAHA.113.004488
Machida T, Takata F, Matsumoto J et al (2015) Brain pericytes are the most thrombin-sensitive matrix metalloproteinase-9-releasing cell type constituting the blood-brain barrier in vitro. Neurosci Lett 599:109–114. https://doi.org/10.1016/j.neulet.2015.05.028
Machida T, Dohgu S, Takata F et al (2017) Role of thrombin-PAR1-PKCθ/δ axis in brain pericytes in thrombin-induced MMP-9 production and blood-brain barrier dysfunction in vitro. Neuroscience 350:146–157. https://doi.org/10.1016/j.neuroscience.2017.03.026
Mammadova-Bach E, Ollivier V, Loyau S et al (2015) Platelet glycoprotein VI binds to polymerized fibrin and promotes thrombin generation. Blood 126(5):683–691. https://doi.org/10.1182/blood-2015-02-629717
McConnell HL, Kersch CN, Woltjer RL et al (2017) The Translational Significance of the Neurovascular Unit. The Journal of biological chemistry 292(3):762–770. https://doi.org/10.1074/jbc.R116.760215
Möller T, Hanisch UK, Ransom BR (2000) Thrombin-induced activation of cultured rodent microglia. J Neurochem 75(4):1539–1547. https://doi.org/10.1046/j.1471-4159.2000.0751539.x
Montagne A, Nikolakopoulou AM, Zhao Z et al (2018) Pericyte degeneration causes white matter dysfunction in the mouse central nervous system. Nat Med 24(3):326–337. https://doi.org/10.1038/nm.4482
Moran CS, Seto SW, Krishna SM et al (2017) Parenteral administration of factor Xa/IIa inhibitors limits experimental aortic aneurysm and atherosclerosis. Scientific reports 7:43079. https://doi.org/10.1038/srep43079
Mozaffarian D, Benjamin EJ, Go AS et al (2016) Executive Summary: Heart Disease and Stroke Statistics–2016 Update: A Report From the American Heart Association. Circulation 133(4):447–454. https://doi.org/10.1161/CIR.0000000000000366
Muoio V, Persson PB, Sendeski MM (2014) The neurovascular unit - concept review. Acta physiologica (Oxford, England) 210(4):790–798. https://doi.org/10.1111/apha.12250
Muradashvili N, Tyagi SC, Lominadze D (2017) Localization of Fibrinogen in the Vasculo-Astrocyte Interface after Cortical Contusion Injury in Mice. Brain sciences 7(7):77. https://doi.org/10.3390/brainsci7070077
Nakano T, Nakamura Y, Irie K et al (2020) Antithrombin gamma attenuates macrophage/microglial activation and brain damage after transient focal cerebral ischemia in mice. Life Sci 252:117665. https://doi.org/10.1016/j.lfs.2020.117665
National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group (1995) Tissue plasminogen activator for acute ischemic stroke. N Engl J Med 333(24):1581–1588
Nazir S, Gadi I, Al-Dabet MM et al (2017) Cytoprotective activated protein C averts Nlrp3 inflammasome-induced ischemia-reperfusion injury via mTORC1 inhibition. Blood 130(24):2664–2677. https://doi.org/10.1182/blood-2017-05-782102
Neal M, Richardson JR (2018) Epigenetic regulation of astrocyte function in neuroinflammation and neurodegeneration Biochimica et biophysica acta. Molecular basis of disease 1864(2):432–443. https://doi.org/10.1016/j.bbadis.2017.11.004
Negrier C, Shima M, Hoffman M (2019) The central role of thrombin in bleeding disorders. Blood Rev 38:100582. https://doi.org/10.1016/j.blre.2019.05.006
Nieman MT (2016) Protease-activated receptors in hemostasis. Blood 128(2):169–177. https://doi.org/10.1182/blood-2015-11-636472
Norris EH, Strickland S (2017) Fibrinogen in the Nervous System: Glia Beware. Neuron 96(5):951–953. https://doi.org/10.1016/j.neuron.2017.11.021
Otsuka S, Sakakima H, Terashi T et al (2019) Preconditioning exercise reduces brain damage and neuronal apoptosis through enhanced endogenous 14–3-3γ after focal brain ischemia in rats. Brain structure & function 224(2):727–738. https://doi.org/10.1007/s00429-018-1800-4
Pan Z, Cui M, Dai G et al (2018) Protective Effect of Anthocyanin on Neurovascular Unit in Cerebral Ischemia/Reperfusion Injury in Rats. Frontiers in neuroscience 12:947. https://doi.org/10.3389/fnins.2018.00947
Pera J, Undas A, Topor-Madry R et al (2012) Fibrin clot properties in acute stroke: what differs cerebral hemorrhage from cerebral ischemia? Stroke 43(5):1412–1414. https://doi.org/10.1161/STROKEAHA.111.646729
Periayah MH, Halim AS, Saad AZM (2017) Mechanism Action of Platelets and Crucial Blood Coagulation Pathways in Hemostasis. International journal of hematology-oncology and stem cell research 11(4):319–327
Piao C, Ralay Ranaivo H, Rusie A et al (2015) Thrombin decreases expression of the glutamate transporter GLAST and inhibits glutamate uptake in primary cortical astrocytes via the Rho kinase pathway. Exp Neurol 273:288–300. https://doi.org/10.1016/j.expneurol.2015.09.009
Pijet B, Stefaniuk M, Kostrzewska-Ksiezyk A et al (2018) Elevation of MMP-9 Levels Promotes Epileptogenesis After Traumatic Brain Injury. Mol Neurobiol 55(12):9294–9306. https://doi.org/10.1007/s12035-018-1061-5
Puech C, Delavenne X, He Z et al (2019) Direct oral anticoagulants are associated with limited damage of endothelial cells of the blood-brain barrier mediated by the thrombin/PAR-1 pathway. Brain Res 1719:57–63. https://doi.org/10.1016/j.brainres.2019.05.024
Puig B, Brenna S, Magnus T (2018) Molecular Communication of a Dying Neuron in Stroke. Int J Mol Sci 19(9):2834. https://doi.org/10.3390/ijms19092834
Qi Z, Liu KJ (2019) The interaction of zinc and the blood-brain barrier under physiological and ischemic conditions. Toxicol Appl Pharmacol 364:114–119. https://doi.org/10.1016/j.taap.2018.12.018
Radak D, Katsiki N, Resanovic I et al (2017) Apoptosis and Acute Brain Ischemia in Ischemic Stroke. Curr Vasc Pharmacol 15(2):115–122. https://doi.org/10.2174/1570161115666161104095522
Radulovic M, Yoon H, Wu J et al (2016) Targeting the thrombin receptor modulates inflammation and astrogliosis to improve recovery after spinal cord injury. Neurobiology of disease 93:226–242. https://doi.org/10.1016/j.nbd.2016.04.010
Rajput PS, Kothari S et al (2020) Abstract TP468: Differential Effect of Ischemia and Thrombin Mediated Toxicity on Neurovascular Unit. Stroke 51:ATP468
Rajput PS, Lamb J, Kothari S et al (2020) Neuron-generated thrombin induces a protective astrocyte response via protease activated receptors. Glia 68(2):246–262. https://doi.org/10.1002/glia.23714
Rossi D (2015) Astrocyte physiopathology: At the crossroads of intercellular networking, inflammation and cell death. Prog Neurobiol 130:86–120. https://doi.org/10.1016/j.pneurobio.2015.04.003
Ryu JK, Petersen MA, Murray SG et al (2015) Blood coagulation protein fibrinogen promotes autoimmunity and demyelination via chemokine release and antigen presentation. Nature communications 6:8164. https://doi.org/10.1038/ncomms9164
Schnoor M, García Ponce A, Vadillo E et al (2017) Actin dynamics in the regulation of endothelial barrier functions and neutrophil recruitment during endotoxemia and sepsis. Cellular and molecular life sciences: CMLS 74(11):1985–1997. https://doi.org/10.1007/s00018-016-2449-x
Shavit Stein E, Ben Shimon M, Artan Furman A et al (2018) Thrombin Inhibition Reduces the Expression of Brain Inflammation Markers upon Systemic LPS Treatment. Neural plasticity 2018:7692182. https://doi.org/10.1155/2018/7692182
Stamatovic SM, Philips C, Martinez-Revollar G et al (2019) Involvement of Epigenetic Mechanisms and Non-coding RNAs in Blood-Brain Barrier and Neurovascular Unit Injury and Recovery After Stroke. Frontiers in neuroscience 13:864. https://doi.org/10.3389/fnins.2019.00864
Stein ES, Itsekson-Hayosh Z, Aronovich A et al (2015) Thrombin induces ischemic LTP (iLTP): implications for synaptic plasticity in the acute phase of ischemic stroke. Scientific reports 5:7912. https://doi.org/10.1038/srep07912
Su X, Huang L, Qu Y et al (2019) Pericytes in Cerebrovascular Diseases: An Emerging Therapeutic Target. Frontiers in cellular neuroscience 13:519. https://doi.org/10.3389/fncel.2019.00519
Suo Z, Wu M, Ameenuddin S et al (2002) Participation of protease-activated receptor-1 in thrombin-induced microglial activation. J Neurochem 80(4):655–666. https://doi.org/10.1046/j.0022-3042.2001.00745.x
Sweeney MD, Ayyadurai S, Zlokovic BV (2016) Pericytes of the neurovascular unit: key functions and signaling pathways. Nat Neurosci 19(6):771–783. https://doi.org/10.1038/nn.4288
ten Cate H, Hackeng TM, de Frutos PG (2017) Coagulation factor and protease pathways in thrombosis and cardiovascular disease. Thromb Haemost 117(7):1265–1271. https://doi.org/10.1160/TH17-02-0079
Tomaiuolo M, Brass LF, Stalker TJ (2017) Regulation of Platelet Activation and Coagulation and Its Role in Vascular Injury and Arterial Thrombosis. Interventional cardiology clinics 6(1):1–12. https://doi.org/10.1016/j.iccl.2016.08.001
Travers RJ, Smith SA, Morrissey JH (2015) Polyphosphate, platelets, and coagulation. International journal of laboratory hematology 37:31–35. https://doi.org/10.1111/ijlh.12349
Underly RG, Levy M, Hartmann DA et al (2017) Pericytes as Inducers of Rapid, Matrix Metalloproteinase-9-Dependent Capillary Damage during Ischemia. The Journal of neuroscience: the official journal of the Society for Neuroscience 37(1):129–140. https://doi.org/10.1523/JNEUROSCI.2891-16.2016
Utepbergenov DI, Mertsch K, Sporbert A et al (1998) Nitric oxide protects blood-brain barrier in vitro from hypoxia/reoxygenation-mediated injury. FEBS Lett 424(3):197–201. https://doi.org/10.1016/s0014-5793(98)00173-2
Vidale S, Consoli A, Arnaboldi M, Consoli D (2017) Postischemic Inflammation in Acute Stroke. Journal of clinical neurology (Seoul, Korea) 13(1):1–9. https://doi.org/10.3988/jcn.2017.13.1.1
Vilar R, Fish RJ, Casini A et al (2020) Fibrin(ogen) in human disease: both friend and foe. Haematologica 105(2):284–296. https://doi.org/10.3324/haematol.2019.236901
Wang Z, Higashikawa K, Yasui H et al (2020) FTY720 Protects Against Ischemia-Reperfusion Injury by Preventing the Redistribution of Tight Junction Proteins and Decreases Inflammation in the Subacute Phase in an Experimental Stroke Model. Translational stroke research 11(5):1103–1116. https://doi.org/10.1007/s12975-020-00789-x
Webb AH et al (2017) Inhibition of MMP-2 and MMP-9 decreases cellular migration, and angiogenesis in in vitro models of retinoblastoma. BMC cancer 17(1):434. https://doi.org/10.1186/s12885-017-3418-y
Weekman EM, Wilcock DM (2016) Matrix Metalloproteinase in Blood-Brain Barrier Breakdown in Dementia. Journal of Alzheimer’s disease: JAD 49(4):893–903. https://doi.org/10.3233/JAD-150759
Wolter J, Schild L, Bock F et al (2016) Thrombomodulin-dependent protein C activation is required for mitochondrial function and myelination in the central nervous system. Journal of thrombosis and haemostasis : JTH 14(11):2212–2226. https://doi.org/10.1111/jth.13494
Wu C, Chen J, Chen C et al (2015) Wnt/β-catenin coupled with HIF-1α/VEGF signaling pathways involved in galangin neurovascular unit protection from focal cerebral ischemia. Scientific reports 5:16151. https://doi.org/10.1038/srep16151
Wu C, Yan X, Liao Y et al (2019) Increased perihematomal neuron autophagy and plasma thrombin-antithrombin levels in patients with intracerebral hemorrhage: An observational study. Medicine 98(39):e17130. https://doi.org/10.1097/MD.0000000000017130
Xi G, Reiser G, Keep RF (2003) The role of thrombin and thrombin receptors in ischemic, hemorrhagic and traumatic brain injury: deleterious or protective? J Neurochem 84(1):3–9. https://doi.org/10.1046/j.1471-4159.2003.01268.x
Xie CL, Li JH, Wang WW et al (2015) Neuroprotective effect of ginsenoside-Rg1 on cerebral ischemia/reperfusion injury in rats by downregulating protease-activated receptor-1 expression. Life Sci 121:145–151. https://doi.org/10.1016/j.lfs.2014.12.002
Yang Y, Zhang M, Kang X et al (2015) Thrombin-induced microglial activation impairs hippocampal neurogenesis and spatial memory ability in mice. Behavioral and brain functions: BBF 11(1):30. https://doi.org/10.1186/s12993-015-0075-7
Yang CC, Hsiao LD, Yang CM et al (2017) Thrombin Enhanced Matrix Metalloproteinase-9 Expression and Migration of SK-N-SH Cells via PAR-1, c-Src, PYK2, EGFR, Erk1/2 and AP-1. Mol Neurobiol 54(5):3476–3491. https://doi.org/10.1007/s12035-016-9916-0
Yang S, Jin H, Zhu Y et al (2017) Diverse Functions and Mechanisms of Pericytes in Ischemic Stroke. Curr Neuropharmacol 15(6):892–905. https://doi.org/10.2174/1570159X15666170112170226
Yin X, Wright J, Wall T et al (2010) Brain endothelial cells synthesize neurotoxic thrombin in Alzheimer’s disease. The American journal of pathology 176(4):1600–1606. https://doi.org/10.2353/ajpath.2010.090406
Yoo AJ, Andersson T (2017) Thrombectomy in Acute Ischemic Stroke: Challenges to Procedural Success. Journal of stroke 19(2):121–130. https://doi.org/10.5853/jos.2017.00752
Zagrean AM, Hermann DM, Opris I et al (2018) Multicellular Crosstalk Between Exosomes and the Neurovascular Unit After Cerebral Ischemia. Therapeutic Implications Frontiers in neuroscience 12:811. https://doi.org/10.3389/fnins.2018.00811
Zbesko JC, Nguyen TV, Yang T et al (2018) Glial scars are permeable to the neurotoxic environment of chronic stroke infarcts. Neurobiology of disease 112:63–78. https://doi.org/10.1016/j.nbd.2018.01.007
Zhang S (2019) Microglial activation after ischaemic stroke. Stroke and vascular neurology 4(2):71–74. https://doi.org/10.1136/svn-2018-000196
Zhao B, Shi QJ, Zhang ZZ et al (2018) Protective effects of paeonol on subacute/chronic brain injury during cerebral ischemia in rats. Experimental and therapeutic medicine 15(4):3836–3846. https://doi.org/10.3892/etm.2018.5893
Zhu J, Pan X, Lin B et al (2019) The effect of hirudin on antagonisting thrombin induced apoptosis of human microvascular endothelial cells1. Acta cirurgica brasileira 34(1):e20190010000006. https://doi.org/10.1590/s0102-865020190010000006
Zuo P, Zuo Z, Wang X et al (2015) Factor Xa induces pro-inflammatory cytokine expression in RAW 2647 macrophages via protease-activated receptor-2 activation. American journal of translational research 7(11):2326–2334
Funding
Young Scientists Fund (81603617); Fundamental Research Funds for Central Universities of the Central South University (ZZ13-YQ-007). This work was partially supported by a grant (RC Ref. No. CC-CSTH-2018-145(E)) from the College of Professional and Continuing Education, the Hong Kong Polytechnic University.
Author information
Authors and Affiliations
Contributions
Wenting SONG had the idea for the article. Hui CAO and Sai Wang SETO performed the literature search and drafted this paper, who contributed equally to this work. Deep Jyoti BHUYAN and Hoi Huen CHAN critically revised the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, H., Seto, S.W., Bhuyan, D.J. et al. Effects of Thrombin on the Neurovascular Unit in Cerebral Ischemia. Cell Mol Neurobiol 42, 973–984 (2022). https://doi.org/10.1007/s10571-020-01019-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-01019-6