Abstract
Key message
Isoflavones are not involved in rhizobial signaling in red clover, but likely play a role in defense in the rhizosphere.
Abstract
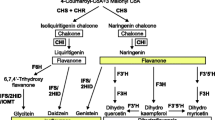
Red clover (Trifolium pratense) is a high-quality forage legume, well suited for grazing and hay production in the temperate regions of the world. Like many legumes, red clover produces a number of phenylpropanoid compounds including anthocyanidins, flavan-3-ols, flavanols, flavanones, flavones, and isoflavones. The study of isoflavone biosynthesis and accumulation in legumes has come into the forefront of biomedical and agricultural research due to potential for medicinal, antimicrobial, and environmental implications. CRISPR/Cas9 was used to knock out the function of a key enzyme in the biosynthesis of isoflavones, isoflavone synthase (IFS1). A hemizygous plant carrying a 9-bp deletion in the IFS1 gene was recovered and was intercrossed to obtain homozygous mutant plants. Levels of the isoflavones formononetin, biochanin A and genistein were significantly reduced in the mutant plants. Wild-type and mutant plants were inoculated with rhizobia to test the effect of the mutation on nodulation, but no significant differences were observed, suggesting that these isoflavones do not play important roles in nodulation. Gene expression profiling revealed an increase in expression of the upstream genes producing the precursors for IFS1, namely, phenylalanine ammonium lyase and chalcone synthase, but there were no significant differences in IFS1 gene expression or in the downstream genes in the production of specific isoflavones. Higher expression in genes involved in ethylene response was observed in the mutant plants. This response is normally associated with biotic stress, suggesting that the plants may have been responding to cues in the surrounding rhizosphere due to lower levels of isoflavones.





Similar content being viewed by others
Data availability
Raw RNA-seq reads are deposited in the Short Read Archive database at GenBank under BioProject PRJNA657870.
References
Ambawat S, Sharma P, Yadav NR, Yadav RC (2013) MYB transcription factor genes as regulators for plant responses: an overview. Physiol Mol Biol Plants 19(3):307–321. https://doi.org/10.1007/s12298-013-0179-1
Ball DM, Hoveland CS, Lacefield GD (2007) Southern forages, 4th edn. International Plant Nutrition Institute, Norcross
Bennetts HW, Underwood EJ, Shier FL (1946) A specific breeding problem of sheep on subterranean clover pastures in Western Australia. Aust Vet J 22(1):2–12. https://doi.org/10.1111/j.1751-0813.1946.tb15473.x
Biała W, Jasiński M (2018) The phenylpropanoid case—it is transport that matters. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01610
Cady N, Peterson SR, Freedman SN, Mangalam AK (2020) Beyond metabolism: the complex interplay between dietary phytoestrogens, gut bacteria, and cells of nervous and immune systems. Front Neurol. https://doi.org/10.3389/fneur.2020.00150
Carlsson G, Huss-Danell K (2003) Nitrogen fixation in perennial forage legumes in the field. Plant Soil 253(2):353–372. https://doi.org/10.1023/a:1024847017371
Cesco S, Neumann G, Tomasi N, Pinton R, Weisskopf L (2010) Release of plant-borne flavonoids into the rhizosphere and their role in plant nutrition. Plant Soil 329(1–2):1–25. https://doi.org/10.1007/s11104-009-0266-9
Chakrabarti M, Dinkins RD, Hunt AG (2016) De novo transcriptome assembly and dynamic spatial gene expression analysis in red clover. Plant Genome US 9(2):1–12. https://doi.org/10.3835/plantgenome2015.06.0048
Chao Y, Yuan J, Li S, Jia S, Han L, Xu L (2018) Analysis of transcripts and splice isoforms in red clover (Trifolium pratense L.) by single-molecule long-read sequencing. BMC Plant Biol 18(1):300. https://doi.org/10.1186/s12870-018-1534-8
Cooper JE (2004) Multiple responses of rhizobia to flavonoids during legume root infection. Adv Bot Res 41:1–62. https://doi.org/10.1016/s0065-2296(04)41001-5
De Vega JJ, Ayling S, Hegarty M, Kudrna D, Goicoechea JL, Ergon A, Rognli OA, Jones C, Swain M, Geurts R, Lang C, Mayer KF, Rossner S, Yates S, Webb KJ, Donnison IS, Oldroyd GE, Wing RA, Caccamo M, Powell W, Abberton MT, Skot L (2015) Red clover (Trifolium pratense L.) draft genome provides a platform for trait improvement. Sci Rep 5:17394. https://doi.org/10.1038/srep17394
Deavours BE, Dixon RA (2005) Metabolic engineering of isoflavonoid biosynthesis in alfalfa. Plant Physiol 138(4):2245–2259. https://doi.org/10.1104/pp.105.062539
Deavours BE, Liu CJ, Naoumkina MA, Tang Y, Farag MA, Sumner LW, Noel JP, Dixon RA (2006) Functional analysis of members of the isoflavone and isoflavanone O-methyltransferase enzyme families from the model legume Medicago truncatula. Plant Mol Biol 62(4–5):715–733. https://doi.org/10.1007/s11103-006-9050-x
Debbarma J, Sarki YN, Saikia B, Boruah HPD, Singha DL, Chikkaputtaiah C (2019) Ethylene response factor (ERF) family proteins in abiotic stresses and CRISPR–Cas9 genome editing of ERFs for multiple abiotic stress tolerance in crop plants: a review. Mol Biotechnol 61(2):153–172. https://doi.org/10.1007/s12033-018-0144-x
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell. https://doi.org/10.1105/tpc.7.7.1085
Dixon RA, Lamb CJ, Masoud S, Sewalt VJH, Paiva NL (1996) Metabolic engineering: prospects for crop improvement through the genetic manipulation of phenylpropanoid biosynthesis and defense responses—a review. Gene 179(1):61–71. https://doi.org/10.1016/s0378-1119(96)00327-7
Evers GW (2011) Forage legumes: forage quality, fixed nitrogen, or both. Crop Sci 51:403–409. https://doi.org/10.2135/cropsci2010.06.0380
Graham TL (1991) Flavonoid and isoflavonoid distribution in developing soybean seedling tissues and in seed and root exudates. Plant Physiol 95(2):594–603. https://doi.org/10.1104/pp.95.2.594
Gronquist M, Bezzerides A, Attygalle A, Meinwald J, Eisner M, Eisner T (2001) Attractive and defensive functions of the ultraviolet pigments of a flower (Hypericum calycinum). Proc Natl Acad Sci USA 98(24):13745–13750. https://doi.org/10.1073/pnas.231471698
Harlow BE, Flythe MD, Kagan IA, Goodman JP, Klotz JL, Aiken GE (2020) Isoflavone supplementation, via red clover hay, alters the rumen microbial community and promotes weight gain of steers grazing mixed grass pastures. PLoS ONE 15(3):e0229200. https://doi.org/10.1371/journal.pone.0229200
He X-Z, Dixon RA (2000) Genetic manipulation of isoflavone 7-O-methyltransferase enhances biosynthesis of 4′-O-methylated isoflavonoid phytoalexins and disease resistance in Alfalfa. Plant Cell 12(9):1689–1702. https://doi.org/10.1105/tpc.12.9.1689
Istvanek J, Jaros M, Krenek A, Repkova J (2014) Genome assembly and annotation for red clover (Trifolium pratense; Fabaceae). Am J Bot 101(2):327–337. https://doi.org/10.3732/ajb.1300340
Kim BG, Kim SY, Song HS, Lee C, Hur HG, Kim SI, Ahn JH (2003) Cloning and expression of the isoflavone synthase gene (IFS-Tp) from Trifolium pratense. Mol Cells 15(3):301–306
Kim JH, Nguyen NH, Jeong CY, Nguyen NT, Hong S-W, Lee H (2013) Loss of the R2R3 MYB, AtMyb73, causes hyper-induction of the SOS1 and SOS3 genes in response to high salinity in Arabidopsis. J Plant Physiol 170(16):1461–1465. https://doi.org/10.1016/j.jplph.2013.05.011
Knight DC, Eden JA (1995) Phytoestrogens—a short review. Maturitas 22(3):167–175. https://doi.org/10.1016/0378-5122(95)00937-g
Liu Y, Hassan S, Kidd BN, Garg G, Mathesius U, Singh KB, Anderson JP (2017) Ethylene signaling is important for isoflavonoid-mediated resistance torhizoctonia solaniin roots of Medicago truncatula. Mol Plant Microbe Interact 30(9):691–700. https://doi.org/10.1094/mpmi-03-17-0057-r
Marini H, Polito F, Adamo EB, Bitto A, Squadrito F, Benvenga S (2012) Update on genistein and thyroid: an overall message of safety. Front Endocrinol 3:94. https://doi.org/10.3389/fendo.2012.00094
Nottle MC (1976) Composition of some urinary calculi of ruminants in Western Australia. Res Vet Sci 21(3):309–317
Quesenberry KH, Smith RR (1993) Recurrent selection for plant regeneration from red clover tissue culture. Crop Sci 33(3):585–589. https://doi.org/10.2135/cropsci1993.0011183X003300030033x
Riday H, Krohn AL (2010) Genetic map-based location of the red clover (Trifolium pratense L.) gametophytic self-incompatibility locus. TAG Theor Appl Genet 121(4):761–767. https://doi.org/10.1007/s00122-010-1347-0
Sarkar MAR, Watanabe S, Suzuki A, Hashimoto F, Anai T (2019) Identification of novel MYB transcription factors involved in the isoflavone biosynthetic pathway by using the combination screening system with agroinfiltration and hairy root transformation. Plant Biotechnol 36(4):241–251. https://doi.org/10.5511/plantbiotechnology.19.1025a
Saviranta NMM, Anttonen MJ, von Wright A, Karjalainen RO (2008) Red clover (Trifolium pratense L.) isoflavones: determination of concentrations by plant stage, flower colour, plant part and cultivar. J Sci Food Agric 88:125–132. https://doi.org/10.1002/jsfa.3056
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez J-Y, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. https://doi.org/10.1038/nmeth.2019
Setchell KDR, Brown NM, Lydeking-Olsen E (2002) The clinical importance of the metabolite equol—a clue to the effectiveness of soy and its isoflavones. J Nutr 132(12):3577–3584. https://doi.org/10.1093/jn/132.12.3577
Smith RR, Quensenberry KH (1995) Registration of NEWRC red clover germplasm. Crop Sci 35(1):295–295. https://doi.org/10.2135/cropsci1995.0011183X003500010078x
Subramanian S, Graham MY, Yu O, Graham TL (2005) RNA interference of soybean isoflavone synthase genes leads to silencing in tissues distal to the transformation site and to enhanced susceptibility to Phytophthora sojae. Plant Physiol 137(4):1345–1353. https://doi.org/10.1104/pp.104.057257
Subramanian S, Stacey G, Yu O (2006) Endogenous isoflavones are essential for the establishment of symbiosis between soybean and Bradyrhizobium japonicum. Plant J 48(2):261–273
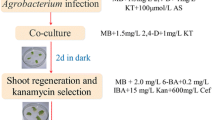
Sullivan ML, Quesenberry KH (2006) Red clover (Trifolium pratense). In: Wang K (ed) Methods in molecular biology: agrobacterium protocols, vol 343. Humana Press, Totowa, pp 369–384
Taylor N (2008) A century of clover breeding developments in the United States. Crop Sci 48:1–13. https://doi.org/10.2135/cropsci2007.08.0446
Tian T, Liu Y, Yan H, You Q, Yi X, Du Z, Xu W, Su Z (2017) agriGO v2.0: a GO analysis toolkit for the agricultural community, 2017 update. Nucleic Acids Res 45(W1):W122–W129. https://doi.org/10.1093/nar/gkx382
Townsend CE, Taylor NL (1985) Incompatibility and plant breeding. In: Taylor N (ed) Clover science and technology. ASA-CSSA-SSSA, Madison, WI, pp 365–381. https://doi.org/10.2134/agronmonogr25.c14
Tsanuo MK, Hassanali A, Hooper AM, Khan Z, Kaberia F, Pickett JA, Wadhams LJ (2003) Isoflavanones from the allelopathic aqueous root exudate of Desmodium uncinatum. Phytochemistry 64(1):265–273. https://doi.org/10.1016/s0031-9422(03)00324-8
Tsao R, Papadopoulos Y, Yang R, Young JC, McRae K (2006) Isoflavone profiles of red clovers and their distribution in different parts harvested at different growing stages. J Agric Food Chem 54(16):5797–5805. https://doi.org/10.1021/jf0614589
Xing HL, Dong L, Wang ZP, Zhang HY, Han CY, Liu B, Wang XC, Chen QJ (2014) A CRISPR/Cas9 toolkit for multiplex genome editing in plants. BMC Plant Biol 14:327. https://doi.org/10.1186/s12870-014-0327-y
Yang Y, Zhang L, Chen P, Liang T, Li X, Liu H (2019) UV-B photoreceptor UVR8 interacts with MYB73/MYB77 to regulate auxin responses and lateral root development. EMBO J 39(2):e101928. https://doi.org/10.15252/embj.2019101928
Yates SA, Swain MT, Hegarty MJ, Chernukin I, Lowe M, Allison GG, Ruttink T, Abberton MT, Jenkins G, Skot L (2014) De novo assembly of red clover transcriptome based on RNA-seq data provides insight into drought response, gene discovery and marker identification. BMC Genom 15:453. https://doi.org/10.1186/1471-2164-15-453
Yu O, Shi J, Hession AO, Maxwell CA, McGonigle B, Odell JT (2003) Metabolic engineering to increase isoflavone biosynthesis in soybean seed. Phytochemistry 63(7):753–763. https://doi.org/10.1016/s0031-9422(03)00345-5
Zhang J, Subramanian S, Stacey G, Yu O (2009) Flavones and flavonols play distinct critical roles during nodulation of Medicago truncatula by Sinorhizobium meliloti. Plant J 57(1):171–183. https://doi.org/10.1111/j.1365-313X.2008.03676.x
Acknowledgements
The authors wish to thank Dr. David McNair in the Plant and Soil Science Department at the University of Kentucky for advice and support on the root nodulation count procedures. This project was funded by the USDA-ARS Project no. 5042-21000-004-00D.
Author information
Authors and Affiliations
Contributions
RDD and HZ conceived and designed the experiments. JH, BLC, JBM, JPG, WTB, JL, YF and QZ performed the experiments and BLC, WTB and JL supervised the work. Data analysis was done by RDD, JBM, JPG and WTB. Manuscript was written by RDD, BLC, WTB and HZ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Additional information
Communicated by Baochun Li.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dinkins, R.D., Hancock, J., Coe, B.L. et al. Isoflavone levels, nodulation and gene expression profiles of a CRISPR/Cas9 deletion mutant in the isoflavone synthase gene of red clover. Plant Cell Rep 40, 517–528 (2021). https://doi.org/10.1007/s00299-020-02647-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02647-4