Abstract
Objective
To evaluate the role of the biocontrol agent Bacillus subtilis CtpxS2-1 in inducing lupin systemic resistance against anthracnose caused by Colletotrichum acutatum by lipopeptide production.
Results
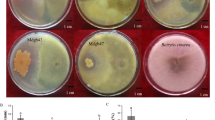
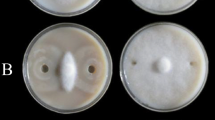
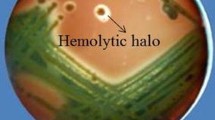
First, growth inhibition and thin layer chromatography-bioautography analysis confirmed that CtpxS2-1 cultures and their lipopeptide extracts, specifically fengycin, have strong antifungal activity against C. acutatum. Subsequent microscopic examination of these fungal inhibition zones showed mycelial pathogen deformations. PCR amplification of CtpxS2-1 confirmed the presence of genes encoding fengycins E and C, bacillomycin C, iturin A, and surfactins B and C. Based on this evidence, the effect of CtpxS2-1 and its lipopeptides on the induction of the lupin defence- and growth-related genes PR-1, PR-4, SOD-2, PIN-1 and PIN-3 was evaluated by RT-qPCR. In seedlings from roots treated with CtxpS2-1, a significant increase in the expression of these genes was induced. Efficacy assays showed that CtpxS2-1 treatment completely controlled anthracnose incidence (0.0%) compared with the untreated control. Furthermore, root and shoot growth in treated seedlings with CtpxS2-1 significantly increased due to disease control, as did the synthesis of the defence enzymes catalase, peroxidase and superoxide dismutase.
Conclusion
B. subtilis CtpxS2-1 is a key factor enhancing Andean lupin health by producing lipopeptides that damage C. acutatum cellular structures and inhibit their growth, as well as by inducing the expression of defence-related genes of lupin plants involved in systemic acquired resistance (SAR) against anthracnose.


Similar content being viewed by others
References
Afzal I, Shinwari ZK, Sikandar S, Shahzad S (2019) Plant beneficial endophytic bacteria: mechanisms, diversity, host range and genetic determinants. Microbiol Res 221:36–49
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26:1–20
Ali S, Ganai BA, Kamili AN et al (2018) Pathogenesis-related proteins and peptides as promising tools for engineering plants with multiple stress tolerance. Microbiol Res 212–213:29–37
Arguelles-Arias A, Ongena M, Halimi B et al (2009) Bacillus amyloliquefaciens GA1 as a source of potent antibiotics and other secondary metabolites for biocontrol of plant pathogens. Microb Cell Fact. https://doi.org/10.1186/1475-2859-8-63
Arnoldi A, Boschin G, Zanoni C, Lammi C (2015) The health benefits of sweet lupin seed flours and isolated proteins. J Funct Foods 18:550–563
Ashwini N, Srividya S (2014) Potentiality of Bacillus subtilis as biocontrol agent for management of anthracnose disease of chilli caused by Colletotrichum gloeosporioides OGC1. 3 Biotech. https://doi.org/10.1007/s13205-013-0134-4
Balseiro-Romero M, Gkorezis P, Kidd PS et al (2017) Use of plant growth promoting bacterial strains to improve Cytisusstriatus and Lupinusluteus development for potential application in phytoremediation. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2016.12.180
Begum M, Sariah M, Puteh AB, ZainalAbidin MA (2007) Detection of seed-borne fungi and site of infection by Colletotrichumtruncatum in naturally-infected soybean seeds. Int J Agric Res 2:812–819
Choudhary DK, Johri BN (2009) Interactions of Bacillus spp. and plants—with special reference to induced systemic resistance (ISR). Microbiol Res 164:493–513
Chowdappa P, Mohan Kumar SP, Jyothi Lakshmi M, Upreti KK (2013) Growth stimulation and induction of systemic resistance in tomato against early and late blight by Bacillus subtilis OTPB1 or Trichodermaharzianum OTPB3. Biol Control 65:109–117. https://doi.org/10.1016/j.biocontrol.2012.11.009
Chowdhury SP, Hartmann A, Gao XW, Borriss R (2015) Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42—a review. Front Microbiol. https://doi.org/10.3389/fmicb.2015.00780
Deleu M, Paquot M, Nylander T (2005) Fengycin interaction with lipid monolayers at the air–aqueous interface—implications for the effect of fengycin on biological membranes. J Colloid Interface Sci 283:358–365. https://doi.org/10.1016/J.JCIS.2004.09.036
Elanchezhiyan K, Keerthana U, Nagendran K et al (2018) Multifaceted benefits of Bacillus amyloliquefaciens strain FBZ24 in the management of wilt disease in tomato caused by Fusariumoxysporum f. sp. lycopersici. Physiol Mol Plant Pathol. https://doi.org/10.1016/j.pmpp.2018.05.008
Falconí CE, Yánez-Mendizábal V (2016) Dry heat treatment of Andean lupin seed to reduce anthracnose infection. Crop Prot. https://doi.org/10.1016/j.cropro.2016.07.021
Falconí CE, Yánez-Mendizábal V (2018) Efficacy of UV-C radiation to reduce seedborne anthracnose (Colletotrichumacutatum) from Andean lupin (Lupinusmutabilis). Plant Pathol. https://doi.org/10.1111/ppa.12793
Falconí CE, Yánez-Mendizábal V (2019) Solar UV-B radiation limits seedborne anthracnose infection and induces physiological and biochemical responses in Lupinusmutabilis. Plant Pathol 68:1635–1644. https://doi.org/10.1111/ppa.13086
Falconi CE, Visser RGF, Van Heusden S (2015) Influence of plant growth stage on resistance to anthracnose in Andean lupin (Lupinusmutabilis). Crop Past Sci. https://doi.org/10.1071/CP14104
Fira D, Dimkić I, Berić T et al (2018) Biological control of plant pathogens by Bacillus species. J Biotechnol. https://doi.org/10.1016/j.jbiotec.2018.07.044
Gao T, Li Y, Ding M et al (2017) The phosphotransferase system gene ptsI in Bacillus cereus regulates expression of sodA2 and contributes to colonization of wheat roots. Res Microbiol 168:524–535. https://doi.org/10.1016/j.resmic.2017.04.003
Gond SK, Bergen MS, Torres MS, White JF (2015) Endophytic Bacillus spp. produce antifungal lipopeptides and induce host defence gene expression in maize. Microbiol Res 172:79–87. https://doi.org/10.1016/j.micres.2014.11.004
Góth L (1991) A simple method for determination of serum catalase activity and revision of reference range. Clin Chim Acta 196:143–151. https://doi.org/10.1016/0009-8981(91)90067-M
Jacobsen S, Mujica A (2006) El tarwi (Lupinusmutabilis Sweet.) y sus parientessilvestres. In: Moraes M, Ollgaard B, Kvist LP, Borchsenius F, Balslev H (eds) Botánica Económica de los Andes Centrales. Universidad Mayor de San Andrés, La Paz, pp 58–482
Jacques P, Hbid C, Destain J et al (1999) Optimization of biosurfactant lipopeptide production from Bacillus subtilis S499 by Plackett-Burman design
Jomová K, Feszterová M, Morovič M (2011) Expression of pathogenesis-related protein genes and changes of superoxide dismutase activity induced by toxic elements in Lupinus luteus L.
Keller J, Imperial J, Ruiz-Argüeso T et al (2018) RNA sequencing and analysis of three Lupinusnodulomes provide new insights into specific host-symbiont relationships with compatible and incompatible Bradyrhizobium strains. Plant Sci 266:102–116. https://doi.org/10.1016/j.plantsci.2017.10.015
Kloepper JW, Ryu C-M, Zhang S (2004) Induced systemic resistance and promotion of plant growth by Bacillusspp. Phytopathology. https://doi.org/10.1094/PHYTO.2004.94.11.1259
Liu X, Huang B (2000) Heat stress injury in relation to membrane lipid peroxidation in creeping bentgrass. Crop Sci 40:503–510
Modi A, Kanani P, Kumar A (2020) 10—Fungal endophytes-induced gene expression studies in biotic and abiotic stress management. In: Kumar AEKR (eds) Microbial endophytes. Woodhead Publishing, pp 251–271
Ongena M, Jacques P (2008) Bacillus lipopeptides: versatile weapons for plant disease biocontrol. Trends Microbiol
Ongena M, Henry G, Thonart P (2010) The roles of cyclic lipopeptides in the biocontrol activity of Bacillus subtilis. Recent developments in management of plant diseases. Springer, Netherlands, pp 59–69
Peralta IE, Mazón N, Murillo IA et al (2008) Manual agrícola de granos andinos: Chocho, quinua, amaranto y ataco. Cultivos, variedades y costos de producción, Manual no. 69. EC: INIAP, Estación Experimental Santa Catalina, Programa Nacional de Leguminosas y Granos Andinos, Quito
Przymusiński R, Rucińska R, Gwóźdź EA (2004) Increased accumulation of pathogenesis-related proteins in response of lupine roots to various abiotic stresses. Environ Exp Bot 52:53–61. https://doi.org/10.1016/j.envexpbot.2004.01.006
Redmile-Gordon MA, Armenise E, White RP et al (2013) A comparison of two colorimetric assays, based upon Lowry and Bradford techniques, to estimate total protein in soil extracts. Soil Biol Biochem. https://doi.org/10.1016/j.soilbio.2013.08.017
Sajitha KL, Dev SA (2016) Quantification of antifungal lipopeptide gene expression levels in Bacillus subtilis B1 during antagonism against sapstain fungus on rubberwood. Biol Control 96:78–85. https://doi.org/10.1016/j.biocontrol.2016.02.007
Sałek K, Euston SR (2019) Sustainable microbial biosurfactants and bioemulsifiers for commercial exploitation. Process Biochem 85:143–155
Vanneste S, Friml J (2009) Reviewauxin: a trigger for change in plant development. Cell. https://doi.org/10.1016/j.cell.2009.03.001
Wang X, Yuan Z, Shi Y et al (2020) Bacillus amyloliquefaciens HG01 induces resistance in loquats against anthracnose rot caused by Colletotrichumacutatum. Postharvest Biol Technol. https://doi.org/10.1016/j.postharvbio.2019.111034
Wu Q, Zhi Y, Xu Y (2019) Systematically engineering the biosynthesis of a green biosurfactant surfactin by Bacillus subtilis 168. Metab Eng 52:87–97. https://doi.org/10.1016/j.ymben.2018.11.004
Yánez-Mendizábal V, Falconí CE (2018) Efficacy of Bacillus spp. to biocontrol of anthracnose and enhance plant growth on Andean lupin seeds by lipopeptide production. Biol Control 122:67–75. https://doi.org/10.1016/j.biocontrol.2018.04.004
Yánez-Mendizábal V, Zeriouh H, Viñas I et al (2012) Biological control of peach brown rot (Monilinia spp.) by Bacillus subtilis CPA-8 is based on production of fengycin-like lipopeptides. Eur J Plant Pathol 132:609–619. https://doi.org/10.1007/s10658-011-9905-0
Yesuf M, Sangchote S (2007) Seed transmission and epidemics of Colletotrichum lindemuthianum in the major common bean growing areas of Ethiopia. Witthayasan Kasetsart (Sakha Witthayasat) 34–45
Acknowledgements
This research was supported by the Secretaria de Educación Superior, Ciencia, Tecnología e Innovación (SENESCYT), Universidad de las Fuerzas Armadas - ESPE and Universidad de Las Américas - UDLA through Proyecto PIC-15-ESPE-001 “Mejora de la cadena productiva del chocho (Lupinus mutabilis) en Ecuador”. The authors thank the Ministerio del Ambiente of Ecuador for authorization to collect microorganisms. Additionally, the authors thank Eng. Byron Freire and Sebastian Cueva for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yánez-Mendizábal, V., Falconí, C.E. Bacillus subtilis CtpxS2-1 induces systemic resistance against anthracnose in Andean lupin by lipopeptide production. Biotechnol Lett 43, 719–728 (2021). https://doi.org/10.1007/s10529-020-03066-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-03066-x