Abstract
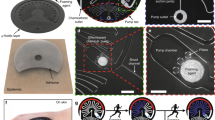
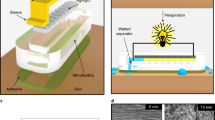
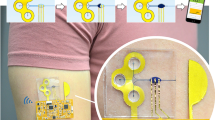
Skin-compatible microfluidic valving systems with on-demand sweat capture are necessary to understand the temporal variation of biomarkers. Here, we demonstrate solution-based electrowetting valves with rapid actuation integrated into a flexible microfluidic sweat collection patch. The valve is produced by inkjet-printing a pair of silver electrodes with spacings of 0.2–2 mm and modifying the downstream electrode with a hydrophobic self-assembled monolayer. To complete the valve, a microfluidic channel is fabricated from laser ablation of adhesive layers and pressed over the silver electrodes. Artificial perspiration is driven by capillary action within the channel until stopped by the electrowetting valve. A low voltage is applied to the electrodes, decreasing the surface energy of the hydrophobic monolayer and allowing the fluid front to continue through the microchannel. Statistical analysis demonstrated that applied voltage, and not electrode spacing, influenced valve actuation time, with 17 ± 8 s for 4 V and 40 ± 16 s for 1 V. Inkjet-printing conditions were also optimized to achieve a valve fluid retention time of 9 h. Using four electrowetting valves, an integrated wearable device is designed for artificial perspiration collection through valve actuation at distinct time points over 40 min. Finally, these inexpensive, user-friendly, and disposable electrowetting valves offer exciting opportunities for non-invasive point-of-care sweat monitoring.






Similar content being viewed by others
References
Alizadeh A, Burns A, Lenigk R et al (2018) A wearable patch for continuous monitoring of sweat electrolytes during exertion. Lab Chip 18:2632–2641. https://doi.org/10.1039/C8LC00510A
Anastasova S, Crewther B, Bembnowicz P et al (2017) A wearable multisensing patch for continuous sweat monitoring. Biosens Bioelectron 93:139–145. https://doi.org/10.1016/j.bios.2016.09.038
Atabakhsh S, Jafarabadi Ashtiani S (2018) Thermal actuation and confinement of water droplets on paper-based digital microfluidics devices. Microfluid Nanofluidics 22:1–9. https://doi.org/10.1007/s10404-018-2060-6
Baker LB (2017) Sweating rate and sweat sodium concentration in athletes : a review of methodology and intra/interindividual variability. Sport Med 47:111–128. https://doi.org/10.1007/s40279-017-0691-5
Bandodkar AJ, Gutruf P, Choi J et al (2019) Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci Adv 5:1–16. https://doi.org/10.1126/sciadv.aav3294
Bandodkar AJ, Wang J (2014) Non-invasive wearable electrochemical sensors: a review. Trends Biotechnol 32:363–371. https://doi.org/10.1016/j.tibtech.2014.04.005
Bariya M, Nyein HYY, Javey A (2018) Wearable sweat sensors. Nat Electron 1:160–171
Chandekar A, Sengupta SK, Whitten JE (2010) Thermal stability of thiol and silane monolayers: a comparative study. Appl Surf Sci 256:2742–2749. https://doi.org/10.1016/j.apsusc.2009.11.020
Chen J, Zhou Y, Wang D et al (2015) UV-nanoimprint lithography as a tool to develop flexible microfluidic devices for electrochemical detection. Lab Chip 15:3086–3094. https://doi.org/10.1039/C5LC00515A
Choi J, Bandodkar AJ, Reeder JT et al (2019) Soft, skin-integrated multifunctional microfluidic systems for accurate colorimetric analysis of sweat biomarkers and temperature. ACS Sensors 4:379–388. https://doi.org/10.1021/acssensors.8b01218
Coyle S, Morris D, Lau KT, et al (2009) Textile sensors to measure sweat pH and sweat-rate during exercise. In: Pervasive Comput Technol Heal 2009 Pervasive Health 2009 3rd Interntional Conference 4–9. https://doi.org/https://doi.org/10.4108/ICST.PERVASIVEHEALTH2009.5957
Di QT, Zhang H, Liang XL, et al (2015) Electrowetting display pixels fabricated by nanoimprint lithography. Int Conf Appl Sci Eng Innov 495–501
Gao W, Emaminejad S, Nyein HYY et al (2016) Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529:509–514. https://doi.org/10.1038/nature16521
Guo ZH, Jiao YC, Wang HL et al (2019) Self-powered electrowetting valve for instantaneous and simultaneous actuation of paper-based microfluidic assays. Adv Funct Mater 29:1–7. https://doi.org/10.1002/adfm.201808974
Haynes W (2012) CRC handbook of chemistry and physics (92 ed). CRC Press, New York, pp 5–80
He F, Grimes J, Alcaine SD, Nugen SR (2014) A hybrid paper and microfluidic chip with electrowetting valves and colorimetric detection. Analyst 139:3002–3008. https://doi.org/10.1039/c3an01516e
He F, Nugen SR (2014) Automating fluid delivery in a capillary microfluidic device using low-voltage electrowetting valves. Microfluid Nanofluidics 16:879–886. https://doi.org/10.1007/s10404-013-1317-3
Heikenfeld J (2016) Non-invasive analyte access and sensing through eccrine sweat: challenges and outlook circa 2016. Electroanalysis. https://doi.org/10.1002/elan.201600018
Hendriks BHW, Kuiper S, Van As MAJ et al (2005) Electrowetting-based variable-focus lens for miniature systems. Opt Rev 12:255–259. https://doi.org/10.1007/s10043-005-0255-z
Jadoon S, Karim S, Akram MR et al (2015) Recent developments in sweat analysis and its applications. Int J Anal Chem 2015:1–7. https://doi.org/10.1155/2015/164974
Kim SB, Koo J, Yoon J et al (2020) Soft, skin-interfaced microfluidic systems with integrated enzymatic assays for measuring the concentration of ammonia and ethanol in sweat. Lab Chip 20:84–92. https://doi.org/10.1039/c9lc01045a
Koh A, Kang D, Xue Y et al (2016) A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci Transl Med 8:1–14. https://doi.org/10.1126/scitranslmed.aaf2593
Koo CKW, He F, Nugen SR (2013) An inkjet-printed electrowetting valve for paper-fluidic sensors. Analyst 138:4998–5004. https://doi.org/10.1039/c3an01114c
Liu H, Dharmatilleke S, Maurya DK, Tay AAO (2010) Dielectric materials for electrowetting-on-dielectric actuation. Microsyst Technol 16:449–460. https://doi.org/10.1007/s00542-009-0933-z
Love JC, Estroff LA, Kriebel JK et al (2005) Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem Inform. https://doi.org/10.1002/chin.200532281
Mach P, Krupenkin T, Yang S, Rogers JA (2002) Dynamic tuning of optical waveguides with electrowetting pumps and recirculating fluid channels. Appl Phys Lett 81:202–204. https://doi.org/10.1063/1.1491608
Merian T, He F, Yan H et al (2012) Development and surface characterization of an electrowetting valve for capillary-driven microfluidics. Colloids Surf Physicochem Eng Asp 414:251–258. https://doi.org/10.1016/j.colsurfa.2012.08.020
Moon I, Kim J (2006) Using EWOD (electrowetting-on-dielectric) actuation in a micro conveyor system. Sens Actuators A 130–131:537–544. https://doi.org/10.1016/j.sna.2005.12.016
Mugele F, Baret J-C (2005) Electrowetting: from basics to applications. J Phys Condens Matter 17:R705–R774. https://doi.org/10.1088/0953-8984/17/28/R01
Quilliet C, Berge B (2001) Electrowetting: a recent outbreak. Colloid Interface Sci 6:34–39. https://doi.org/10.1016/S1359-0294(00)00085-6
Rossmeisl J, Logadottir A, Nørskov JK (2005) Electrolysis of water on (oxidized) metal surfaces. Chem Phys 319:178–184. https://doi.org/10.1016/j.chemphys.2005.05.038
Seshadri DR, Li RT, Voos JE et al (2019) Wearable sensors for monitoring the physiological and biochemical profile of the athlete. NPJ Digit Med. https://doi.org/10.1038/s41746-019-0150-9
Shamai R, Andelman D, Berge B, Hayes R (2008) Water, electricity, and between… On electrowetting and its applications. Soft Matter 4:38–45. https://doi.org/10.1039/b714994h
Tohgha UN, Alvino EL, Jarnagin CC et al (2019) Electrowetting behavior and digital microfluidic applications of fluorescent, polymer-encapsulated quantum dot nanofluids. ACS Appl Mater Interfaces 11:28487–28498. https://doi.org/10.1021/acsami.9b07983
Torabinia M, Asgari P, Dakarapu US et al (2019) On-chip organic synthesis enabled using an engine-and-cargo system in an electrowetting-on-dielectric digital microfluidic device. Lab Chip 19:3054–3064. https://doi.org/10.1039/c9lc00428a
Zhang Y, Guo H, Kim SB et al (2019) Passive sweat collection and colorimetric analysis of biomarkers relevant to kidney disorders using a soft microfluidic system. Lab Chip 19:1545–1555. https://doi.org/10.1039/c9lc00103d
Acknowledgements
We would like to thank Gregory Larson for assistance with the ANOVA test and Xiyu Hu for performing the adhesion tape test. This research was funded by the Nano-Bio Manufacturing Consortium (NBMC), NextFlex, the Air Force Research Laboratory (AFRL), and US Department of Defense (DoD) (FA86500 13273 11-12). A.R.N, B.W, and J.J.W. acknowledge funding from the National Science Foundation (NSF) Center for Hierarchical Manufacturing at the University of Massachusetts at Amherst (CMMI-1025020). The U.S. Government is authorized to reproduce and distribute reprints for governmental purposes notwithstanding any copyright notation thereon. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements, either expressed or implied, of the sponsors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Naik, A.R., Warren, B., Burns, A. et al. Electrowetting valves for sweat-based microfluidics. Microfluid Nanofluid 25, 2 (2021). https://doi.org/10.1007/s10404-020-02403-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02403-w