Abstract
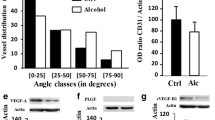
Ethanol consumption during pregnancy or lactation period can induce permanent damage to the development of the central nervous system (CNS), resulting in fetal alcohol spectrum disorders (FASD). CNS development depends on proper neural cells and blood vessel (BV) development and blood-brain barrier (BBB) establishment; however, little is known about how ethanol affects these events. Here, we investigated the impact of ethanol exposure to endothelial cells (ECs) function and to ECs interaction with astrocytes in the context of BBB establishment. Cerebral cortex of newborn mice exposed in utero to ethanol (FASD model) presented a hypervascularized phenotype, revealed by augmented vessel density, length, and branch points. Further, aberrant distribution of the tight junction ZO-1 protein along BVs and increased rates of perivascular astrocytic endfeet around BVs were observed. In vitro exposure of human brain microcapillary ECs (HBMEC) to ethanol significantly disrupted ZO-1 distribution, decreased Claudin-5 and GLUT-1 expression and impaired glucose uptake, and increased nitric oxide secretion. These events were accompanied by upregulation of angiogenesis-related secreted proteins by ECs in response to ethanol exposure. Treatment of cortical astrocytes with conditioned medium (CM) from ethanol exposed ECs, upregulated astrocyte’s expression of GFAP, Cx43, and Lipocalin-2 genes, as well as the pro-inflammatory genes, IL-1beta, IL-6, and TNF-alpha, which was accompanied by NF-kappa B protein nuclear accumulation. Our findings suggest that ethanol triggers a dysfunctional phenotype in brain ECs, leading to impairment of cortical vascular network formation, and promotes ECs-induced astrocyte dysfunction, which could dramatically affect BBB establishment in the developing brain.





Similar content being viewed by others
Data Availability
All data generated and analyzed during this study are available from the corresponding author upon reasonable request.
Abbreviations
- FASD:
-
Fetal alcohol spectrum disorder
- ECs:
-
Endothelial cells
- CNS:
-
Central nervous system
- ZO:
-
Zonula occludens
- CM:
-
Conditioned medium
- BBB:
-
Blood-brain barrier
- BV:
-
Blood vessel
- GLUT:
-
Glucose transporter
- HBMEC:
-
Human brain microcapillary endothelial cell
- MBEC:
-
Mouse brain endothelial cell
- EC-CM:
-
Endothelial cell-conditioned medium
- TiJOR:
-
Tight junction organization rate
- GFAP:
-
Glial fibrillary acidic protein
- MTT:
-
3- (4,5-dimethylthiazol-2-yl) -2,5-diphenyltetrazolium bromide
- 2-NBDG:
-
2-(N-(7-Nitrobenz-2-oxa-1,3- diazol-4-yl)amino)-2-deoxyglucose
References
De Guio F, Mangin JF, Riviere D et al (2014) A study of cortical morphology in children with fetal alcohol spectrum disorders. Hum Brain Mapp 35:2285–2296. https://doi.org/10.1002/hbm.22327
Lewis CE, Thomas KG, Dodge NC et al (2015) Verbal learning and memory impairment in children with fetal alcohol spectrum disorders. Alcohol Clin Exp Res 39:724–732. https://doi.org/10.1111/acer.12671
Hoyme HE, Kalberg WO, Elliott AJ, Blankenship J, Buckley D, Marais AS, Manning MA, Robinson LK et al (2016) Updated clinical guidelines for diagnosing fetal alcohol spectrum disorders. Pediatrics 138. https://doi.org/10.1542/peds.2015-4256
Walchli T, Wacker A, Frei K et al (2015) Wiring the vascular network with neural cues: a CNS perspective. Neuron 87:271–296. https://doi.org/10.1016/j.neuron.2015.06.038
Obermeier B, Daneman R, Ransohoff RM (2013) Development, maintenance and disruption of the blood-brain barrier. Nat Med 19:1584–1596. https://doi.org/10.1038/nm.3407
Wolburg H, Noell S, Mack A, Wolburg-Buchholz K, Fallier-Becker P (2009) Brain endothelial cells and the glio-vascular complex. Cell Tissue Res 335:75–96. https://doi.org/10.1007/s00441-008-0658-9
Abbott NJ, Patabendige AA, Dolman DE et al (2010) Structure and function of the blood-brain barrier. Neurobiol Dis 37:13–25. https://doi.org/10.1016/j.nbd.2009.07.030
McCarty JH (2005) Cell biology of the neurovascular unit: implications for drug delivery across the blood-brain barrier. Assay Drug Dev Technol 3:89–95. https://doi.org/10.1089/adt.2005.3.89
Dick AP, Harik SI, Klip A, Walker DM (1984) Identification and characterization of the glucose transporter of the blood-brain barrier by cytochalasin B binding and immunological reactivity. Proc Natl Acad Sci U S A 81:7233–7237. https://doi.org/10.1073/pnas.81.22.7233
Mathiisen TM, Lehre KP, Danbolt NC, Ottersen OP (2010) The perivascular astroglial sheath provides a complete covering of the brain microvessels: an electron microscopic 3D reconstruction. Glia 58:1094–1103. https://doi.org/10.1002/glia.20990
Alvarez JI, Katayama T, Prat A (2013) Glial influence on the blood brain barrier. Glia 61:1939–1958. https://doi.org/10.1002/glia.22575
Cambier S, Gline S, Mu D, Collins R, Araya J, Dolganov G, Einheber S, Boudreau N et al (2005) Integrin alpha(v)beta8-mediated activation of transforming growth factor-beta by perivascular astrocytes: an angiogenic control switch. Am J Pathol 166:1883–1894
Diniz LP, Matias I, Siqueira M, Stipursky J, Gomes FCA (2019) Astrocytes and the TGF-beta1 pathway in the healthy and diseased brain: a double-edged sword. Mol Neurobiol 56:4653–4679. https://doi.org/10.1007/s12035-018-1396-y
Mi H, Haeberle H, Barres BA (2001) Induction of astrocyte differentiation by endothelial cells. J Neurosci 21:1538–1547
Solonskii AV, Logvinov SV, Kutepova NA (2008) Development of brain vessels in human embryos and fetuses in conditions of prenatal exposure to alcohol. Neurosci Behav Physiol 38:373–376. https://doi.org/10.1007/s11055-008-0053-8
Welch JH, Mayfield JJ, Leibowitz AL, Baculis BC, Valenzuela CF (2016) Third trimester-equivalent ethanol exposure causes micro-hemorrhages in the rat brain. Neuroscience 324:107–118. https://doi.org/10.1016/j.neuroscience.2016.03.004
Haorah J, Heilman D, Knipe B, Chrastil J, Leibhart J, Ghorpade A, Miller DW, Persidsky Y (2005) Ethanol-induced activation of myosin light chain kinase leads to dysfunction of tight junctions and blood-brain barrier compromise. Alcohol Clin Exp Res 29:999–1009
Haorah J, Knipe B, Leibhart J et al (2005) Alcohol-induced oxidative stress in brain endothelial cells causes blood-brain barrier dysfunction. J Leukoc Biol 78:1223–1232. https://doi.org/10.1189/jlb.0605340
Topper LA, Baculis BC, Valenzuela CF (2015) Exposure of neonatal rats to alcohol has differential effects on neuroinflammation and neuronal survival in the cerebellum and hippocampus. J Neuroinflammation 12:160. https://doi.org/10.1186/s12974-015-0382-9
Wilhelm CJ, Guizzetti M (2015) Fetal alcohol Spectrum disorders: an overview from the glia perspective. Front Integr Neurosci 9:65. https://doi.org/10.3389/fnint.2015.00065
Goodlett CR, Leo JT, O’Callaghan JP et al (1993) Transient cortical astrogliosis induced by alcohol exposure during the neonatal brain growth spurt in rats. Brain Res Dev Brain Res 72:85–97
Guizzetti M, Zhang X, Goeke C, Gavin DP (2014) Glia and neurodevelopment: focus on fetal alcohol spectrum disorders. Front Pediatr 2:123. https://doi.org/10.3389/fped.2014.00123
Trindade P, Hampton B, Manhaes AC, Medina AE (2016) Developmental alcohol exposure leads to a persistent change on astrocyte secretome. J Neurochem 137:730–743. https://doi.org/10.1111/jnc.13542
Guerri C, Pascual M, Renau-Piqueras J (2001) Glia and fetal alcohol syndrome. Neurotoxicology 22:593–599
Abbott NJ, Ronnback L, Hansson E (2006) Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 7:41–53. https://doi.org/10.1038/nrn1824
Silva Siqueira MD, Gisbert DC, Gomes FCA, Stipursky J (2019) Radial glia-endothelial cells bidirectional interactions control vascular maturation and astrocyte differentiation: Impact for blood brain barrier formation. Curr Neurovasc Res 16:291–300. https://doi.org/10.2174/1567202616666191014120156
Jegou S, El Ghazi F, de Lendeu PK et al (2012) Prenatal alcohol exposure affects vasculature development in the neonatal brain. Ann Neurol 72:952–960. https://doi.org/10.1002/ana.23699
Downing C, Balderrama-Durbin C, Hayes J, Johnson TE, Gilliam D (2009) No effect of prenatal alcohol exposure on activity in three inbred strains of mice. Alcohol Alcohol 44:25–33. https://doi.org/10.1093/alcalc/agn082
Siqueira M, Francis D, Gisbert D, Gomes FCA, Stipursky J (2017) Radial glia cells control angiogenesis in the developing cerebral cortex through TGF-beta1 signaling. Mol Neurobiol. https://doi.org/10.1007/s12035-017-0557-8
Diniz LP, Almeida JC, Tortelli V, Vargas Lopes C, Setti-Perdigão P, Stipursky J, Kahn SA, Romão LF et al (2012) Astrocyte-induced synaptogenesis is mediated by transforming growth factor beta signaling through modulation of D-serine levels in cerebral cortex neurons. J Biol Chem 287:41432–41445. https://doi.org/10.1074/jbc.M112.380824
Adachi J, Mizoi Y, Fukunaga T et al (1991) Degrees of alcohol intoxication in 117 hospitalized cases. J Stud Alcohol 52:448–453. https://doi.org/10.15288/jsa.1991.52.448
Deitrich RA, Harris RA (1996) How much alcohol should I use in my experiments? Alcohol Clin Exp Res 20:1–2. https://doi.org/10.1111/j.1530-0277.1996.tb01033.x
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126:131–138
Noursadeghi M, Tsang J, Haustein T, Miller RF, Chain BM, Katz DR (2008) Quantitative imaging assay for NF-κB nuclear translocation in primary human macrophages. J Immunol Methods 329:194–200. https://doi.org/10.1016/j.jim.2007.10.015
Zudaire E, Gambardella L, Kurcz C, Vermeren S (2011) A computational tool for quantitative analysis of vascular networks. PLoS One 6:e27385. https://doi.org/10.1371/journal.pone.0027385
Yu X, Zhao R, Lin S et al (2016) CXCL16 induces angiogenesis in autocrine signaling pathway involving hypoxia-inducible factor 1alpha in human umbilical vein endothelial cells. Oncol Rep 35:1557–1565. https://doi.org/10.3892/or.2015.4520
Terryn C, Sellami M, Fichel C, Diebold MD, Gangloff S, le Naour R, Polette M, Zahm JM (2013) Rapid method of quantification of tight-junction organization using image analysis. Cytometry A 83:235–241. https://doi.org/10.1002/cyto.a.22239
Abdul Muneer PM, Alikunju S, Szlachetka AM, Haorah J (2011) Inhibitory effects of alcohol on glucose transport across the blood-brain barrier leads to neurodegeneration: preventive role of acetyl-L: -carnitine. Psychopharmacology 214:707–718. https://doi.org/10.1007/s00213-010-2076-4
Parathath SR, Parathath S, Tsirka SE (2006) Nitric oxide mediates neurodegeneration and breakdown of the blood-brain barrier in tPA-dependent excitotoxic injury in mice. J Cell Sci 119:339–349. https://doi.org/10.1242/jcs.02734
Hanahan D, Folkman J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86:353–364. https://doi.org/10.1016/s0092-8674(00)80108-7
Boulay AC, Saubamea B, Adam N et al (2017) Translation in astrocyte distal processes sets molecular heterogeneity at the gliovascular interface. Cell Discov 3:17005. https://doi.org/10.1038/celldisc.2017.5
Sofroniew MV (2015) Astrocyte barriers to neurotoxic inflammation. Nat Rev Neurosci 16:249–263. https://doi.org/10.1038/nrn3898
Lau LT, Yu AC (2001) Astrocytes produce and release interleukin-1, interleukin-6, tumor necrosis factor alpha and interferon-gamma following traumatic and metabolic injury. J Neurotrauma 18:351–359. https://doi.org/10.1089/08977150151071035
Argaw AT, Zhang Y, Snyder BJ, Zhao ML, Kopp N, Lee SC, Raine CS, Brosnan CF et al (2006) IL-1beta regulates blood-brain barrier permeability via reactivation of the hypoxia-angiogenesis program. J Immunol 177:5574–5584. https://doi.org/10.4049/jimmunol.177.8.5574
Li K, Li J, Zheng J, Qin S (2019) Reactive astrocytes in neurodegenerative diseases. Aging Dis 10:664–675. https://doi.org/10.14336/AD.2018.0720
Mohr T, Haudek-Prinz V, Slany A, Grillari J, Micksche M, Gerner C (2017) Proteome profiling in IL-1beta and VEGF-activated human umbilical vein endothelial cells delineates the interlink between inflammation and angiogenesis. PLoS One 12:e0179065. https://doi.org/10.1371/journal.pone.0179065
Tata M, Ruhrberg C, Fantin A (2015) Vascularisation of the central nervous system. Mech Dev 138(Pt 1):26–36. https://doi.org/10.1016/j.mod.2015.07.001
Paredes I, Himmels P, Ruiz de Almodovar C (2018) Neurovascular communication during CNS development. Dev Cell 45:10–32. https://doi.org/10.1016/j.devcel.2018.01.023
Garcez PP, Stolp HB, Sravanam S, Christoff RR, Ferreira JCCG, Dias AA, Pezzuto P, Higa LM et al (2018) Zika virus impairs the development of blood vessels in a mouse model of congenital infection. Sci Rep 8:12774. https://doi.org/10.1038/s41598-018-31149-3
Bennett RE, Robbins AB, Hu M, Cao X, Betensky RA, Clark T, Das S, Hyman BT (2018) Tau induces blood vessel abnormalities and angiogenesis-related gene expression in P301L transgenic mice and human Alzheimer’s disease. Proc Natl Acad Sci U S A 115:E1289–E1298. https://doi.org/10.1073/pnas.1710329115
Estato V, Stipursky J, Gomes F, Mergener TC, Frazão-Teixeira E, Allodi S, Tibiriçá E, Barbosa HS et al (2018) The neurotropic parasite Toxoplasma gondii induces sustained neuroinflammation with microvascular dysfunction in infected mice. Am J Pathol 188:2674–2687. https://doi.org/10.1016/j.ajpath.2018.07.007
Tornavaca O, Chia M, Dufton N, Almagro LO, Conway DE, Randi AM, Schwartz MA, Matter K et al (2015) ZO-1 controls endothelial adherens junctions, cell-cell tension, angiogenesis, and barrier formation. J Cell Biol 208:821–838. https://doi.org/10.1083/jcb.201404140
Guillemot L, Paschoud S, Pulimeno P, Foglia A, Citi S (2007) The cytoplasmic plaque of tight junctions: a scaffolding and signalling center. Biochim Biophys Acta Biomembr 1778:601–613
George AK, Behera J, Kelly KE, Mondal NK, Richardson KP, Tyagi N (2018) Exercise mitigates alcohol induced endoplasmic reticulum stress mediated cognitive impairment through ATF6-Herp signaling. Sci Rep 8:5158. https://doi.org/10.1038/s41598-018-23568-z
Haorah J, Schall K, Ramirez SH, Persidsky Y (2008) Activation of protein tyrosine kinases and matrix metalloproteinases causes blood-brain barrier injury: novel mechanism for neurodegeneration associated with alcohol abuse. Glia 56:78–88. https://doi.org/10.1002/glia.20596
Yu H, Wang C, Wang X, Wang H, Zhang C, You J, Wang P, Feng C et al (2017) Long-term exposure to ethanol downregulates tight junction proteins through the protein kinase C alpha signaling pathway in human cerebral microvascular endothelial cells. Exp Ther Med 14:4789–4796. https://doi.org/10.3892/etm.2017.5180
Katsuno T, Umeda K, Matsui T, Hata M, Tamura A, Itoh M, Takeuchi K, Fujimori T et al (2008) Deficiency of zonula occludens-1 causes embryonic lethal phenotype associated with defected yolk sac angiogenesis and apoptosis of embryonic cells. Mol Biol Cell 19:2465–2475. https://doi.org/10.1091/mbc.E07-12-1215
Zimatkin SM, Pronko SP, Vasiliou V, Gonzalez FJ, Deitrich RA (2006) Enzymatic mechanisms of ethanol oxidation in the brain. Alcohol Clin Exp Res 30:1500–1505. https://doi.org/10.1111/j.1530-0277.2006.00181.x
Han J, Shuvaev VV, Muzykantov VR (2011) Catalase and superoxide dismutase conjugated with platelet-endothelial cell adhesion molecule antibody distinctly alleviate abnormal endothelial permeability caused by exogenous reactive oxygen species and vascular endothelial growth factor. J Pharmacol Exp Ther 338:82–91. https://doi.org/10.1124/jpet.111.180620
Haorah J, Ramirez SH, Schall K, Smith D, Pandya R, Persidsky Y (2007) Oxidative stress activates protein tyrosine kinase and matrix metalloproteinases leading to blood-brain barrier dysfunction. J Neurochem 101:566–576. https://doi.org/10.1111/j.1471-4159.2006.04393.x
Drever N, Yin H, Kechichian T et al (2012) The expression of antioxidant enzymes in a mouse model of fetal alcohol syndrome. Am J Obstet Gynecol 206:358.e19–358.e22. https://doi.org/10.1016/j.ajog.2012.01.017
Dermietzel R, Krause D, Kremer M, Wang C, Stevenson B (1992) Pattern of glucose transporter (Glut 1) expression in embryonic brains is related to maturation of blood-brain barrier tightness. Dev Dyn 193:152–163. https://doi.org/10.1002/aja.1001930207
Veys K, Fan Z, Ghobrial M, Bouché A, García-Caballero M, Vriens K, Conchinha NV, Seuwen A et al (2020) Role of the GLUT1 glucose transporter in postnatal CNS angiogenesis and blood-brain barrier integrity. Circ Res 127:466–482. https://doi.org/10.1161/CIRCRESAHA.119.316463
Zhuge X, Murayama T, Arai H, Yamauchi R, Tanaka M, Shimaoka T, Yonehara S, Kume N et al (2005) CXCL16 is a novel angiogenic factor for human umbilical vein endothelial cells. Biochem Biophys Res Commun 331:1295–1300. https://doi.org/10.1016/j.bbrc.2005.03.200
Rosito M, Deflorio C, Limatola C, Trettel F (2012) CXCL16 orchestrates adenosine A3 receptor and MCP-1/CCL2 activity to protect neurons from excitotoxic cell death in the CNS. J Neurosci 32:3154–3163. https://doi.org/10.1523/JNEUROSCI.4046-11.2012
Sokolowski JD, Chabanon-Hicks CN, Han CZ et al (2014) Fractalkine is a “find-me” signal released by neurons undergoing ethanol-induced apoptosis. Front Cell Neurosci 8. https://doi.org/10.3389/fncel.2014.00360
Mandal C, Halder D, Jung KH, Chai YG (2017) Gestational alcohol exposure altered DNA methylation status in the developing fetus. Int J Mol Sci 18. https://doi.org/10.3390/ijms18071386
Basavarajappa BS, Subbanna S (2016) Epigenetic mechanisms in developmental alcohol-induced neurobehavioral deficits. Brain Sci 6. https://doi.org/10.3390/brainsci6020012
Tomas M, Marin P, Megias L et al (2005) Ethanol perturbs the secretory pathway in astrocytes. Neurobiol Dis 20:773–784. https://doi.org/10.1016/j.nbd.2005.05.012
Hubbard JA, Hsu MS, Seldin MM, Binder DK (2015) Expression of the astrocyte water channel Aquaporin-4 in the mouse brain. ASN Neuro 7:175909141560548. https://doi.org/10.1177/1759091415605486
Ezan P, Andre P, Cisternino S et al (2012) Deletion of astroglial connexins weakens the blood-brain barrier. J Cereb Blood Flow Metab 32:1457–1467. https://doi.org/10.1038/jcbfm.2012.45
Bi F, Huang C, Tong J, Qiu G, Huang B, Wu Q, Li F, Xu Z et al (2013) Reactive astrocytes secrete lcn2 to promote neuron death. Proc Natl Acad Sci U S A 110:4069–4074. https://doi.org/10.1073/pnas.1218497110
Kim JH, Ko PW, Lee HW, Jeong JY, Lee MG, Kim JH, Lee WH, Yu R et al (2017) Astrocyte-derived lipocalin-2 mediates hippocampal damage and cognitive deficits in experimental models of vascular dementia. Glia 65:1471–1490. https://doi.org/10.1002/glia.23174
Mooney SM, Miller MW (2003) Ethanol-induced neuronal death in organotypic cultures of rat cerebral cortex. Brain Res Dev Brain Res 147:135–141
Sofroniew MV (2009) Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci 32:638–647. https://doi.org/10.1016/j.tins.2009.08.002
Liu T, Zhang L, Joo D, Sun SC (2017) NF-kappaB signaling in inflammation. Signal Transduct Target Ther 2. https://doi.org/10.1038/sigtrans.2017.23
Liu S, Agalliu D, Yu C, Fisher M (2012) The role of pericytes in blood-brain barrier function and stroke. Curr Pharm Des 18:3653–3662. https://doi.org/10.2174/138161212802002706
Acknowledgments
We thank Dr. Ana Paula Cabral de Araújo Lima (Universidade Federal do Rio de Janeiro, Instituto de Biofísica Carlos Chagas Filho, Brazil) that with the consent of Dr. Dennis Grab (Associate Professor, Johns Hopkins University, Department of Pathology, Baltimore, USA) provided HBMEC cell line. We also thank Marcelo Meloni, Carolina Moraes, and Grasiela Ventura for technical assistance.
Funding
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (MS, JS, FG); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (MS, JS, FCAG); Departamento de Ciência e Tecnologia do Ministério da Saúde (Decit) (APBA); Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) (MS, JS, FCAG); and International Society for Neurochemistry (ISN) (JS).
Author information
Authors and Affiliations
Contributions
Conceptualization, JS; methodology, JS and MS; formal analysis and investigation, APBA and MS; visualization, MS; writing (original draft), MS; writing (review and editing), FCAG and JS; funding acquisition, FCAG and JS; resources, FCAG and JS; project administration, JS; supervision, JS
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
All animal protocols were approved by the Animal Use Ethics Committee of the Federal University of Rio de Janeiro (protocol n°. CEUA 067/16).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary figure 1
Treatment of astrocytes with ethanol modulates BBB and pro-inflammatory related genes in a less extent than ECs derived factors. Confluent cortical astrocytes cultures were treated with control medium (Control) or ethanol for 24 h, followed by RT-qPCR analysis. Ethanol significatively enhanced Aqp4, Cx43, GFAP and TNF-α and did not modulate Lcn2, IL-1β and IL-6 gene expression (a-b). Immunocytochemistry assays revealed that ethanol exposure did not induce NF-κB nuclear accumulation in astrocyte cultures (b). Binary masks of nuclear and cytoplasmic labeling intensities of NF-κB ROI (g-j). Data are expressed as Mean ± SEM (n = 3–4 control and ethanol-treated) independent astrocyte cultures. Scale bar: 20 μm (c-j). (PNG 460 kb)
Rights and permissions
About this article
Cite this article
Siqueira, M., Araujo, A.P.B., Gomes, F.C.A. et al. Ethanol Gestational Exposure Impairs Vascular Development and Endothelial Potential to Control BBB-Associated Astrocyte Function in the Developing Cerebral Cortex. Mol Neurobiol 58, 1755–1768 (2021). https://doi.org/10.1007/s12035-020-02214-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-02214-8