Abstract
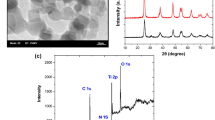
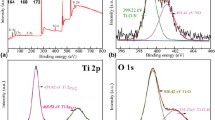
The hydrothermal method has been used to prepare Ni(OH)2 photocathode. The photoelectrochemical (PEC) reduction of CO2 over Ni(OH)2 has been conducted in 0.2 M LiClO4 in aqueous and N,N-dimethylformamide (DMF) medium under visible light irradiation. The thin film was characterized by XRD, UV–Vis, FTIR, FESEM-EDX, BET analysis, and electrochemical method for the determination of phases, bandgap energy, chemical bonding, surface morphology, elemental compositions, surface area, and electrochemical properties, respectively. Based on UV–Vis spectroscopy, the bandgap energy of Ni(OH)2 was 1.8 eV which enabled efficient visible light absorption for the photoreaction. The photocurrent density in aqueous and DMF solution at 0.2 V (vs. Ag/AgCl) was 24 mA cm−2 and 5 mA cm−2, respectively. Acetaldehyde and methanol are the products in aqueous solution, while formic acid and methanol are the products in DMF, after 6 h of photoelectrolysis. The product formations from the photoelectrochemical reduction of dissolved CO2 were 612 and 854 ppm in aqueous and DMF, respectively, where the Faradaic efficiency in aqueous and DMF is 24 and 33%, respectively. Furthermore, throughout the PEC study, the transformation of Ni(OH)2 to NiO plays a significant role in the formation of organic oxygenates from the reduction reaction of CO2.
Graphic abstract







Similar content being viewed by others
References
Z. He, J. Tang, J. Shen, J. Chen, S. Song, Enhancement of photocatalytic reduction of CO2 to CH4 over TiO2 nanosheets by modifying with sulfuric acid. Appl. Surf. Sci. 364, 416–427 (2016)
Q. Zhang, J. Du, A. He, Z. Liu, C. Tao, Low overpotential electrochemical CO2 reduction to formate on Co3O4–CeO2/low graphitic carbon catalyst with oxygen vacancies. J. Solid State Chem. 279, 120946–120954 (2019)
E. Szaniawska, K. Bienkowski, I.A. Rutkowska, P.J. Kulesza, R. Solarska, Enhanced photoelectrochemical CO2-reduction system based on mixed Cu2O–nonstoichiometric TiO2 photocathode. Catal. Today 300, 145–151 (2018)
N.T.T. Truc, L.G. Bach, N.T. Hanh, T.-D. Pham, N.T.P. Le Chi, D.T. Tran, M.V. Nguyen, V.N. Nguyen, The superior photocatalytic activity of Nb doped TiO2/g-C3N4 direct Z-scheme system for efficient conversion of CO2 into valuable fuels. J. Colloid Interface Sci. 540, 1–8 (2019)
L. Yu, X. Ba, M. Qiu, Y. Li, L. Shuai, W. Zhang, Z. Ren, Y. Yu, Visible-light driven CO2 reduction coupled with water oxidation on Cl-doped Cu2O nanorods. Nano Energy 60, 576–582 (2019)
D.H. Apaydin, E. Tordin, E. Portenkirchner, G. Aufischer, S. Schlager, M. Weichselbaumer, K. Oppelt, N.S. Sariciftci, Photoelectrochemical Reduction of CO2 Using Third-Generation Conjugated Polymers. ChemistrySelect 1, 1156–1162 (2016)
S. Kaneco, H. Katsumata, T. Suzuki, K. Ohta, Electrochemical reduction of CO2 to methane at the Cu electrode in methanol with sodium supporting salts and its comparison with other alkaline salts. Energy Fuels 20, 409–414 (2006)
J. Qu, X. Zhang, Y. Wang, C. Xie, Electrochemical reduction of CO2 on RuO2/TiO2 nanotubes composite modified Pt electrode. Electrochim. Acta 50, 3576–3580 (2005)
N. Furuya, S. Koide, Electroreduction of carbon dioxide by metal phthalocyanines. Electrochim. Acta 36, 1309–1313 (1991)
A.A. Peterson, J.K. Nørskov, Activity descriptors for CO2 electroreduction to methane on transition-metal catalysts. J. Phys. Chem. Lett. 3, 251–258 (2012)
T. Yamamoto, D.A. Tryk, A. Fujishima, H. Ohata, Production of syngas plus oxygen from CO2 in a gas-diffusion electrode-based electrolytic cell. Electrochim. Acta 47, 3327–3334 (2002)
Y. Zhang, L. Sun, K. Lv, Y. Zhang, One-pot synthesis of Ni(OH)2 flakes embedded in highly-conductive carbon nanotube/graphene hybrid framework as high performance electrodes for supercapacitors. Mater. Lett. 213, 131–134 (2018)
H. Wu, Y. Li, J. Ren, D. Rao, Q. Zheng, L. Zhou, D. Lin, CNT-assembled dodecahedra core@ nickel hydroxide nanosheet shell enabled sulfur cathode for high-performance lithium-sulfur batteries. Nano Energy 55, 82–92 (2019)
V. Ganesh, S. Farzana, S. Berchmans, Nickel hydroxide deposited indium tin oxide electrodes as electrocatalysts for direct oxidation of carbohydrates in alkaline medium. J. Power Sources 196, 9890–9899 (2011)
S. Ramesh, K. Karuppasamy, H.M. Yadav, J.-J. Lee, H.-S. Kim, H.-S. Kim, J.-H. Kim, Ni(OH)2-decorated nitrogen doped MWCNT nanosheets as an efficient electrode for high performance supercapacitors. Sci. Rep. 9, 1–10 (2019)
X. Zhou, Z. Xia, Z. Zhang, Y. Ma, Y. Qu, One-step synthesis of multi-walled carbon nanotubes/ultra-thin Ni(OH)2 nanoplate composite as efficient catalysts for water oxidation. J. Mater. Chem. A 2, 11799–11806 (2014)
H. Zhou, J. Peng, X. Qiu, Y. Gao, L. Lu, W. Wang, β-Ni(OH)2 nanosheets: an effective sensing platform for constructing nucleic acid-based optical sensors. J. Mater. Chem. B 5, 7426–7432 (2017)
Q. Xu, H. Jiang, H. Zhang, Y. Hu, C. Li, Heterogeneous interface engineered atomic configuration on ultrathin Ni(OH)2/Ni3S2 nanoforests for efficient water splitting. Appl. Catal. B 242, 60–66 (2019)
I. Vamvasakis, I.T. Papadas, T. Tzanoudakis, C. Drivas, S.A. Choulis, S. Kennou, G.S. Armatas, Visible-light photocatalytic H2 production activity of β-Ni(OH)2-modified CdS mesoporous nanoheterojunction networks. ACS Catal. 8, 8726–8738 (2018)
N.I.M. Abdallah, Y. Li, X.-T. Wang, X. Li, C.-W. Wang, Design and fabrication of Ni(OH)2/BiVO4 heterostructured nano-photocatalyst for high-efficient removal of organic dyes, J. Alloys Compd., (2020) 154828-154836
M. Barakat, M. Anjum, R. Kumar, Z. Alafif, M. Oves, M.O. Ansari, Design of ternary Ni(OH)2/graphene oxide/TiO2 nanocomposite for enhanced photocatalytic degradation of organic, microbial contaminants, and aerobic digestion of dairy wastewater. J. Clean. Prod. 258, 120588–120597 (2020)
A. Meng, S. Wu, B. Cheng, J. Yu, J. Xu, Hierarchical TiO2/Ni(OH)2 composite fibers with enhanced photocatalytic CO2 reduction performance. J. Mater. Chem. A 6, 4729–4736 (2018)
S. Palanisamy, S. Srinivasan, Electrochemical reduction of CO2 on Ni(OH)2 doped water dispersible graphene under different electrolyte conditions. SN Appl. Sci. 1, 837–843 (2019)
L. Dai, Q. Qin, P. Wang, X. Zhao, C. Hu, P. Liu, R. Qin, M. Chen, D. Ou, C. Xu, Ultrastable atomic copper nanosheets for selective electrochemical reduction of carbon dioxide. Sci. Adv. 3, e1701069–e1701078 (2017)
M.S. Akple, T. Ishigaki, P. Madhusudan, Bio-inspired honeycomb-like graphitic carbon nitride for enhanced visible light photocatalytic CO2 reduction activity, Environ. Sci. Pollut. Res., (2020) 1-15
S.N. Habisreutinger, L. Schmidt-Mende, J.K. Stolarczyk, Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 52, 7372–7408 (2013)
L. Kong, Z. Jiang, H.H. Lai, R.J. Nicholls, T. Xiao, M.O. Jones, P.P. Edwards, Unusual reactivity of visible-light-responsive AgBr–BiOBr heterojunction photocatalysts. J. Catal. 293, 116–125 (2012)
X.-J. Wen, C.-G. Niu, M. Ruan, L. Zhang, G.-M. Zeng, AgI nanoparticles-decorated CeO2 microsheets photocatalyst for the degradation of organic dye and tetracycline under visible-light irradiation. J. Colloid Interface Sci. 497, 368–377 (2017)
Y. Qi, H. Qi, C. Lu, Y. Yang, Y. Zhao, Photoluminescence and magnetic properties of β-Ni(OH)2 nanoplates and NiO nanostructures, J. Mater. Sci.: Mater. Electron, 20 (2009) 479-483
G. Lakshminarayana, S. Buddhudu, Spectral analysis of Mn2+, Co2+ and Ni2+: B2O3–ZnO–PbO glasses. Spectrochim. Spectrochimica Acta Part A 63, 295–304 (2006)
A. De Moura, R. Lima, E. Paris, M. Li, J.A. Varela, E. Longo, Formation of β-nickel hydroxide plate-like structures under mild conditions and their optical properties. J. Solid State Chem. 184, 2818–2823 (2011)
Z. Liu, R. Ma, M. Osada, K. Takada, T. Sasaki, Selective and controlled synthesis of α-and β-cobalt hydroxides in highly developed hexagonal platelets. J. Am. Chem. Soc. 127, 13869–13874 (2005)
D. Dubal, V. Fulari, C. Lokhande, Effect of morphology on supercapacitive properties of chemically grown β-Ni(OH)2 thin films. Microporous Mesoporous Mater. 151, 511–516 (2012)
M. Chatterjee, D. Enkhtuvshin, B. Siladitya, D. Ganguli, Hollow alumina microspheres from boehmite sols. J. Mater. Sci. 33, 4937–4942 (1998)
S. Tamura, A. Mori, N. Imanaka, Li+ ion conduction in (Gd, La)2O3–LiNO3 system. Solid State Ion. 175, 467–470 (2004)
M. Sulaiman, A. Rahman, N. Mohamed, Effect of water-based sol gel method on structural, thermal and conductivity properties of LiNO3–Al2O3 composite solid electrolytes. Arab. J. Chem. 10, 1147–1152 (2017)
T. Ramesh, P.V. Kamath, Synthesis of nickel hydroxide: effect of precipitation conditions on phase selectivity and structural disorder. J. Power Sources 156, 655–661 (2006)
P. Jeevanandam, Y. Koltypin, A. Gedanken, Synthesis of nanosized α-nickel hydroxide by a sonochemical method. Nano Lett. 1, 263–266 (2001)
D. Yang, R. Wang, M. He, J. Zhang, Z. Liu, Ribbon-and boardlike nanostructures of nickel hydroxide: synthesis, characterization, and electrochemical properties. J. Phys. Chem. B 109, 7654–7658 (2005)
E. Shangguan, H. Tang, Z. Chang, X.-Z. Yuan, H. Wang, Effects of different Ni(OH)2 precursors on the structure and electrochemical properties of NiOOH. Int. J. Hydrog. Energy 36, 10057–10064 (2011)
M.K. Motlagh, A. Youzbashi, F. Hashemzadeh, L. Sabaghzadeh, Structural properties of nickel hydroxide/oxyhydroxide and oxide nanoparticles obtained by microwave-assisted oxidation technique. Powder Technol. 237, 562–568 (2013)
M. Zhou, Z. Wei, H. Qiao, L. Zhu, H. Yang, T. Xia, Particle size and pore structure characterization of silver nanoparticles prepared by confined arc plasma. J. Nanomater. 2009, 3 (2009)
M.R. Hasan, S.B.A. Hamid, W.J. Basirun, S.H.M. Suhaimy, A.N.C. Mat, A sol–gel derived, copper-doped, titanium dioxide–reduced graphene oxide nanocomposite electrode for the photoelectrocatalytic reduction of CO2 to methanol and formic acid. RSC Adv. 5, 77803–77813 (2015)
C. Jiang, J. Wu, S.J. Moniz, D. Guo, M. Tang, Q. Jiang, S. Chen, H. Liu, A. Wang, T. Zhang, Stabilization of GaAs photoanodes by in situ deposition of nickel-borate surface catalysts as hole trapping sites. Sustain. Energy Fuels 3, 814–822 (2019)
Y. Ji, Y. Luo, Theoretical Study on the Mechanism of Photoreduction of CO2 to CH4 on the Anatase TiO2 (101) Surface. ACS Catal. 6, 2018–2025 (2016)
S.t. Neaţu, J.A. Maciá-Agulló, P. Concepción, H. Garcia, Gold–copper nanoalloys supported on TiO2 as photocatalysts for CO2 reduction by water, J. Am. Chem. Soc., 136 (2014) 15969-15976
J. Mao, K. Li, T. Peng, Recent advances in the photocatalytic CO2 reduction over semiconductors. Catal. Sci. Technol. 3, 2481–2498 (2013)
N. Zhang, R. Long, C. Gao, Y. Xiong, Recent progress on advanced design for photoelectrochemical reduction of CO2 to fuels. Sci. China Mater. 61, 771–805 (2018)
J.L. White, M.F. Baruch, J.E. Pander III, Y. Hu, I.C. Fortmeyer, J.E. Park, T. Zhang, K. Liao, J. Gu, Y. Yan, Light-driven heterogeneous reduction of carbon dioxide: photocatalysts and photoelectrodes. Chem. Rev. 115, 12888–12935 (2015)
M. Gattrell, N. Gupta, A. Co, A review of the aqueous electrochemical reduction of CO2 to hydrocarbons at copper. J. Electroanal. Chem. 594, 1–19 (2006)
M. Subrahmanyam, S. Kaneco, N. Alonso-Vante, A screening for the photo reduction of carbon dioxide supported on metal oxide catalysts for C1–C3 selectivity. Appl. Catal. B 23, 169–174 (1999)
N. Sasirekha, S.J.S. Basha, K. Shanthi, Photocatalytic performance of Ru doped anatase mounted on silica for reduction of carbon dioxide. Appl. Catal. B 62, 169–180 (2006)
Acknowledgments
The authors would like to thank University of Malaya and the Ministry of Higher Education Malaysia for funding this research through Grants RP020D-16SUS and FP039-2016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflicting interest and look forward to your positive response.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Che Mat, A.N., Basirun, W.J. & Shahid, M.M. Photoelectrochemical reduction of dissolved carbon dioxide over Ni(OH)2 into organic oxygenates. J IRAN CHEM SOC 18, 1363–1372 (2021). https://doi.org/10.1007/s13738-020-02117-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-020-02117-4